REVIEW
Year: 2019 I Volume: 2 I Issue: 1 I Page: 01-06
Sunscreens: The current scenario.
Kumari Pramila1 , Suvirya Swastika1 , Verma Parul1 , Pathania Sucheta1 , Shukla Prakriti1
1 Department of Dermatology, King George’s medical university (KGMU), Lucknow, Uttar Pradesh
Corresponding Author:
Dr. Swastika Suvirya
Department of Dermatology, Venereology and Leprosy, K.G. Medical University, Sitapur, Lucknow, Uttar Pradesh, India Email : swastika.p@gmail.com
Email: swastika.p@gmail.com
How to cite this article:
Kumari P, Suvirya S, Verma P, Pathania S, Shukla P.Sunscreens: The current scenario. JDA Indian Journal of Clinical Dermatology 2019;2:01-06
Abstract:
The increasing incidence of cutaneous malignancies and detrimental effects caused by ultraviolet radiation (UV) has increased the use of sunscreens. Many organic and inorganic filters are used as a measure of photoprotection, but their efficacy and safety profile still raise questions. Concerns have been raised regarding safety of nanotechnology in sunscreen, detrimental effects on environment, photocarcinogenic potential of UV filter to name a few. New developments in formulations of sunscreens along with changes in the guidelines of the regulatory bodies like The United States Food and Drug Administration and European Union have prompted us to revisit this topic. Continuous public education is still needed about proper application technique of sunscreen along with behavioral measures. In this article, the authors try to shed light on classification, pharmacological actions, various related terminologies, indications, emerging concerns and correct application technique as well as usefulness of oral sunscreens.
Key words: Ultraviolet radiation, sunscreen, photoprotection, controversies.
Introduction:
Photoprotection is crucial to prevent detrimental effects of ultraviolet (UV) radiations like photo-carcinogenesis, photoageing and photosensitivity. Sunscreens have become an integral part of not only the dermatologists’ therapeutic armamentarium but are also distributed as over the counter and cosmetic products because of increasing awareness against harmful impact of radiations. Nevertheless, we need to continue educating the general population about photo protective measures as there are concerns of inadequate application of sunscreen. In this article we attempt to compile basic aspects of sunscreens along with an effort to understand debatable issues associated with it.
Why do we need photo protection?
Ultraviolet radiations (wavelength 200-400nm) are a small part of electromagnetic radiation spectrum, classified as UVA, UVB and UVC. Most detrimental and probably extensively studied part of UV radiations is its role as a major causal factor of skin cancer.[1]Unprotected chronic sun exposure leads to development of non melanoma skin cancers.[2] Direct photochemical damage to DNA is caused by UVB leading to gene mutations by means of pyrimidine dimers and development of precancerous and cancerous lesions while UVA penetrates deeper into skin; acting indirectly at cellular level by generation of free radical species.[3]Ultraviolet radiations cause both acute and chronic effects on skin which are elaborated in [Table 1].[4], [5]
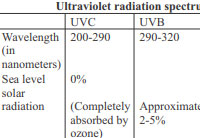 |
Table 1: Effects of ultraviolet radiations on skin |
Majority of current research and preventive strategies are centered around detrimental effects of UV radiations on skin due to its higher photon energy and relatively visible macroscopic changes. However, visible light which has been less studied so far, has a significant role in disease pathogenesis like solar urticaria, porphyria and idiopathic photodermatoses.[6] Infrared A (IRA) can cause photoageing and photocarcinogenesis through its ability to induce gene alterations.[7]Education about wholesome photo protective measures is needed to attain overall protection against solar radiations.
Sunscreen usage: indications
Sunscreens have become an integral part of day to day activity, primarily used for protection against immediate and long term ill effects of ultraviolet radiations. With current trends of leisure activities like sunbathing, tan beddings and increased awareness of skin cancers, markets are flooded with more and more sunscreens. Sunscreens are primarily indicated in prevention and management of freckling, sunburn, photoageing, photocarcinogenesis, photosensitive and photo-aggravated dermatoses. Sunscreens have become indispensable in procedure driven dermatology to prevent post procedural hyper pigmentation. Strict photoprotection is needed to prevent development or aggravation of certain dermatoses, few examples of which are tabulated [Table 2].
 |
Table 2: Indications of strict photoprotection |
Sunscreen: classification and characteristics
Sunscreen agents are broadly divided into topical and systemic. According to the Food and Drug Administration (FDA), topical sunscreens are classified as organic and inorganic, discarding the previously used terms like chemical and physical sunscreens [Figure 1]. An organic sunscreen agent is an active chemical which depending on their chemical characteristic absorbs UV radiation thereby moving into higher energy state from ground state. Depending on the fate of higher energy excited state, these are further divided into photo-stable, photo-unstable and photo-reactive.
Photo-stable sunscreen:
- > It returns to the ground state after dissipating its absorbed radiation to the environment as heat.
- > Subsequently becomes capable of absorbing UVR again (recycles
Photo-unstable sunscreen:
- > It degrades or undergoes conformational change after absorbing UV energy.
- > It cannot enter in next cycle
Photo-reactive sunscreen:
- > These agents produce free radicals by interactions of their excited state to surrounding biological molecules.
- > They can exert unwanted biological effects.
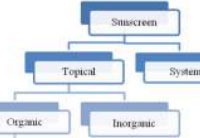 |
Figure 1: Classification of sunscreens |
Inorganic agents exert their protective function by means of reflecting, scattering or absorbing UV radiation. To enhance user’s experience their “whitening effect” may be lessened by using their micronized or ultrafine particles. [8] Incorporation of inorganic particulates has an added advantage of scattering light from the upper layers of epidermis thus enhancing the sunburn protection factor (SPF) value.[9],[10]
On the basis of photo protective quality against particular wavelengths, topical sunscreens are classified as UVA filters, UVB filters and broad spectrum sunscreens. Sunscreen agents are recognized by three widely used nomenclatures which are the US adopted name (USAN), International Nomenclature Cosmetic Ingredient (INCI) name, and trade name [Table 3].[11]
 |
Table 3: Classification and nomenclatures of topical sunscreen agents |
Sunscreen related terminology:
For a long time the labeling information over sunscreens was misleading and confusing, making claims like sweat proof, water proof etcetera. Regulatory agencies like the FDA and European Union (EU) issued guidelines to regulate labeling of sunscreens hence enabling people to choose effective agents, to provide optimal sun protection without being misguided by lucrative claims of pharmaceuticals.[12] The FDA has discarded the terms like sweatproof, waterproof and sun blocks for the same reasons. These terminologies have been replaced by water resistant and very water resistant which are adequately defined. According to the FDA, manufacturers should display the effective duration of water resistance and need of reapplication over the sunscreen label. Water resistant sunscreens should be applied before activities like swimming or excessive sweaty conditions. False claims like immediate protection after application or longstanding efficacy of product (lasting >2 hours) are forbidden if not supported by enough evidence. Consumers can confidently choose a product suitable to their needs with help of this information over label and be realistic about photo protective efficacy of the product. The FDA has also made it mandatory to test a product for both UVB and UVA protection before using the terminology ‘broad spectrum’ on the label.
- > Water resistant- withstand 2 sequential water immersion of 20 minutes (40 minutes total) while maintaining claimed SPF value.
- > Very water resistant- maintains claimed SPF after immersion in water for 20 minutes, 4 times (80 minutes total).
- > Critical wavelength- wavelength at which 90% of the total area under the absorbance curve occurs.
- > Broad spectrum- sunscreen with critical wavelength = 370nm with UVA protection factor = 4.
The sunburn protection factor is measured as a ratio of the amount of UVR necessary to burn the protected skin (with sunscreen) to that required to burn the same non protected skin (without sunscreen) with all other parameters being constant. The required amount of UVR is known as MED (minimal erythema dose) which is defined as the minimum UV dose required to produce perceptible erythema of the skin with well defined margins at 16 to 24 hours after UV irradiation.[12] This means that a SPF 30 sunscreen protected skin can tolerate the same amount of UV radiation 30 times more than the unprotected skin. The grading of SPF is done as low (SPF 2-15), medium (SPF 15-30), high (SPF 30-50) and highest (SPF >50).
For measurement of UVA protection, various testing methods have been developed by regulatory bodies in Japan, the European Union (EU), United Kingdom (UK) and Australia.
UVA protection indices:
- 1. Australian/New Zealand Standard: In vitro method. 8-µm and 20-µm thick layers of the product should not transmit greater than 10% and 1% of radiation of 320 to 360 nm, respectively.
- 2. Japanese standard (persistent pigment darkening): In vivo test. It is a ratio of UVA required to induce persistent pigment darkening (PPD) 2 to 24 hrs after irradiation in sunscreen protected skin to unprotected skin. Ratings- PA+, PA++, PA+++, PA++++ (PA= protection grade from UVA).
- 3. European Union guidelines: Based on PPD method. It requires UVA protection factor to be = 1/3 of labeled SPF.
- 4. Boots star rating system (United Kingdom): In vitro method. It measures ratio of UVA absorbance to mean UVB absorbance.
Since 2012, the FDA has mandated that only sunscreens having SPF=15 and critical wavelength =370 nm, can display their claim about protection against development of skin cancers.[12]
Ideal sunscreen:
The notion of ideal sunscreen is that it should provide maximum photo-protection while maintaining its compliance quality. An ideal sunscreen should possess ‘spectral homeostasis’, which refers to uniform protection against UVA and UVB radiations spectrum.[13]The characteristics of an ideal sunscreen also include cosmetic acceptability and non-irritant nature, among others [Figure 2].
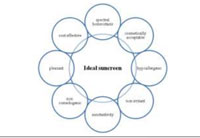 |
Figure 2: Ideal sunscreen |
Guidelines for application:
Apart from SPF and substantivity, the other major factor that determines the protective efficacy of a given sunscreen is its adequate application. There is a lack of basic knowledge about correct application, leading to attainment of only one third to quarter of the recommended dose of photoprotection.[14]Sunscreen needs to be applied about half an hour before going outdoor, in a density of at least 2 mg per square centimeter which can be further simplified by following the “Tea spoon rule.”[15] The rule recommends application of 1 teaspoon of sunscreen to the face/head/neck, 1 teaspoon to each upper limb, 2 teaspoons to trunk (front and back), and 2 teaspoons to each lower limb. The protective efficacy can be boosted significantly by reapplication 20 minutes later, thus correcting the areas of inadequate application.[6]
Sunscreen should always be used along with complete photoprotection package which includes seeking shade, protective clothing, wide-brimmed hat and sunglasses. A continuous effort in public education is required concerning its correct application, reapplication and prevention of unnecessary sun exposure.
Systemic sunscreens:
There is a growing interest in use of orally active ingredients to counter the inherent photochemical reaction, by decreasing free radical injuries. Antioxidants act by reversing the oxidative stress developed by UVR and infrared radiations. Their protective efficacies have been reported in various studies. Still larger studies are needed to confirm their level of protection and long term safety profile. Carotene, antimalarials, vitamin A, C and E, green tea extracts, selenium, retinol and many more have been reported to have photo protective qualities.[8] Three widely used and studied systemic sunscreens are polypodium leucotomos extract, afamelanotide and nicotinamide.
Polypodium leucotomos, belonging to fern group of plants, has been shown to decrease psoralen plus UVA and UVB phototoxicity.[16],[17] The oral administration of the drug as 240 mg twice a day provides a SPF value of 3-8, and has been shown it to be protective against erythemogenic spectra of UVR.[18]
The melanocortin-1 receptor agonist, afamelanotide exerts its photoprotection via increasing synthesis of eumelanin. It has been approved as an adjuvant in adult patients of erythropoietic protoporphyria by the European Union to prevent phototoxicity and has to be administered as 16 mg subcutaneously every 2 months.[19]
When taken orally, nicotinamide (active form of niacin) has been shown to be photo protective by enhancing intracellular adenosine triphosphate,[20] DNA repair and boosting cell energy.[21],[22] Its broad photoprotective effect against development of premalignant lesions is being studied currently.
Sunscreens related controversies:
1. Hormonal effect: There are rising concerns about possible hormonal disruption of sunscreen agents benzophenones specially oxybenzone, which is in widespread use since 1970s. Initial in vitro studies have shown its antiandrogenic and estrogenic effects.[23],[24] However in recent in-vivo studies the claims were not substantiated about cause and effects.[25] Careful observation and further human studies are needed to clarify this aspect.
2. Role of antioxidants: Nowadays, many sunscreen manufactures are using antioxidants beside active ingredient to decrease the adverse effects of free radicals, generated by UV exposure. In vivo studies have shown decreased matrix metalloproteinase-1 activity and less pigment induction with use of stabilized antioxidants[26] but a recent study has proven otherwise because of lack of stability of antioxidants used in sunscreens.[27]
3. Nanoparticulate sunscreen: There are concerns about rising use of nanotechnology in sunscreens (to make it cosmetically elegant) as nanoparticles can produce free radicals on UV irradiation.[28] Various studies have shown that the confinement of nanoparticles is limited to stratum corneum. In addition, the use of coated nanoparticles has made it safe for usage in humans.[29] However application at sites with severely impaired barrier function should be minimized till further data are available.
4. Photocarcinogenic potential of retinyl palmitate (RP): It is a storage form of vitamin A which was approved by the FDA to use in cosmetics and edibles. In view of rising concerns about photo carcinogenicity of compound, Wang et al[30] concluded in-depth review on this topic in addition to a large in-vivo study conducted by the FDA. None of the above could establish conclusive evidence about photo-carcinogenicity of RP. Also there is long standing history of safety profile of the product in humans.[11]
5. Vitamin D deficiency: There have been concerns of vitamin D deficiency regarding universal use of sunscreens as ninety percent of vitamin D production in skin happens as a result of UV exposure. However, review of literature by Norval et al[31] concluded that deficiency doesn’t occur with normal usage of sunscreen most likely due to insufficient application of sunscreen by most individuals. Unprotected exposure to UVR is not recommended to obtain vitamin D and supplementation in individuals at risk is advised as supported by the Institute of Medicine.[19]
6. Pediatric population and sunscreen: Although there are no deleterious effects of sunscreen documented with use in early age, still it is advisable to use sunscreens containing inorganic filters over exposed area, in adjunction to other sun protective measures.[11]
7. Environmental issues: The effects of organic sunscreens on environment have become a burning issue since water sources are found to be contaminated with sunscreens specially oxybenzone in various studies.[32] They can react to chlorine in pools to form brominated transformation products which are hard to remove by usual water filters. In addition to this, studies have indicated possible role of oxybenzone in coral bleaching.[33] The adverse impact is still being studied.
8. Role of higher SPF: The labeling restriction of SPF value more than 50 as SPF50+ by the US FDA has created a stir about significance of higher SPF. Various studies have shown that sunscreen with higher SPF has provided better protection against sunburn and UV induced phototoxicity.[34] The higher SPF can compensate the efficacy of a product in actual use as there are enough evidences to suggest that on an average only one third amount of a given SPF is attained due to insufficient and improper application.[14]
9. Sunscreen and special populations There has always been confusion about prescription of sunscreen in Fitzpatrick skin type IV to VI as these skin types are less prone to sun damage because of inherent protective quality of melanin. However, enough evidence of photodamage including photo ageing has been documented.[35] In addition, malignant melanoma carries a poorer prognosis in POC (people of colour) despite low prevalence. Hence it is recommended to use regular sunscreen with other sun protective measures the same way as in other skin types. Broad spectrum sunscreen with SPF =30, specially containing inorganic filters are better suited for POC as they are more acceptable.[36]
Use of regular sunscreen with other photo protective behavior measures should be followed in patients of organ transplantation and dialysis to decrease the risk of premalignant and malignant changes in skin.[37]
10. Sunscreen and cosmetics: Besides conventional sunscreen cream and lotion, now a day active ingredients are seen as foam, gel, mousse, spray, pastes, oils, butters, sticks and ointments. The pharmaceuticals and cosmetic giants are using sunscreen in range of over the counter products and cosmetics like foundation, compact, shampoo, lipstick, lip balm and wipes, with claim of varied SPF. The efficacy of these are not well established and not approved by the FDA yet. The spray forms are being promoted as convenient to use in children and over relatively non accessible sites like back in adults. For acne prone skin, gel and sprays forms are tolerated well. Sprays have also shown to retain active compounds to superficial layers of epidermis thus decreasing the risk of deeper penetration.[38]
References:
1. Gilchrest BA, Eller MS, Geller AC, Yaar M. The pathogenesis of melanoma induced by ultraviolet radiation. N Engl J Med 1999;340:1341-48.
2. van Kranen HJ, Westerman A, Berg RJ, Kram N, van Kreijl CF, Wester PW, et al. Dose-dependent effects of UVB-induced skin carcinogenesis in hairless p53 knockout mice. Mutat Res 2005;571:81-90.
3. Rhodes LE. Topical and systemic approaches for protection against solar radiation-induced skin damage. Dermatol Clin 1998;16:75-82.
4. Forestier S. Rationale for sunscreen development. J Am Acad Dermatol 2008;58:133-8.
5. Young AR. Photobiology. In: Griffiths C, Barker J, Bleiker T, Chalmers R, Creamer D, editors. Rook’s Textbook of Dermatology. 9th ed. West Sussex (UK): Wiley-Blackwell; 2016. p.9.5-9.7.
6. Rai R, Shanmuga SC, Srinivas C R. Update on photoprotection. Indian J Dermatol 2012;57:335-42.
7. Calles C, Schneider M, Macaluso F, Benesova T, Krutmann J, Schroeder P. Infrared A radiation influences the skin fibroblast transcriptome: mechanisms and consequences. J Invest Dermatol 2010;130:1524-36.
8. Kaimal S, Abraham A. Sunscreens. Indian J Dermatol Venereol Leprol 2011;77:238-43.
9. Lademann J, Schanzer S, Jacobi U, Schaefer H, Pflücker F, Driller H, et al. Synergy effects between organic and inorganic UV filters in sunscreens. J Biomed Opt 2005;10:14008.
10. Vergou T, Antoniou C, Patzelt A, Richter H, Schanzer S, Sterry W, et al. Transfer of ultraviolet photon energy into fluorescent light in the visible path represents a new and efficient protection mechanism of sunscreens. J Biomed Opt 2011;16:105001.
11. Jansen R, Osterwalder U, Wang SQ, Burnett M, Lim HW. Photoprotection: part II. Sunscreen: development, efficacy, and controversies. J Am Acad Dermatol 2013;69:867-e1.
12. Latha MS, Martis J, Shobha V, Shinde RS, Bangera S, Krishnankutty B, et al. Sunscreening agents: a review. J Clin Aesthet Dermatol.2013;6:16-26.
13. Diffey B. The need for sunscreens with broad spectrum protection. In: Urbach F, editor. Biological responses to ultraviolet a radiation. Overland Park (KS): Valdenmar Publication Co; 1992. pp. 321-8.
14. Wang SQ, Dusza SW. Assessment of sunscreen knowledge: a pilot survey. Br J Dermatol 2009;161:28-32.
15. Isedeh P, Osterwalder U, Lim HW. Teaspoon rule revisited: proper amount of sunscreen application. Photodermatol Photoimmunol Photomed 2013;29:55-6.
16. Gonzalez S, Pathak MA, Cuevas J, Villarrubia VG, Fitzpatrick TB. Topical or oral administration with an extract of Polypodium leucotomos prevents acute sunburn and psoralen-induced phototoxic reactions as well as depletion of Langerhans cells in human skin. Photodermatol Photoimmunol Photomed 1997;13:50-60.
17. Middelkamp-Hup MA, Pathak MA, Parrado C, Garcia-Caballero T, Rius-Díaz F, Fitzpatrick TB, et al. Orally administered Polypodium leucotomos extract decreases psoralen-UVA induced phototoxicity, pigmentation, and damage of human skin. J Am Acad Dermatol 2004;50:41-9.
18. Nestor MS, Berman B, Swenson N. Safety and efficacy of oral Polypodium leucotomos extract in healthy adult subjects. J Clin Aesthet Dermatol 2015;8:19-23.
19. Lim HW, Arellano-Mendoza MI, Stengel F. Current challenges in photoprotection. J Am Acad Dermatol 2017;76:91-9.
20. Park J, Halliday GM, Surjana D, Damian DL. Nicotinamide prevents ultraviolet radiation-induced cellular energy loss. Photochem Photobiol 2010;86:942-48.
21. Surjana D, Halliday GM, Damian DL. Nicotinamide enhances repair of ultraviolet radiation-induced DNA damage in human keratinocytes and ex vivo skin. Carcinogenesis 2013;34:1144-49.
22. Thompson BC, Surjana D, Halliday GM, Damian DL. Nicotinamide enhances repair of ultraviolet radiation-induced DNA damage in primary melanocytes. Exp Dermatol 2014;23:509-11.
23. Schlumpf M, Schmid P, Durrer S, Conscience M, Maerkel K, Henseler M, et al. Endocrine activity and developmental toxicity of cosmetic UV filters- an update. Toxicology 2004;205:113-22.
24. Ma R, Cotton B, Lichtensteiger W, Schlumpf M. UV filters with antagonistic action at androgen receptors in the MDA-kb2 cell transcriptional-activation assay. Toxicol Sci 2003;74:43-50.
25. Janjua NR, Mogensen B, Andersson AM, Petersen JH, Henriksen M, Skakkebaek NE, et al. Systemic absorption of the sunscreens benzophenone-3, octyl-methoxycinnamate, and 3-(4-methyl-benzylidene) camphor after whole-body topical application and reproductive hormone levels in humans. J Invest Dermatol 2004;123:57-61.
26. Matsui MS, Hsia A, Miller JD, Hanneman K, Scull H, Cooper KD, et al. Non-sunscreen photoprotection: antioxidants add value to a sunscreen. J Investig Dermatol Symp Proc 2009;14:56-9.
27. Wang SQ, Osterwalder U, Jung K. Ex vivo evaluation of radical sun protection factor in popular sunscreens with antioxidants. J Am Acad Dermatol 2011;65:525-30.
28. Newman MD, Stotland M, Ellis JI. The safety of nanosized particles in titanium dioxide and zinc oxide based sunscreens. J Am Acad Dermatol 2009;61:685-92.
29. Livraghi S, Corazzari I, Paganini MC, Ceccone G, Giamello E, Fubini B, et al. Decreasing the oxidative potential of TiO(2) nanoparticles through modification of the surface with carbon: a new strategy for the production of safe UV filters. Chem Commun 2010;46:8478-80.
30. Wang SQ, Dusza SW, Lim HW. Safety of retinylpalmitate in sunscreens: a critical analysis. J Am Acad Dermatol 2010;63:903-6.
31. Norval M, Wulf HC. Does chronic sunscreen use reduce vitamin D production to insufficient levels? Br J Dermatol 2009;161:732-6.
32. Cuderman P, Heath E. Determination of UV filters and antimicrobial agents in environmental water samples. Anal Bioanal Chem 2007;387:1343-50.
33. DiNardo JC, Downs CA. Dermatological and environmental toxicological impact of the sunscreen ingredient oxybenzone/ benzophenone-3. J Cosmet Dermatol 2018;17:15-9.
34. Williams JD, Maitra P, Atillasoy E, Wu MM, Farberg AS, Rigel DS. SPF 100+ sunscreen is more protective against sunburn than SPF 50+ in actual use: Results of a randomized, double-blind, split-face, natural sunlight exposure clinical trial. J Am Acad Dermatol 2018;78:902-10.
35. Del Bino S, Bernerd F. Variations in skin color and the biological consequences of ultraviolet radiation exposure. Br J Dermatol 2013;169:33-40.
36. Agbai ON, Buster K, Sanchez M, Hernandez C, Kundu RV, Chiu M, et al. Skin cancer and photoprotection in people of color: a review and recommendations for physicians and the public. J Am Acad Dermatol 2014;70:748-62.
37. Ulrich C, Degen A, Patel MJ, Stockfleth E. Sunscreens in organ transplant patients. Nephrol Dial Transplant 2008;23:1805-8.
38. Durand L, Habran N, Henschel V, Amighi K. In vitro evaluation of the cutaneous penetration of sprayable sunscreen emulsions with high concentrations of UV filters. Int J Cosmet Sci2009;31:279-92.

CTGF was a plausible candidate to participate in mediating survival, as it has been implicated as such a signal downstream of S1p signalling and it is expressed in the developing pancreas in both the epithelium and the mesenchyme S5 Fig 35 buy cheap doxycycline
If glipizide is exposed to extremely hot or cold temperatures, it s possible for the chemical structure of the drug to be affected cialis generic reviews
viagra connect near me The use of a combination of a loop diuretic eg, furosemide and the replacement of urine output with a solution that contains a higher Na concentration ie, 3 sodium chloride solution can be dramatically successful in some patients
order arimidex 1mg pills purchase anastrozole without prescription generic arimidex
Very nice post. I just stumbled upon your blog and wanted to say that I’ve really enjoyed browsing your blog posts. In any case I’ll be subscribing to your feed and I hope you write again soon!
Римская аристократия всегда без
особенного сопротивления уступала народу почести; но когда дело коснулось
имуществ, она стала защищать их так упорно
что народу для удовлетворения своего желания пришлось прибегать
к чрезвычайным мерам. Психологичные книги.
Hi visit my site porn
Your article gave me a lot of inspiration, I hope you can explain your point of view in more detail, because I have some doubts, thank you. 20bet
https://amoxil.icu/# azithromycin amoxicillin
buying generic clomid price can i purchase cheap clomid without rx – order generic clomid without insurance
cost of generic clomid without insurance: can i purchase generic clomid – get clomid without rx
http://ciprofloxacin.life/# cipro ciprofloxacin
http://amoxil.icu/# amoxicillin 500mg buy online canada
prednisone 20 mg tablets coupon: buy prednisone 20mg – order prednisone online no prescription
generic for doxycycline doxy how to order doxycycline
where to get zithromax: zithromax drug – zithromax drug
http://lisinoprilbestprice.store/# lisinopril price 10 mg
п»їcytotec pills online: п»їcytotec pills online – buy cytotec over the counter
http://cytotec.icu/# cytotec pills online
п»їcytotec pills online: buy cytotec online fast delivery – buy cytotec pills online cheap
https://nolvadex.fun/# tamoxifen depression
buy generic doxycycline doxycycline without a prescription doxycycline 50 mg
http://doxycyclinebestprice.pro/# generic for doxycycline
doxycycline hyclate 100 mg cap: odering doxycycline – order doxycycline online
tamoxifen hair loss: tamoxifen menopause – where to buy nolvadex
http://zithromaxbestprice.icu/# zithromax 1000 mg online
doxycycline 50 mg: buy cheap doxycycline – doxycycline order online
https://cytotec.icu/# Cytotec 200mcg price
nolvadex side effects tamoxifen citrate nolvadex for sale
cytotec buy online usa: Cytotec 200mcg price – buy cytotec over the counter
https://lisinoprilbestprice.store/# lisinopril tablets india
doxycycline 100mg capsules: doxy 200 – doxycycline 100mg capsules
lisinopril 40 mg coupon: lisinopril tab 5 mg price – buy 20mg lisinopril
nolvadex side effects: tamoxifen breast cancer – tamoxifen cyp2d6
https://lisinoprilbestprice.store/# prinivil 40 mg
doxycycline 100mg buy doxycycline hyclate 100mg without a rx doxycycline monohydrate
tamoxifen alternatives premenopausal: aromatase inhibitors tamoxifen – tamoxifen
https://lisinoprilbestprice.store/# lisinopril 500 mg
https://nolvadex.fun/# buy nolvadex online
cheapest online pharmacy india: India pharmacy of the world – indian pharmacy paypal indiapharm.llc
mexican rx online Purple Pharmacy online ordering mexican mail order pharmacies mexicopharm.com
http://mexicopharm.com/# best online pharmacies in mexico mexicopharm.com
india online pharmacy: Online India pharmacy – indian pharmacy online indiapharm.llc
canada pharmacy online legit: Canadian online pharmacy – canadian world pharmacy canadapharm.life
http://indiapharm.llc/# Online medicine home delivery indiapharm.llc
http://indiapharm.llc/# buy medicines online in india indiapharm.llc
buying from online mexican pharmacy: mexican pharmacy – mexico drug stores pharmacies mexicopharm.com
online shopping pharmacy india: cheapest online pharmacy india – indian pharmacy paypal indiapharm.llc
canadian pharmacy mall canadian pharmacy online reviews canadian compounding pharmacy canadapharm.life
mexican border pharmacies shipping to usa: Purple Pharmacy online ordering – buying prescription drugs in mexico online mexicopharm.com
http://canadapharm.life/# best canadian pharmacy canadapharm.life
india pharmacy mail order: India Post sending medicines to USA – reputable indian online pharmacy indiapharm.llc
https://indiapharm.llc/# pharmacy website india indiapharm.llc
online shopping pharmacy india: top 10 online pharmacy in india – reputable indian pharmacies indiapharm.llc
п»їbest mexican online pharmacies: Medicines Mexico – mexican online pharmacies prescription drugs mexicopharm.com
https://canadapharm.life/# canadian 24 hour pharmacy canadapharm.life
mexican online pharmacies prescription drugs: Purple Pharmacy online ordering – best online pharmacies in mexico mexicopharm.com
mexican pharmaceuticals online: mexican pharmacy – mexican drugstore online mexicopharm.com
indian pharmacies safe India Post sending medicines to USA top 10 online pharmacy in india indiapharm.llc
http://indiapharm.llc/# indianpharmacy com indiapharm.llc
online shopping pharmacy india: indian pharmacy to usa – mail order pharmacy india indiapharm.llc
indian pharmacy paypal: Medicines from India to USA online – buy medicines online in india indiapharm.llc
https://mexicopharm.com/# pharmacies in mexico that ship to usa mexicopharm.com
buying prescription drugs in mexico: mexican pharmacy – mexico pharmacies prescription drugs mexicopharm.com
https://indiapharm.llc/# best india pharmacy indiapharm.llc
Kamagra 100mg price: kamagra oral jelly – super kamagra
tadalafil 20mg canada Tadalafil 20mg price in Canada tadalafil tablets 20 mg buy
https://edpillsdelivery.pro/# best ed treatment pills
Levitra 20 mg for sale: Buy generic Levitra online – Levitra 10 mg buy online
https://sildenafildelivery.pro/# sildenafil 50 mg tablet cost
herbal ed treatment: best over the counter ed pills – ed drugs list
http://tadalafildelivery.pro/# tadalafil 20 mg buy online
https://kamagradelivery.pro/# Kamagra Oral Jelly
men’s ed pills: erection pills over the counter – buy ed pills online
sildenafil buy usa can you buy sildenafil otc sildenafil buy cheap
п»їkamagra: kamagra oral jelly – sildenafil oral jelly 100mg kamagra
https://edpillsdelivery.pro/# best erection pills
best drug for ed: pills erectile dysfunction – mens ed pills
http://tadalafildelivery.pro/# tadalafil 10 mg canadian pharmacy
Buy Vardenafil 20mg online: Generic Levitra 20mg – Levitra generic best price
buy viagra over the counter canada
https://levitradelivery.pro/# Levitra tablet price
https://kamagradelivery.pro/# cheap kamagra
Cheap Levitra online: Levitra best price – Vardenafil online prescription
tadalafil canadian pharmacy online: Tadalafil 20mg price in Canada – tadalafil online price
Levitra online USA fast Buy Vardenafil 20mg Cheap Levitra online
https://kamagradelivery.pro/# Kamagra Oral Jelly
buy Levitra over the counter: Cheap Levitra online – Levitra 20 mg for sale
http://amoxil.guru/# amoxicillin 500 mg online
https://prednisone.auction/# prednisone 50 mg for sale
https://clomid.auction/# how to buy cheap clomid without prescription
paxlovid for sale paxlovid buy paxlovid covid
http://stromectol.guru/# price of ivermectin
paxlovid india: paxlovid best price – paxlovid india
https://stromectol.guru/# ivermectin price canada
http://clomid.auction/# can i get generic clomid prices
paxlovid cost without insurance buy paxlovid online paxlovid india
https://prednisone.auction/# prednisone 5 mg tablet without a prescription
http://stromectol.guru/# ivermectin tablets order
ivermectin drug: buy ivermectin online – order minocycline 50 mg online
https://clomid.auction/# cost clomid pills
https://clomid.auction/# how to get clomid pills
Paxlovid buy online Buy Paxlovid privately paxlovid cost without insurance
http://prednisone.auction/# prednisone rx coupon
http://prednisone.auction/# prednisone 2.5 mg
buy clomid without prescription: order clomid without a prescription – how can i get cheap clomid online
https://prednisone.auction/# prednisone pill
zithromax 500 without prescription: Azithromycin 250 buy online – zithromax online usa
buy zithromax online: zithromax best price – zithromax generic price
http://misoprostol.shop/# buy cytotec over the counter
buy cheap lisinopril 40mg cheapest lisinopril lisinopril 100 mg
https://azithromycin.store/# how to get zithromax
buy cheap propecia pill: Buy finasteride 1mg – propecia cost
https://finasteride.men/# buy cheap propecia for sale
https://misoprostol.shop/# Abortion pills online
buy cytotec in usa: cheap cytotec – purchase cytotec
lisinopril 125 mg: cheapest lisinopril – lisinopril 10mg daily
http://azithromycin.store/# where to buy zithromax in canada
buy generic propecia without insurance: Finasteride buy online – order propecia without a prescription
https://misoprostol.shop/# cytotec buy online usa
https://finasteride.men/# order propecia pills
http://finasteride.men/# buy cheap propecia pill
order propecia without dr prescription: buy propecia – cheap propecia no prescription
cost of cheap propecia prices: Finasteride buy online – cost of generic propecia online
zithromax 1000 mg online: buy zithromax over the counter – zithromax 600 mg tablets
https://azithromycin.store/# zithromax capsules australia
cost generic propecia online buy propecia buy propecia without a prescription
lasix online: Buy Furosemide – lasix 100 mg
http://azithromycin.store/# generic zithromax over the counter
lisinopril 20 mg price online: High Blood Pressure – cheap lisinopril 40 mg
https://lisinopril.fun/# lisinopril 20 mg online
https://lisinopril.fun/# zestril 20 mg tab
https://misoprostol.shop/# Cytotec 200mcg price
zithromax tablets for sale: average cost of generic zithromax – zithromax cost australia
cytotec pills buy online: Misoprostol best price in pharmacy – buy cytotec pills
order generic propecia no prescription Buy Finasteride 5mg cost of propecia online
how to get zithromax online: Azithromycin 250 buy online – zithromax online australia
http://azithromycin.store/# zithromax drug
buy cytotec in usa: buy misoprostol – buy cytotec over the counter
http://finasteride.men/# order generic propecia for sale
http://lisinopril.fun/# lisinopril price in india
lasix 100 mg Buy Lasix furosemide 100mg
cost of generic propecia without a prescription: buy propecia – buy propecia pill
zithromax for sale cheap: buy zithromax without presc – where can i get zithromax
https://misoprostol.shop/# order cytotec online
http://furosemide.pro/# lasix 100 mg
lasix generic: Buy Furosemide – lasix side effects
kamagra senza ricetta in farmacia sildenafil 100mg prezzo viagra generico in farmacia costo
http://kamagraitalia.shop/# top farmacia online
acquisto farmaci con ricetta: farmacia online piu conveniente – п»їfarmacia online migliore
comprare farmaci online con ricetta: kamagra oral jelly consegna 24 ore – farmacia online piГ№ conveniente
https://kamagraitalia.shop/# migliori farmacie online 2023
http://kamagraitalia.shop/# farmacia online più conveniente
https://avanafilitalia.online/# farmacia online miglior prezzo
viagra originale in 24 ore contrassegno: viagra prezzo – miglior sito per comprare viagra online
farmacie online affidabili: avanafil prezzo in farmacia – migliori farmacie online 2023
https://sildenafilitalia.men/# viagra subito
farmacie on line spedizione gratuita cialis prezzo farmaci senza ricetta elenco
comprare farmaci online all’estero: farmacia online – acquistare farmaci senza ricetta
farmacia online migliore: farmacia online miglior prezzo – acquistare farmaci senza ricetta
https://tadalafilitalia.pro/# farmacia online senza ricetta
cialis farmacia senza ricetta: viagra consegna in 24 ore pagamento alla consegna – viagra 100 mg prezzo in farmacia
http://avanafilitalia.online/# п»їfarmacia online migliore
http://farmaciaitalia.store/# migliori farmacie online 2023
farmaci senza ricetta elenco Farmacie a roma che vendono cialis senza ricetta comprare farmaci online con ricetta
п»їfarmacia online migliore: Tadalafil generico – п»їfarmacia online migliore
cialis farmacia senza ricetta: viagra generico – viagra 100 mg prezzo in farmacia
https://farmaciaitalia.store/# farmacia online miglior prezzo
comprare farmaci online con ricetta: Farmacie a roma che vendono cialis senza ricetta – farmacia online piГ№ conveniente
farmacie online affidabili kamagra oral jelly farmacia online
https://kamagraitalia.shop/# comprare farmaci online all’estero
https://farmaciaitalia.store/# farmacia online migliore
http://sildenafilitalia.men/# viagra generico prezzo piГ№ basso
comprare farmaci online all’estero: cialis generico consegna 48 ore – migliori farmacie online 2023
https://tadalafilitalia.pro/# farmacia online migliore
http://indiapharm.life/# indian pharmacy
top 10 pharmacies in india: indianpharmacy com – best online pharmacy india
canadian online pharmacy canadian pharmacy 365 legitimate canadian pharmacy
https://indiapharm.life/# top 10 pharmacies in india
http://mexicanpharm.store/# mexican pharmaceuticals online
mexico pharmacies prescription drugs: buying from online mexican pharmacy – mexico pharmacies prescription drugs
india online pharmacy: pharmacy website india – indian pharmacy paypal
https://indiapharm.life/# india pharmacy
best online pharmacy india: pharmacy website india – online pharmacy india
http://mexicanpharm.store/# best online pharmacies in mexico
pharmacies in mexico that ship to usa: mexico drug stores pharmacies – buying prescription drugs in mexico
buy medicines online in india: world pharmacy india – п»їlegitimate online pharmacies india
mexican online pharmacies prescription drugs mexican drugstore online reputable mexican pharmacies online
http://mexicanpharm.store/# mexican drugstore online
https://mexicanpharm.store/# pharmacies in mexico that ship to usa
indianpharmacy com: pharmacy website india – pharmacy website india
indian pharmacies safe: best online pharmacy india – indian pharmacies safe
pharmacies in canada that ship to the us: canadian pharmacy service – canadian pharmacy world reviews
http://mexicanpharm.store/# mexican drugstore online
canada drug pharmacy: cheap canadian pharmacy – my canadian pharmacy
https://mexicanpharm.store/# buying prescription drugs in mexico
buying prescription drugs in mexico online: mexico pharmacy – mexican drugstore online
http://mexicanpharm.store/# mexican pharmaceuticals online
mexican mail order pharmacies reputable mexican pharmacies online mexico drug stores pharmacies
http://mexicanpharm.store/# mexican rx online
п»їbest mexican online pharmacies: reputable mexican pharmacies online – mexican pharmacy
top 10 pharmacies in india: buy medicines online in india – indian pharmacy paypal
https://mexicanpharm.store/# mexican pharmacy
top 10 online pharmacy in india: reputable indian pharmacies – Online medicine home delivery
best online pharmacies in mexico: mexican online pharmacies prescription drugs – buying prescription drugs in mexico
https://mexicanpharm.store/# buying prescription drugs in mexico online
mexican online pharmacies prescription drugs: mexico drug stores pharmacies – mexico drug stores pharmacies
http://canadapharm.shop/# vipps canadian pharmacy
reputable indian online pharmacy Online medicine home delivery indian pharmacy paypal
best canadian online pharmacy reviews: canadian pharmacy ltd – the canadian pharmacy
http://canadapharm.shop/# adderall canadian pharmacy
https://prednisonepharm.store/# prednisone 20mg buy online
otc prednisone cream: buy prednisone online india – prednisone 20mg tab price
buy misoprostol over the counter buy cytotec online fast delivery cytotec online
http://prednisonepharm.store/# price of prednisone 5mg
Their online refill system is straightforward https://nolvadex.pro/# does tamoxifen make you tired
buying clomid tablets: how can i get generic clomid online – buying generic clomid without prescription
Their global pharmacists’ network is commendable https://prednisonepharm.store/# prednisone otc uk
http://prednisonepharm.store/# average price of prednisone
https://cytotec.directory/# purchase cytotec
how can i get generic clomid without insurance: get cheap clomid without dr prescription – cost clomid pills
They always offer alternatives and suggestions https://cytotec.directory/# buy cytotec
http://cytotec.directory/# п»їcytotec pills online
zithromax online pharmacy canada: where can i purchase zithromax online – zithromax for sale 500 mg
They always offer alternatives and suggestions https://nolvadex.pro/# tamoxifen
can you buy cheap clomid without rx where to buy cheap clomid without rx cost cheap clomid online
http://clomidpharm.shop/# can you get cheap clomid price
The gold standard for international pharmaceutical services http://zithromaxpharm.online/# zithromax online pharmacy canada
cost cheap clomid price: generic clomid online – where to get cheap clomid for sale
https://clomidpharm.shop/# generic clomid without a prescription
http://prednisonepharm.store/# can i buy prednisone online without a prescription
The best place for quality health products https://prednisonepharm.store/# prednisone 40mg
tamoxifenworld: how to prevent hair loss while on tamoxifen – tamoxifen breast cancer
http://cytotec.directory/# order cytotec online
Their free health check-ups are a wonderful initiative http://nolvadex.pro/# tamoxifen buy
tamoxifen citrate pct tamoxifen cancer is nolvadex legal
zithromax online paypal: where can you buy zithromax – zithromax 500 without prescription
https://zithromaxpharm.online/# zithromax over the counter uk
Their staff is so knowledgeable and friendly http://nolvadex.pro/# tamoxifen 20 mg tablet
prednisone 5mg price: prednisone 5 mg – prednisone cost in india
http://nolvadex.pro/# tamoxifen and bone density
http://reputablepharmacies.online/# the discount pharmacy
erectile dysfunction medications best erectile dysfunction pills ed treatments
canadian pharmacies that deliver to the us https://reputablepharmacies.online/# buy canadian pharmacy
legitimate mexican pharmacy online
canadian mail order drug companies: canadian pharmacy without a prescription – cheap drugs online
best pill for ed: what is the best ed pill – gnc ed pills
real viagra without a doctor prescription non prescription erection pills buy prescription drugs
http://edwithoutdoctorprescription.store/# prescription without a doctor’s prescription
viagra without doctor prescription amazon: prescription drugs without doctor approval – meds online without doctor prescription
https://edwithoutdoctorprescription.store/# real viagra without a doctor prescription usa
buy prescription drugs without doctor canada drugs online mail order prescription drugs from canada
non prescription erection pills: best ed drug – ed meds online
http://edwithoutdoctorprescription.store/# tadalafil without a doctor’s prescription
best canadian online pharmacy viagra https://reputablepharmacies.online/# canadian pharmacy 365
online pharmacies reviews
canadian drug prices on line pharmacy with no prescriptions verified canadian pharmacies
100mg viagra without a doctor prescription: prescription drugs online without – levitra without a doctor prescription
http://reputablepharmacies.online/# overseas no rx drugs online
cialis order
viagra without doctor prescription viagra without a doctor prescription prescription drugs online without
ed meds online without prescription or membership: viagra without a prescription – prescription drugs online without doctor
natural ed remedies ed pills otc over the counter erectile dysfunction pills
male ed drugs: drugs for ed – ed drugs
cialis alternative
http://edpills.bid/# ed dysfunction treatment
https://edwithoutdoctorprescription.store/# ed prescription drugs
buy prescription drugs without doctor prescription drugs online without doctor buy prescription drugs from canada
canadian prescriptions online: best internet pharmacies – canadian pharmacy world reviews
https://edpills.bid/# non prescription ed pills
cheapest ed pills online pills for ed cheap erectile dysfunction pill
canadian prescriptions: online pharmacies canadian – canadian pharma companies
canada drugs online review: pharmacy online – medicine canada
https://edwithoutdoctorprescription.store/# buy prescription drugs from india
new ed treatments herbal ed treatment best ed pills online
http://mexicanpharmacy.win/# medicine in mexico pharmacies mexicanpharmacy.win
canada pharmacy online no script
http://indianpharmacy.shop/# reputable indian pharmacies indianpharmacy.shop
best online pharmacy india Best Indian pharmacy buy prescription drugs from india indianpharmacy.shop
mexico pharmacy: mexican drugstore online – mexican pharmacy mexicanpharmacy.win
top 10 online pharmacy in india indian pharmacy best online pharmacy india indianpharmacy.shop
http://indianpharmacy.shop/# indian pharmacy online indianpharmacy.shop
mexican online pharmacies prescription drugs: mexican online pharmacies prescription drugs – mexican border pharmacies shipping to usa mexicanpharmacy.win
http://canadianpharmacy.pro/# canadian pharmacy checker canadianpharmacy.pro
mexican border pharmacies shipping to usa Mexico pharmacy best online pharmacies in mexico mexicanpharmacy.win
https://mexicanpharmacy.win/# mexican pharmaceuticals online mexicanpharmacy.win
best canadian pharmacy no prescription
canadian pharmacy store: Cheapest drug prices Canada – safe canadian pharmacies canadianpharmacy.pro
http://canadianpharmacy.pro/# northwest pharmacy canada canadianpharmacy.pro
mexican mail order pharmacies Medicines Mexico medicine in mexico pharmacies mexicanpharmacy.win
https://indianpharmacy.shop/# indian pharmacy paypal indianpharmacy.shop
buy prescription drugs from canada cheap Cheapest drug prices Canada my canadian pharmacy canadianpharmacy.pro
http://mexicanpharmacy.win/# mexico pharmacies prescription drugs mexicanpharmacy.win
best online pharmacy india indian pharmacies safe reputable indian pharmacies indianpharmacy.shop
http://canadianpharmacy.pro/# canadian pharmacy world canadianpharmacy.pro
https://canadianpharmacy.pro/# onlinecanadianpharmacy canadianpharmacy.pro
online shopping pharmacy india
https://mexicanpharmacy.win/# mexico pharmacies prescription drugs mexicanpharmacy.win
http://indianpharmacy.shop/# best online pharmacy india indianpharmacy.shop
online ed medication no prescription
mexico drug stores pharmacies Mexico pharmacy mexico pharmacies prescription drugs mexicanpharmacy.win
http://canadianpharmacy.pro/# canadian pharmacy meds canadianpharmacy.pro
online shopping pharmacy india
https://mexicanpharmacy.win/# purple pharmacy mexico price list mexicanpharmacy.win
best online pharmacies in mexico medicine in mexico pharmacies mexican pharmaceuticals online mexicanpharmacy.win
https://mexicanpharmacy.win/# mexican pharmacy mexicanpharmacy.win
world pharmacy india
https://canadianpharmacy.pro/# canada pharmacy reviews canadianpharmacy.pro
canada discount pharmacy canadian pharmacies comparison canadian pharmacy ed medications canadianpharmacy.pro
http://indianpharmacy.shop/# india pharmacy mail order indianpharmacy.shop
https://mexicanpharmacy.win/# mexico pharmacies prescription drugs mexicanpharmacy.win
indianpharmacy com
http://indianpharmacy.shop/# cheapest online pharmacy india indianpharmacy.shop
http://canadianpharmacy.pro/# canadian pharmacy mall canadianpharmacy.pro
fda approved canadian pharmacies
buying from online mexican pharmacy medicine in mexico pharmacies medicine in mexico pharmacies mexicanpharmacy.win
https://indianpharmacy.shop/# indian pharmacy indianpharmacy.shop
indian pharmacy online
https://indianpharmacy.shop/# india online pharmacy indianpharmacy.shop
canada rx pharmacy world canadapharmacyonline canadian pharmacy king canadianpharmacy.pro
http://indianpharmacy.shop/# best online pharmacy india indianpharmacy.shop
reputable indian online pharmacy
https://indianpharmacy.shop/# online shopping pharmacy india indianpharmacy.shop
Viagra homme sans prescription Prix du Viagra en pharmacie en France Viagra femme sans ordonnance 24h
Pharmacie en ligne France: Levitra acheter – Pharmacie en ligne fiable
https://levitrasansordonnance.pro/# Pharmacie en ligne pas cher
Acheter mГ©dicaments sans ordonnance sur internet kamagra livraison 24h acheter medicament a l etranger sans ordonnance
pharmacie ouverte 24/24: Pharmacie en ligne livraison rapide – acheter mГ©dicaments Г l’Г©tranger
https://pharmadoc.pro/# acheter médicaments à l’étranger
https://acheterkamagra.pro/# pharmacie ouverte
pharmacie ouverte 24/24
Viagra sans ordonnance livraison 48h: viagra sans ordonnance – Viagra sans ordonnance 24h suisse
Pharmacie en ligne sans ordonnance: cialis sans ordonnance – Pharmacie en ligne France
pharmacie ouverte 24/24 Cialis sans ordonnance 24h п»їpharmacie en ligne
https://viagrasansordonnance.pro/# Viagra pas cher inde
acheter mГ©dicaments Г l’Г©tranger: kamagra oral jelly – pharmacie ouverte
http://levitrasansordonnance.pro/# Pharmacie en ligne France
Viagra prix pharmacie paris: viagrasansordonnance.pro – Viagra pas cher livraison rapide france
Pharmacie en ligne livraison 24h kamagra livraison 24h Pharmacie en ligne livraison 24h
http://acheterkamagra.pro/# Pharmacie en ligne France
pharmacie ouverte 24/24
Pharmacie en ligne fiable: Medicaments en ligne livres en 24h – pharmacie ouverte 24/24
Acheter mГ©dicaments sans ordonnance sur internet: Levitra sans ordonnance 24h – Pharmacie en ligne sans ordonnance
Pharmacies en ligne certifiГ©es acheter medicament a l etranger sans ordonnance acheter medicament a l etranger sans ordonnance
http://viagrasansordonnance.pro/# Viagra Pfizer sans ordonnance
Pharmacie en ligne livraison gratuite: Medicaments en ligne livres en 24h – Acheter mГ©dicaments sans ordonnance sur internet
Pharmacie en ligne livraison 24h Pharmacie en ligne livraison 24h acheter medicament a l etranger sans ordonnance
5 mg prednisone daily can you buy prednisone in canada 2.5 mg prednisone daily
minocycline 100mg pills: stromectol nz – minocycline 100mg tablets
buy amoxil: medicine amoxicillin 500mg – amoxicillin medicine over the counter
https://azithromycin.bid/# buy zithromax online with mastercard
stromectol coronavirus price of ivermectin liquid ivermectin brand
buy zithromax online with mastercard: can i buy zithromax over the counter in canada – zithromax 500 price
https://prednisonetablets.shop/# prednisone online for sale
http://clomiphene.icu/# can you get clomid no prescription
where can i get cheap clomid without a prescription get generic clomid buy clomid pills
can i get clomid without insurance: can i order clomid pill – where can i buy cheap clomid price
where can i buy cheap clomid without prescription: where can i get generic clomid without insurance – can i get cheap clomid without prescription
http://amoxicillin.bid/# amoxicillin pharmacy price
ivermectin 4000 mcg stromectol xl ivermectin 1
amoxicillin 875 mg tablet: amoxil generic – amoxicillin no prescipion
https://ivermectin.store/# ivermectin pills
cost clomid price: how to buy clomid for sale – where to get clomid prices
buy generic zithromax online average cost of generic zithromax can you buy zithromax over the counter
ivermectin 0.1: minocycline 100mg tablets for human – minocycline drug
https://amoxicillin.bid/# amoxicillin for sale online
https://amoxicillin.bid/# amoxicillin tablet 500mg
can i get cheap clomid without insurance: cost generic clomid pill – where buy clomid no prescription
order cheap clomid no prescription can you buy generic clomid pills cost generic clomid pill
https://prednisonetablets.shop/# where to buy prednisone without prescription
can you get generic clomid prices: cheap clomid price – can i buy generic clomid no prescription
minocycline 50 mg tabs: minocycline 100 mg without prescription – stromectol 3mg tablets
http://amoxicillin.bid/# amoxicillin 250 mg price in india
how to get prednisone without a prescription prednisone tablets 2.5 mg 50mg prednisone tablet
can i purchase prednisone without a prescription: prednisone otc uk – prednisone purchase canada
http://amoxicillin.bid/# cheap amoxicillin 500mg
buy prednisone without rx: compare prednisone prices – can you buy prednisone without a prescription
http://prednisonetablets.shop/# prednisone 50 mg prices
where to get zithromax over the counter zithromax capsules 250mg zithromax azithromycin
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article. https://www.binance.com/ph/register?ref=T7KCZASX
my canadian pharmacy review Canadian Pharmacy best canadian online pharmacy canadianpharm.store
canada ed drugs: Canada Pharmacy online – reputable canadian online pharmacies canadianpharm.store
best canadian online pharmacy reviews: Licensed Online Pharmacy – thecanadianpharmacy canadianpharm.store
http://indianpharm.store/# pharmacy website india indianpharm.store
canadian drug Pharmacies in Canada that ship to the US reliable canadian pharmacy reviews canadianpharm.store
india online pharmacy: international medicine delivery from india – top 10 online pharmacy in india indianpharm.store
mexico drug stores pharmacies: Online Mexican pharmacy – best online pharmacies in mexico mexicanpharm.shop
https://canadianpharm.store/# buying drugs from canada canadianpharm.store
indian pharmacy online international medicine delivery from india pharmacy website india indianpharm.store
Online medicine home delivery: Indian pharmacy to USA – indian pharmacy online indianpharm.store
https://mexicanpharm.shop/# buying from online mexican pharmacy mexicanpharm.shop
http://indianpharm.store/# india pharmacy mail order indianpharm.store
mexican mail order pharmacies: Certified Pharmacy from Mexico – mexican drugstore online mexicanpharm.shop
best india pharmacy order medicine from india to usa indian pharmacy indianpharm.store
mexico pharmacy: Online Pharmacies in Mexico – purple pharmacy mexico price list mexicanpharm.shop
http://mexicanpharm.shop/# buying prescription drugs in mexico mexicanpharm.shop
pharmacies in mexico that ship to usa: Certified Pharmacy from Mexico – medicine in mexico pharmacies mexicanpharm.shop
purple pharmacy mexico price list: Certified Pharmacy from Mexico – mexican drugstore online mexicanpharm.shop
canadian medications Pharmacies in Canada that ship to the US pharmacy com canada canadianpharm.store
https://canadianpharm.store/# legal canadian pharmacy online canadianpharm.store
prescription drugs canada buy online: Licensed Online Pharmacy – canadian pharmacies compare canadianpharm.store
https://canadianpharm.store/# canadian pharmacy oxycodone canadianpharm.store
mexico pharmacies prescription drugs Certified Pharmacy from Mexico mexican pharmacy mexicanpharm.shop
medication canadian pharmacy: Canadian Pharmacy – northwest canadian pharmacy canadianpharm.store
https://canadianpharm.store/# prescription drugs canada buy online canadianpharm.store
cheapest online pharmacy india: Indian pharmacy to USA – indianpharmacy com indianpharm.store
medication from mexico pharmacy Online Mexican pharmacy mexican online pharmacies prescription drugs mexicanpharm.shop
buy prescription drugs from india: indianpharmacy com – indianpharmacy com indianpharm.store
http://mexicanpharm.shop/# best mexican online pharmacies mexicanpharm.shop
cheapest online pharmacy india: Indian pharmacy to USA – world pharmacy india indianpharm.store
mexico drug stores pharmacies Certified Pharmacy from Mexico mexican rx online mexicanpharm.shop
top 10 online pharmacy in india: order medicine from india to usa – world pharmacy india indianpharm.store
https://canadianpharm.store/# trusted canadian pharmacy canadianpharm.store
top 10 pharmacies in india: Indian pharmacy to USA – indian pharmacy indianpharm.store
https://mexicanpharm.shop/# mexican mail order pharmacies mexicanpharm.shop
safe canadian pharmacy Best Canadian online pharmacy legitimate canadian online pharmacies canadianpharm.store
reputable indian pharmacies: international medicine delivery from india – reputable indian online pharmacy indianpharm.store
https://canadianpharm.store/# canadian online pharmacy canadianpharm.store
india online pharmacy: Indian pharmacy to USA – cheapest online pharmacy india indianpharm.store
reputable indian pharmacies: international medicine delivery from india – top 10 online pharmacy in india indianpharm.store
http://mexicanpharm.shop/# pharmacies in mexico that ship to usa mexicanpharm.shop
medication from mexico pharmacy buying from online mexican pharmacy mexico drug stores pharmacies mexicanpharm.shop
mexican online pharmacies prescription drugs: Online Pharmacies in Mexico – buying prescription drugs in mexico online mexicanpharm.shop
http://mexicanpharm.shop/# pharmacies in mexico that ship to usa mexicanpharm.shop
canadian pharmacy ed medications Canada Pharmacy online legitimate canadian pharmacy canadianpharm.store
fda approved canadian online pharmacies canadian pharmacy online canada online canadian pharcharmy
trust online pharmacy: canadian pharmacy androgel – buy prescription drugs online
http://canadadrugs.pro/# canada pharmacy online no script
canadian pharmacy for sildenafil true canadian pharmacy canadian prescription drugstore
reputable online canadian pharmacy: best online pharmacy stores – online pharmacy without precriptions
legal canadian pharmacy online: legitimate canadian pharmacy – canadian pharmacy
order drugs online: canadian pharmaceuticals online reviews – top 10 mail order pharmacies
https://canadadrugs.pro/# cheap prescription drugs
buy medicine canada family pharmacy online prescription drug price comparison
canada drugs online: no prescription canadian drugs – legitimate canadian mail order pharmacy
http://canadadrugs.pro/# list of 24 hour pharmacies
north canadian pharmacy canadian drug store online canadian drugstore online
online prescriptions: aarp approved canadian pharmacies – prescription online
http://canadadrugs.pro/# canadian pharmacies that are legit
canadian drugstore pharmacy: certified canadian drug stores – safe reliable canadian pharmacy
pharmacy in canada: high street discount pharmacy – mail order pharmacy list
online pharmacy without precriptions approved canadian pharmacies online list of canadian pharmacies online
https://canadadrugs.pro/# best canadian online pharmacy viagra
overseas online pharmacy: prescription drugs without doctor approval – canada pharmacy not requiring prescription
canada online pharmacy: best canadian mail order pharmacy – canadian pharmacy no prescription
https://canadadrugs.pro/# certified online canadian pharmacies
canadapharmacyonline.com: reputable online pharmacy – best online canadian pharmacies
http://canadadrugs.pro/# canadian wholesale pharmacy
canadian pharmacies without prescriptions: best mexican online pharmacies – online pharmacy usa
canadian pharmacy world: online pharmacies canada reviews – best mail order canadian pharmacy
canadian discount cialis: accutane mexican pharmacy – recommended canadian pharmacies
http://canadadrugs.pro/# list of safe online pharmacies
no script pharmacy: overseas online pharmacies – top rated online pharmacy
my canadian pharmacy: good online mexican pharmacy – northeast discount pharmacy
https://canadadrugs.pro/# canadian drug store coupon
verified canadian pharmacy: best online canadian pharcharmy – prescription drugs without the prescription
http://canadadrugs.pro/# top rated canadian mail order pharmacies
canadian drug: canadian xanax – approved canadian online pharmacies
cheapest canadian online pharmacy canadian internet pharmacies prescription drug pricing
https://canadadrugs.pro/# canadian drugs
buy prescription drugs without doctor cialis without a doctor prescription canada ed meds online without prescription or membership
indian pharmacies safe: cheapest online pharmacy india – india pharmacy
http://medicinefromindia.store/# reputable indian online pharmacy
buying ed pills online erectile dysfunction drugs ed meds
canada pharmacy online: legitimate canadian pharmacy online – pet meds without vet prescription canada
http://edwithoutdoctorprescription.pro/# prescription drugs
mexican border pharmacies shipping to usa medicine in mexico pharmacies pharmacies in mexico that ship to usa
cheap erectile dysfunction pill: best otc ed pills – ed dysfunction treatment
http://canadianinternationalpharmacy.pro/# drugs from canada
pharmacy website india: top 10 pharmacies in india – reputable indian online pharmacy
http://edpill.cheap/# gnc ed pills
canadian valley pharmacy safe reliable canadian pharmacy reddit canadian pharmacy
https://canadianinternationalpharmacy.pro/# canada pharmacy world
mexican drugstore online mexico drug stores pharmacies mexico pharmacies prescription drugs
http://medicinefromindia.store/# reputable indian pharmacies
mexican rx online: mexico drug stores pharmacies – mexico pharmacies prescription drugs
best online pharmacy india indianpharmacy com cheapest online pharmacy india
https://edwithoutdoctorprescription.pro/# legal to buy prescription drugs without prescription
best online pharmacies in mexico: buying prescription drugs in mexico online – mexican online pharmacies prescription drugs
http://certifiedpharmacymexico.pro/# medicine in mexico pharmacies
cheapest online pharmacy india world pharmacy india indian pharmacies safe
https://certifiedpharmacymexico.pro/# medicine in mexico pharmacies
new ed drugs treatment for ed male ed drugs
online canadian pharmacy reviews: cross border pharmacy canada – canadian pharmacy service
https://edwithoutdoctorprescription.pro/# prescription drugs
cheap canadian pharmacy online reliable canadian pharmacy reliable canadian pharmacy
http://edpill.cheap/# impotence pills
ed drugs compared ed medications herbal ed treatment
https://edpill.cheap/# the best ed pill
https://canadianinternationalpharmacy.pro/# canada ed drugs
best medication for ed: cure ed – ed pills cheap
cheapest online pharmacy india mail order pharmacy india indianpharmacy com
https://canadianinternationalpharmacy.pro/# canada ed drugs
canadian pharmacies online canadapharmacyonline canadian pharmacy ratings
https://edwithoutdoctorprescription.pro/# prescription drugs without doctor approval
canadian online pharmacy reviews best rated canadian pharmacy canada drugs online review
http://edpill.cheap/# non prescription erection pills
best ed pills non prescription: ed pills without doctor prescription – buy prescription drugs without doctor
buy prescription drugs online cheap cialis ed prescription drugs
http://medicinefromindia.store/# top 10 online pharmacy in india
Online medicine order online pharmacy india top 10 pharmacies in india
https://canadianinternationalpharmacy.pro/# canadian pharmacy review
Before we can get to understand more about PayPal casinos, we need to get to the bottom of what PayPal is and what type of online casino payment method category it falls into. To start with, PayPal is unlike any debit card or mainstream banking payment option. Instead, it is a US-founded company that provides you with e-wallet payment capabilities to shop spend with at retailers around the world. It acts as a middle-man between your bank account and the operator, providing a secure gateway that offers both speedy deposits and withdrawals of real cash. We are glad to add one more country to our list of the luckiest who can make the fastest and the safest online casino deposits, and this country is Belgium. As we can see, Europeans have a more positive attitude towards PayPal, and thus we have a decent number of the online casinos that take PayPal in Belgium. The deposits in PayPal casinos in Belgium may not always bring you the real money wins, but you’ll be sure to have a great gambling experience and a lot of fun.
https://landenheby639639.blogpixi.com/20817617/goldwin-slot
Free Spin Casino is considering an old-fashioned casino that has been around for quite some time, since the original release of the platform in 2004. Many things have changed since the time of its release. The casino has upgraded its graphics and design as well as received an approval from Curacao’s most prestigious casino, and forged the partnership with Real Time Gaming. This partnership has helped Free Spin Casino to achieve international fame. Today, the version that is played online is not just one of the most well-known games at casinos online, but it is also one of the most watched. Casinos can choose any number of pre-selected slot machines for you to enjoy your extra spins on. There are a couple of slot staples that will regularly pop up for free spins online casino bonuses.
ed meds online without doctor prescription gnc ed pills new ed treatments
https://edwithoutdoctorprescription.pro/# buy prescription drugs online legally
non prescription ed drugs: ed pills without doctor prescription – buy prescription drugs online without
http://edpill.cheap/# buying ed pills online
mexican rx online pharmacies in mexico that ship to usa medication from mexico pharmacy
https://canadianinternationalpharmacy.pro/# canadapharmacyonline com
non prescription erection pills cialis without a doctor prescription viagra without doctor prescription
https://edpill.cheap/# ed pill
mexico pharmacy mexican rx online mexican drugstore online
pharmacies in mexico that ship to usa buying prescription drugs in mexico medication from mexico pharmacy
medication from mexico pharmacy mexican pharmaceuticals online best online pharmacies in mexico
mexico drug stores pharmacies mexico pharmacies prescription drugs buying from online mexican pharmacy
http://mexicanph.shop/# п»їbest mexican online pharmacies
best online pharmacies in mexico
mexican pharmaceuticals online best mexican online pharmacies medication from mexico pharmacy
mexican border pharmacies shipping to usa mexican border pharmacies shipping to usa mexico drug stores pharmacies
mexican rx online mexican border pharmacies shipping to usa buying from online mexican pharmacy
mexican mail order pharmacies medication from mexico pharmacy buying prescription drugs in mexico
reputable mexican pharmacies online mexico drug stores pharmacies mexican pharmaceuticals online
reputable mexican pharmacies online mexican drugstore online mexican rx online
http://mexicanph.shop/# п»їbest mexican online pharmacies
mexican border pharmacies shipping to usa
mexico pharmacies prescription drugs buying prescription drugs in mexico online mexican pharmaceuticals online
medicine in mexico pharmacies pharmacies in mexico that ship to usa mexican drugstore online
http://mexicanph.com/# mexican pharmaceuticals online
п»їbest mexican online pharmacies
mexican rx online mexico pharmacy п»їbest mexican online pharmacies
medication from mexico pharmacy mexican online pharmacies prescription drugs mexico drug stores pharmacies
zithromax 500 dosage
mexico pharmacies prescription drugs mexico drug stores pharmacies mexican border pharmacies shipping to usa
buying prescription drugs in mexico online mexican border pharmacies shipping to usa medicine in mexico pharmacies
mexico pharmacy mexican border pharmacies shipping to usa mexican border pharmacies shipping to usa
http://mexicanph.com/# mexican drugstore online
mexican pharmaceuticals online
mexico pharmacies prescription drugs reputable mexican pharmacies online buying from online mexican pharmacy
mexican online pharmacies prescription drugs mexican pharmaceuticals online mexico drug stores pharmacies
buying prescription drugs in mexico mexico pharmacies prescription drugs mexican drugstore online
mexico pharmacy mexican pharmacy purple pharmacy mexico price list
mexican mail order pharmacies purple pharmacy mexico price list mexican pharmaceuticals online
mexico drug stores pharmacies mexico drug stores pharmacies mexican mail order pharmacies
mexican drugstore online buying prescription drugs in mexico mexico pharmacies prescription drugs
buying from online mexican pharmacy mexican drugstore online mexico drug stores pharmacies
mexican pharmacy best online pharmacies in mexico mexican drugstore online
mexican pharmaceuticals online medication from mexico pharmacy mexico pharmacy
mexican online pharmacies prescription drugs mexican online pharmacies prescription drugs medicine in mexico pharmacies
http://mexicanph.com/# medication from mexico pharmacy
mexican border pharmacies shipping to usa
pharmacies in mexico that ship to usa buying from online mexican pharmacy mexico drug stores pharmacies
purple pharmacy mexico price list best online pharmacies in mexico mexican border pharmacies shipping to usa
best mexican online pharmacies mexican border pharmacies shipping to usa mexican pharmacy
reputable mexican pharmacies online mexican online pharmacies prescription drugs purple pharmacy mexico price list
mexican border pharmacies shipping to usa best mexican online pharmacies mexican pharmacy
mexican online pharmacies prescription drugs buying from online mexican pharmacy mexico drug stores pharmacies
best online pharmacies in mexico medication from mexico pharmacy buying prescription drugs in mexico online
buying from online mexican pharmacy mexican pharmaceuticals online medication from mexico pharmacy
mexican drugstore online buying from online mexican pharmacy mexico pharmacy
buying prescription drugs in mexico mexican drugstore online mexican pharmacy
mexico pharmacy mexican online pharmacies prescription drugs buying prescription drugs in mexico
medicine in mexico pharmacies mexico drug stores pharmacies mexican rx online
http://mexicanph.com/# mexican pharmaceuticals online
п»їbest mexican online pharmacies
pharmacies in mexico that ship to usa buying prescription drugs in mexico online best online pharmacies in mexico
mexican mail order pharmacies mexican online pharmacies prescription drugs medicine in mexico pharmacies
mexican online pharmacies prescription drugs mexican pharmaceuticals online best online pharmacies in mexico
reputable mexican pharmacies online mexican border pharmacies shipping to usa mexico pharmacies prescription drugs
buying prescription drugs in mexico п»їbest mexican online pharmacies medicine in mexico pharmacies
mexican pharmacy mexico pharmacy mexico drug stores pharmacies
mexican pharmacy medicine in mexico pharmacies mexican mail order pharmacies
mexican rx online mexico drug stores pharmacies purple pharmacy mexico price list
reputable mexican pharmacies online buying prescription drugs in mexico mexican pharmaceuticals online
https://mexicanph.com/# mexican mail order pharmacies
mexican drugstore online
mexico pharmacies prescription drugs reputable mexican pharmacies online mexican pharmaceuticals online
buying prescription drugs in mexico mexican border pharmacies shipping to usa mexico drug stores pharmacies
mexico drug stores pharmacies mexico pharmacies prescription drugs mexico drug stores pharmacies
best mexican online pharmacies reputable mexican pharmacies online mexico drug stores pharmacies
mexican mail order pharmacies mexican drugstore online pharmacies in mexico that ship to usa
mexican pharmaceuticals online mexico drug stores pharmacies mexican mail order pharmacies
mexico drug stores pharmacies reputable mexican pharmacies online mexican border pharmacies shipping to usa
medication from mexico pharmacy mexico drug stores pharmacies mexican drugstore online
mexico pharmacy buying prescription drugs in mexico pharmacies in mexico that ship to usa
buying prescription drugs in mexico mexico pharmacies prescription drugs mexican border pharmacies shipping to usa
mexico drug stores pharmacies mexican drugstore online mexico pharmacies prescription drugs
best online pharmacies in mexico mexico pharmacies prescription drugs mexican border pharmacies shipping to usa
mexican drugstore online medication from mexico pharmacy mexican drugstore online
https://mexicanph.shop/# reputable mexican pharmacies online
mexican pharmaceuticals online
mexican pharmacy medication from mexico pharmacy pharmacies in mexico that ship to usa
mexican mail order pharmacies mexican pharmacy п»їbest mexican online pharmacies
buying prescription drugs in mexico online purple pharmacy mexico price list mexican pharmacy
mexico pharmacy mexico pharmacies prescription drugs mexican rx online
buying prescription drugs in mexico online mexican online pharmacies prescription drugs mexican rx online
pharmacies in mexico that ship to usa mexico pharmacy mexico drug stores pharmacies
mexico drug stores pharmacies mexico pharmacy best mexican online pharmacies
mexican pharmacy medication from mexico pharmacy pharmacies in mexico that ship to usa
mexico pharmacy medicine in mexico pharmacies buying prescription drugs in mexico
mexican pharmacy buying prescription drugs in mexico online mexican pharmaceuticals online
https://amoxil.cheap/# can you buy amoxicillin over the counter
amoxicillin 775 mg: amoxicillin 800 mg price – amoxicillin 200 mg tablet
https://buyprednisone.store/# prednisone 20mg price in india
purchase amoxicillin online without prescription amoxicillin discount amoxicillin 500 mg brand name
ivermectin otc: stromectol uk buy – buy ivermectin cream for humans
http://buyprednisone.store/# prednisone 5 mg
buy furosemide online: furosemida 40 mg – lasix online
https://buyprednisone.store/# prednisone cream brand name
prednisone 20 mg tablets: generic prednisone otc – can you buy prednisone over the counter in usa
https://buyprednisone.store/# prednisone drug costs
ivermectin otc purchase stromectol buy ivermectin uk
http://lisinopril.top/# lisinopril 102
prednisone 20mg capsule: 40 mg prednisone pill – prednisone 2.5 mg tab
cost of generic lisinopril 10 mg: generic lisinopril online – prinivil
prednisone 20mg online pharmacy 5 mg prednisone daily prednisone 500 mg tablet
https://furosemide.guru/# lasix 40 mg
lisinopril 40 mg brand name in india: lisinopril in mexico – buy lisinopril 20 mg online
https://amoxil.cheap/# amoxicillin online without prescription
http://amoxil.cheap/# amoxicillin discount coupon
amoxicillin online purchase generic for amoxicillin amoxicillin 500mg prescription
furosemide 40mg: lasix side effects – lasix pills
where can i get prednisone over the counter: prednisone 10mg online – where can i buy prednisone online without a prescription
https://stromectol.fun/# ivermectin ireland
https://amoxil.cheap/# amoxicillin 500mg capsule buy online
where can i buy amoxicillin over the counter uk: amoxicillin 500mg buy online uk – can i buy amoxicillin over the counter
generic amoxicillin online amoxicillin no prescription amoxicillin 500mg capsules uk
lisinopril 40 mg cost: lisinopril 80 mg tablet – lisinopril 12.5 tablet
https://furosemide.guru/# lasix tablet
furosemide 40mg: Over The Counter Lasix – lasix 40 mg
https://amoxil.cheap/# amoxicillin 500mg capsules
buy lisinopril online no prescription india zestril tab 10mg lisinopril diuretic
prednisone 2 5 mg: purchase prednisone canada – prednisone tablets 2.5 mg
http://amoxil.cheap/# amoxicillin over the counter in canada
over the counter amoxicillin canada: amoxicillin cephalexin – buy amoxicillin online cheap
buy ivermectin pills: order stromectol – ivermectin medicine
http://lisinopril.top/# online lisinopril
ivermectin buy nz ivermectin virus stromectol price usa
10mg generic 10mg lisinopril: lisinopril 20mg 25mg – price of lisinopril 30 mg
http://buyprednisone.store/# prednisone buy no prescription
order stromectol online: stromectol pill – ivermectin cost
https://lisinopril.top/# lisinopril 80mg tablet
price for 15 prednisone prednisone 10mg tabs prednisone cost us
amoxicillin 500mg buy online canada: cost of amoxicillin 30 capsules – medicine amoxicillin 500
http://amoxil.cheap/# buy amoxicillin online without prescription
lisinopril 10 12.5 mg: benicar lisinopril – lisinopril 419
azithromycin amoxicillin: amoxicillin 875 mg tablet – buy amoxicillin online cheap
http://lisinopril.top/# cost of lisinopril in mexico
lisinopril 5 mg tablet price lisinopril 3973 lisinopril 20 12.5 mg
buy lisinopril 20 mg online uk: prinivil 10 mg tablet – zestril 10 mg price in india
http://furosemide.guru/# lasix generic
http://lisinopril.top/# lisinopril 422
lasix furosemide: Buy Lasix No Prescription – lasix furosemide 40 mg
lasix online: Buy Lasix – furosemide 40 mg
lisinopril 60 mg daily lisinopril discount lisinopril 40 mg
http://buyprednisone.store/# prednisone 10mg buy online
lisinopril 5 mg over the counter: 16 lisinopril – lisinopril 20 25 mg
https://amoxil.cheap/# order amoxicillin online no prescription
amoxicillin 500 mg where to buy can you buy amoxicillin uk where to buy amoxicillin 500mg
lisinopril 40 mg prices: zestril 40 mg tablet – lisinopril 5mg pill
20mg prednisone: buying prednisone – prednisone 60 mg
http://stromectol.fun/# minocin 50 mg for scabies
http://furosemide.guru/# lasix 40 mg
ampicillin amoxicillin: amoxicillin discount coupon – generic amoxicillin
prednisone 10mg canada: prednisone oral – where can i buy prednisone without prescription
https://amoxil.cheap/# amoxicillin 500 mg cost
https://furosemide.guru/# lasix 20 mg
buy prednisone online fast shipping: can you buy prednisone in canada – prednisone 12 tablets price
http://lisinopril.top/# lisinopril 25 mg tablet
prednisone 30 mg daily prednisone canada prescription prednisone in uk
https://stromectol.fun/# stromectol 3mg tablets
amoxacillian without a percription: where can i get amoxicillin – amoxicillin 500 mg purchase without prescription
buy amoxicillin 500mg canada: amoxicillin 500mg for sale uk – amoxicillin pharmacy price
http://stromectol.fun/# ivermectin 0.08 oral solution
how to get amoxicillin: amoxicillin online no prescription – buy amoxicillin from canada
lisinopril 60 mg tablet zestril 10 mg online lisinopril 80mg tablet
http://amoxil.cheap/# cost of amoxicillin 875 mg
amoxicillin 500mg price in canada: amoxicillin 200 mg tablet – amoxicillin for sale
http://buyprednisone.store/# prednisone 2.5 mg daily
how much is lisinopril 5 mg: lisinopril generic – lisinopril 5 mg tabs
http://lisinopril.top/# lisinopril 40 mg on line
furosemide 100 mg lasix side effects lasix side effects
http://furosemide.guru/# lasix 100 mg tablet
lasix online: Buy Lasix No Prescription – lasix 20 mg
glucophage espaГ±a
http://stromectol.fun/# stromectol 3 mg price
buy lasix online: Buy Lasix – lasix pills
http://indianph.com/# top 10 pharmacies in india
best online pharmacy india
online shopping pharmacy india indian pharmacy online п»їlegitimate online pharmacies india
Online medicine order buy medicines online in india top online pharmacy india
https://indianph.com/# Online medicine home delivery
buy prescription drugs from india
http://indianph.com/# Online medicine order
online shopping pharmacy india
п»їlegitimate online pharmacies india top 10 pharmacies in india india pharmacy mail order
india pharmacy mail order india online pharmacy buy medicines online in india
https://indianph.xyz/# best india pharmacy
indian pharmacies safe
https://indianph.xyz/# mail order pharmacy india
https://indianph.com/# india pharmacy mail order
reputable indian online pharmacy
https://indianph.xyz/# indian pharmacies safe
indian pharmacies safe
buy medicines online in india top 10 pharmacies in india best india pharmacy
http://indianph.xyz/# indian pharmacy paypal
indianpharmacy com
indian pharmacy indianpharmacy com indian pharmacy
https://indianph.com/# world pharmacy india
buy prescription drugs from india
https://indianph.xyz/# buy medicines online in india
buy medicines online in india
indian pharmacy online top 10 online pharmacy in india mail order pharmacy india
https://indianph.com/# top 10 online pharmacy in india
https://doxycycline.auction/# cheap doxycycline online
doxycycline 50mg: doxycycline hydrochloride 100mg – doxy 200
https://diflucan.pro/# diflucan tablets
buy cipro buy cipro cheap where can i buy cipro online
https://cytotec24.shop/# buy cytotec over the counter
http://cytotec24.shop/# buy misoprostol over the counter
https://cipro.guru/# cipro 500mg best prices
can i eat eggs while taking ciprofloxacin
doxycycline 100mg online buy doxycycline online uk buy doxycycline monohydrate
tamoxifen alternatives: where to get nolvadex – aromatase inhibitors tamoxifen
http://diflucan.pro/# diflucan without get a prescription online
diflucan online: diflucan 200 mg cost – generic diflucan
diflucan pill diflucan 250 mg diflucan 150 mg capsule
https://cytotec24.shop/# buy cytotec pills
http://diflucan.pro/# can i buy diflucan in mexico
doxycycline order online order doxycycline online doxycycline hyclate
http://cytotec24.com/# buy misoprostol over the counter
doxycycline 150 mg: buy doxycycline online 270 tabs – purchase doxycycline online
http://nolvadex.guru/# tamoxifen warning
https://nolvadex.guru/# tamoxifen and weight loss
https://diflucan.pro/# diflucan over the counter singapore
diflucan cost canada buy diflucan online usa diflucan capsule 50 mg
http://cipro.guru/# ciprofloxacin mail online
https://cipro.guru/# cipro ciprofloxacin
http://nolvadex.guru/# aromatase inhibitors tamoxifen
nolvadex half life tamoxifen joint pain is nolvadex legal
http://cytotec24.com/# cytotec pills online
https://doxycycline.auction/# doxycycline 100mg dogs
tamoxifen hot flashes tamoxifen endometriosis tamoxifen men
http://doxycycline.auction/# doxycycline generic
https://cytotec24.shop/# Misoprostol 200 mg buy online
https://abelladanger.online/# Abella Danger
lana rhoades: lana rhoades filmleri – lana rhoades
https://sweetiefox.online/# swetie fox
http://evaelfie.pro/# eva elfie izle
Angela White filmleri: ?????? ???? – Angela White video
http://sweetiefox.online/# Sweetie Fox video
http://angelawhite.pro/# Angela White
sweeti fox: sweeti fox – Sweetie Fox izle
http://sweetiefox.online/# Sweetie Fox video
http://lanarhoades.fun/# lana rhodes
http://angelawhite.pro/# Angela White filmleri
Sweetie Fox filmleri: Sweetie Fox filmleri – sweeti fox
http://abelladanger.online/# abella danger izle
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://www.binance.info/ph/join?ref=B4EPR6J0
http://abelladanger.online/# abella danger izle
http://angelawhite.pro/# Angela White video
Angela White filmleri: Angela White – Angela Beyaz modeli
https://angelawhite.pro/# Angela White
https://abelladanger.online/# Abella Danger
https://lanarhoades.fun/# lana rhoades modeli
Angela Beyaz modeli: Angela White izle – Angela White filmleri
https://angelawhite.pro/# Angela Beyaz modeli
https://angelawhite.pro/# Angela White izle
http://evaelfie.pro/# eva elfie modeli
lana rhoades filmleri: lana rhoades – lana rhoades filmleri
https://angelawhite.pro/# Angela White filmleri
http://evaelfie.pro/# eva elfie filmleri
https://abelladanger.online/# abella danger izle
eva elfie modeli: eva elfie filmleri – eva elfie video
http://sweetiefox.online/# swetie fox
http://sweetiefox.online/# Sweetie Fox modeli
Angela White video: Angela White izle – Angela White
https://abelladanger.online/# abella danger video
https://evaelfie.pro/# eva elfie filmleri
Angela Beyaz modeli: Angela White – Angela White filmleri
https://evaelfie.pro/# eva elfie filmleri
http://sweetiefox.online/# sweeti fox
http://evaelfie.pro/# eva elfie modeli
http://sweetiefox.online/# Sweetie Fox video
http://lanarhoades.fun/# lana rhoades video
https://sweetiefox.online/# Sweetie Fox filmleri
Angela White filmleri: abella danger filmleri – abella danger video
http://angelawhite.pro/# Angela White izle
eva elfie filmleri: eva elfie video – eva elfie izle
http://abelladanger.online/# Abella Danger
http://evaelfie.pro/# eva elfie izle
Sweetie Fox: swetie fox – Sweetie Fox video
eva elfie full video: eva elfie hd – eva elfie new videos
http://evaelfie.site/# eva elfie
meet= dating dating: http://lanarhoades.pro/# lana rhoades unleashed
lana rhoades hot: lana rhoades full video – lana rhoades full video
https://lanarhoades.pro/# lana rhoades solo
http://lanarhoades.pro/# lana rhoades unleashed
eva elfie videos: eva elfie – eva elfie new videos
eva elfie photo: eva elfie – eva elfie
best singles website: http://lanarhoades.pro/# lana rhoades hot
http://sweetiefox.pro/# sweetie fox video
sweetie fox cosplay: sweetie fox full – sweetie fox cosplay
gabapentin 300 mg para que sirve
eva elfie photo: eva elfie videos – eva elfie hd
http://sweetiefox.pro/# sweetie fox new
mia malkova full video: mia malkova full video – mia malkova photos
plentyoffish free online dating site: https://miamalkova.life/# mia malkova
sweetie fox full video: sweetie fox new – sweetie fox video
http://miamalkova.life/# mia malkova latest
eva elfie full videos: eva elfie hd – eva elfie full videos
eva elfie full video: eva elfie videos – eva elfie new video
https://sweetiefox.pro/# sweetie fox new
dating sites contact united states: https://miamalkova.life/# mia malkova videos
lana rhoades pics: lana rhoades solo – lana rhoades pics
http://miamalkova.life/# mia malkova latest
eva elfie photo: eva elfie – eva elfie new video
internet dating: https://lanarhoades.pro/# lana rhoades solo
https://sweetiefox.pro/# sweetie fox video
lana rhoades solo: lana rhoades solo – lana rhoades pics
http://evaelfie.site/# eva elfie photo
eva elfie videos: eva elfie new videos – eva elfie photo
lana rhoades hot: lana rhoades pics – lana rhoades solo
free local dates: https://lanarhoades.pro/# lana rhoades solo
eva elfie hot: eva elfie full video – eva elfie videos
https://sweetiefox.pro/# ph sweetie fox
lana rhoades videos: lana rhoades full video – lana rhoades hot
http://sweetiefox.pro/# ph sweetie fox
eva elfie new video: eva elfie photo – eva elfie full video
vip dating now: http://miamalkova.life/# mia malkova movie
lana rhoades: lana rhoades – lana rhoades boyfriend
aviator moçambique: aviator – jogar aviator
cassino pin up: pin-up casino entrar – pin-up casino
https://aviatorjogar.online/# jogar aviator online
http://aviatormalawi.online/# aviator
aviator bet malawi: aviator bet malawi login – aviator bet
https://aviatormalawi.online/# play aviator
long term effects of citalopram
http://pinupcassino.pro/# pin-up casino login
aviator bet malawi login: play aviator – play aviator
aviator mocambique: aviator mocambique – como jogar aviator
https://aviatoroyunu.pro/# pin up aviator
aviator betting game: aviator game online – aviator
https://aviatormocambique.site/# aviator bet
estrela bet aviator: aviator pin up – jogar aviator online
aviator oyunu: aviator oyunu – aviator oyna slot
aviator bet malawi login: aviator malawi – aviator game online
https://aviatormalawi.online/# aviator game
https://aviatormocambique.site/# como jogar aviator
aviator betting game: play aviator – aviator game
aviator game: aviator betano – aviator pin up
aviator: aviator bet – aviator game online
https://aviatorghana.pro/# aviator
aviator game: aviator – aviator
aviator oyunu: aviator sinyal hilesi – aviator hilesi
jogos que dao dinheiro: jogo de aposta – aplicativo de aposta
aviator bet malawi: aviator game – play aviator
aviator jogo de aposta: melhor jogo de aposta para ganhar dinheiro – jogos que dão dinheiro
http://aviatorghana.pro/# play aviator
como jogar aviator: jogar aviator – aviator bet
can you buy zithromax over the counter in canada: zithromax drug class zithromax prescription
estrela bet aviator: jogar aviator online – aviator bet
aviator game: aviator game online – aviator betting game
http://aviatorghana.pro/# aviator game
zithromax for sale 500 mg: buy zithromax online fast shipping – can you buy zithromax online
aviator jogar: aviator betano – aviator game
jogo de aposta: melhor jogo de aposta para ganhar dinheiro – aviator jogo de aposta
zithromax 1000 mg online: zithromax price south africa – where to get zithromax
https://aviatorghana.pro/# aviator betting game
https://mexicanpharm24.shop/# mexico drug stores pharmacies mexicanpharm.shop
canadian pharmacy checker: CIPA approved pharmacies – precription drugs from canada canadianpharm.store
mexican border pharmacies shipping to usa mexican mail order pharmacies buying from online mexican pharmacy mexicanpharm.shop
canadian pharmacy 24: Canada pharmacy online – canadian online pharmacy canadianpharm.store
http://indianpharm24.com/# Online medicine home delivery indianpharm.store
online pharmacy india: indianpharmacy com – pharmacy website india indianpharm.store
mexico pharmacies prescription drugs pharmacies in mexico that ship to usa mexican rx online mexicanpharm.shop
reputable indian pharmacies: india pharmacy – top 10 pharmacies in india indianpharm.store
http://canadianpharmlk.com/# canadapharmacyonline canadianpharm.store
http://mexicanpharm24.shop/# buying from online mexican pharmacy mexicanpharm.shop
canadian pharmacies online: Cheapest drug prices Canada – canada cloud pharmacy canadianpharm.store
http://indianpharm24.shop/# best india pharmacy indianpharm.store
http://canadianpharmlk.shop/# reliable canadian online pharmacy canadianpharm.store
https://indianpharm24.com/# top online pharmacy india indianpharm.store
http://mexicanpharm24.com/# п»їbest mexican online pharmacies mexicanpharm.shop
indian pharmacies safe: cheapest online pharmacy – top 10 pharmacies in india indianpharm.store
http://indianpharm24.com/# reputable indian online pharmacy indianpharm.store
http://mexicanpharm24.shop/# best online pharmacies in mexico mexicanpharm.shop
mexican pharmaceuticals online: mexican pharmacy – mexico pharmacies prescription drugs mexicanpharm.shop
http://mexicanpharm24.com/# mexican online pharmacies prescription drugs mexicanpharm.shop
http://mexicanpharm24.com/# mexican pharmaceuticals online mexicanpharm.shop
canadian pharmacy online store: pharmacy canadian superstore – canada pharmacy canadianpharm.store
https://mexicanpharm24.com/# mexican drugstore online mexicanpharm.shop
https://mexicanpharm24.shop/# mexico pharmacy mexicanpharm.shop
https://indianpharm24.shop/# india pharmacy indianpharm.store
top online pharmacy india: Best Indian pharmacy – best online pharmacy india indianpharm.store
http://indianpharm24.shop/# reputable indian online pharmacy indianpharm.store
legal canadian pharmacy online Best Canadian online pharmacy best canadian pharmacy canadianpharm.store
http://canadianpharmlk.shop/# canadian mail order pharmacy canadianpharm.store
https://indianpharm24.com/# online shopping pharmacy india indianpharm.store
https://indianpharm24.com/# Online medicine order indianpharm.store
medication from mexico pharmacy: mexico pharmacy – mexico drug stores pharmacies mexicanpharm.shop
canadian pharmacy in canada: Pharmacies in Canada that ship to the US – maple leaf pharmacy in canada canadianpharm.store
https://mexicanpharm24.com/# medication from mexico pharmacy mexicanpharm.shop
http://indianpharm24.shop/# indianpharmacy com indianpharm.store
https://mexicanpharm24.com/# mexico drug stores pharmacies mexicanpharm.shop
world pharmacy india: india pharmacy – indian pharmacy paypal indianpharm.store
https://canadianpharmlk.shop/# best mail order pharmacy canada canadianpharm.store
how to get prednisone tablets: prednisone 5 mg tablet price – buy prednisone 10mg online
prednisone 10 mg over the counter: prednisone tabs 20 mg – prednisone price south africa
can i purchase clomid now can i purchase cheap clomid prices where to get generic clomid pill
amoxicillin online purchase: amoxicillin dose for adults – amoxicillin capsule 500mg price
https://prednisonest.pro/# prednisone 50 mg tablet cost
amoxicillin pharmacy price: fish amoxicillin – amoxicillin 500mg cost
2.5 mg prednisone daily: prednisone dosage for poison ivy – prednisone brand name us
can i purchase generic clomid without dr prescription: where to buy clomid – cheap clomid pill
can you buy prednisone without a prescription: prednisone 20 mg – ordering prednisone
https://prednisonest.pro/# buy prednisone online australia
prednisone cost in india: can i order prednisone – order prednisone 10mg
amoxicillin 500mg no prescription amoxicillin 500 mg purchase without prescription how to buy amoxycillin
where to buy amoxicillin 500mg without prescription: antibiotic amoxicillin – generic amoxicillin cost
cost generic clomid no prescription: how long does it take for clomid to work in males – cost of clomid without insurance
buying cheap clomid online: alternative to clomid – clomid cheap
http://clomidst.pro/# can i order generic clomid without rx
amoxicillin 500 mg without a prescription: amoxicillin for sinus infection – can you buy amoxicillin over the counter in canada
cost of generic clomid no prescription: can i purchase generic clomid without prescription – where can i buy generic clomid without insurance
https://clomidst.pro/# can i buy cheap clomid
can i buy amoxicillin online: amoxicillin 500 mg – amoxicillin over counter
amoxicillin medicine: amoxicillin azithromycin – amoxicillin 500mg prescription
amoxicillin 500 mg tablet: can you buy amoxicillin uk – where can i get amoxicillin
http://clomidst.pro/# where to get clomid prices
amoxicillin without rx: side effects of amoxicillin – amoxicillin 500 tablet
where can i get clomid tablets can you buy generic clomid get clomid without rx
buy cheap clomid: cost of clomid – how to get clomid price
buy amoxicillin online without prescription: amoxicillin and clavulanate potassium tablets – buy amoxicillin 500mg uk
https://amoxilst.pro/# buy amoxicillin 500mg
prednisone 15 mg tablet: tylenol and prednisone – how to buy prednisone online
where to buy cheap clomid pill: clomid women – how to get cheap clomid pills
http://pharmnoprescription.pro/# no prescription canadian pharmacies
canada prescription: canadian rx prescription drugstore – online pharmacy no prescriptions
legitimate online pharmacy no prescription: buy medication online with prescription – buy medications online without prescription
mexican pharmacy no prescription: how to order prescription drugs from canada – canadian pharmacy no prescription needed
http://onlinepharmacy.cheap/# canadian pharmacy coupon code
https://onlinepharmacy.cheap/# mail order pharmacy no prescription
order ed meds online: cheapest ed medication – pills for ed online
canada prescription: best website to buy prescription drugs – online pharmacy without prescription
uk pharmacy no prescription: canada online pharmacy – us pharmacy no prescription
https://pharmnoprescription.pro/# non prescription online pharmacy india
cheap drugs no prescription: canada pharmacy online no prescription – canadian pharmacy no prescription
http://edpills.guru/# cheap ed treatment
effexor and night sweats
http://onlinepharmacy.cheap/# no prescription required pharmacy
canadian online pharmacy no prescription: online mexican pharmacy – uk pharmacy no prescription
online pharmacy no prescriptions: order prescription from canada – canada drugs without prescription
https://pharmnoprescription.pro/# ordering prescription drugs from canada
ed prescription online online erectile dysfunction prescription online erectile dysfunction
low cost ed meds: best online ed medication – ed prescription online
mail order prescription drugs from canada: online pharmacy delivery – online canadian pharmacy coupon
https://pharmnoprescription.pro/# online medication without prescription
prescription drugs canada: canada drugs without prescription – buying prescription medicine online
http://edpills.guru/# buy ed pills
best non prescription online pharmacy: buy medications without prescriptions – buy medication online no prescription
buy medicines online in india: buy medicines online in india – п»їlegitimate online pharmacies india
canada prescription drugs online online drugstore no prescription mexican pharmacy no prescription
buy prescription drugs from india: india pharmacy mail order – online pharmacy india
http://indianpharm.shop/# india pharmacy
https://indianpharm.shop/# world pharmacy india
mexican pharmacy: buying prescription drugs in mexico – medicine in mexico pharmacies
mexico pharmacy: purple pharmacy mexico price list – mexican pharmaceuticals online
https://canadianpharm.guru/# canadian pharmacies
mexican border pharmacies shipping to usa: medication from mexico pharmacy – medication from mexico pharmacy
best online pharmacy india: reputable indian pharmacies – reputable indian pharmacies
buying from online mexican pharmacy best online pharmacies in mexico mexican mail order pharmacies
canadian pharmacy phone number: buy drugs from canada – pharmacy canadian superstore
no prescription pharmacy online: no prescription online pharmacies – prescription meds from canada
https://pharmacynoprescription.pro/# best no prescription online pharmacy
http://pharmacynoprescription.pro/# online pharmacies no prescription usa
pharmacy website india: india pharmacy mail order – indianpharmacy com
canadian pharmacy prescription: canada prescription – canada drugs without prescription
https://mexicanpharm.online/# mexico pharmacy
online shopping pharmacy india: world pharmacy india – reputable indian online pharmacy
mexico pharmacies prescription drugs: best online pharmacies in mexico – medication from mexico pharmacy
buy drugs online no prescription: online prescription canada – best no prescription online pharmacy
canadian pharmacy non prescription: online medication no prescription – no prescription needed pharmacy
buying prescription drugs in mexico online reputable mexican pharmacies online pharmacies in mexico that ship to usa
http://indianpharm.shop/# best india pharmacy
mexico drug stores pharmacies: mexican rx online – medication from mexico pharmacy
mexican border pharmacies shipping to usa: mexican pharmaceuticals online – mexico drug stores pharmacies
india online pharmacy: Online medicine home delivery – pharmacy website india
http://indianpharm.shop/# best india pharmacy
buy pills without prescription: prescription drugs canada – buying prescription drugs in canada
Online medicine order: indianpharmacy com – india pharmacy mail order
best canadian pharmacy to buy from: canadian pharmacy ed medications – canadian family pharmacy
https://mexicanpharm.online/# buying prescription drugs in mexico
canadian drugs online: canadian pharmacy cheap – cheapest pharmacy canada
best online pharmacies in mexico mexican rx online buying from online mexican pharmacy
http://canadianpharm.guru/# canadian pharmacy no rx needed
aspirin or ibuprofen
indian pharmacy online: online pharmacy india – buy medicines online in india
https://canadianpharm.guru/# canadian pharmacies compare
buy pills without prescription: quality prescription drugs canada – online pharmacies without prescriptions
https://indianpharm.shop/# Online medicine home delivery
legitimate online pharmacies india: Online medicine home delivery – Online medicine order
canada drugs online: canadian pharmacy world reviews – thecanadianpharmacy
canadian pharmacy prescription: online no prescription pharmacy – discount prescription drugs canada
canadian rx prescription drugstore: no prescription needed pharmacy – no prescription
http://canadianpharm.guru/# canadapharmacyonline legit
canadian pharmacy: best canadian pharmacy – canadapharmacyonline legit
prescription drugs online canada: online pharmacy with prescription – best no prescription online pharmacies
https://mexicanpharm.online/# medicine in mexico pharmacies
canadian drugs online: canada drugs reviews – canada pharmacy reviews
https://indianpharm.shop/# indian pharmacies safe
buying prescription drugs in mexico: mexican mail order pharmacies – mexican border pharmacies shipping to usa
mexican drugstore online: mexico pharmacies prescription drugs – mexican mail order pharmacies
reaction to augmentin
mexican pharmaceuticals online: best online pharmacies in mexico – mexican online pharmacies prescription drugs
https://pharmacynoprescription.pro/# canada drugs without prescription
best mexican online pharmacies: reputable mexican pharmacies online – medicine in mexico pharmacies
reputable indian online pharmacy: india pharmacy mail order – buy medicines online in india
quality prescription drugs canada: online pharmacies without prescriptions – pharmacy online no prescription
canadian pharmacy ltd canadapharmacyonline legit adderall canadian pharmacy
http://pharmacynoprescription.pro/# no prescription
onlinecanadianpharmacy: trusted canadian pharmacy – certified canadian pharmacy
indian pharmacies safe: india pharmacy – world pharmacy india
http://pharmacynoprescription.pro/# best no prescription online pharmacy
http://pharmacynoprescription.pro/# mexican prescription drugs online
reputable indian pharmacies: Online medicine home delivery – buy prescription drugs from india
canadian pharmacy drugs online: www canadianonlinepharmacy – canadian pharmacy 365
top online pharmacy india: indian pharmacy paypal – indian pharmacies safe
buy meds online without prescription: canadian prescription prices – online doctor prescription canada
http://indianpharm.shop/# buy medicines online in india
http://aviatoroyna.bid/# aviator oyunu
sweet bonanza guncel: guncel sweet bonanza – sweet bonanza demo turkce
https://aviatoroyna.bid/# aviator hilesi
https://aviatoroyna.bid/# aviator oyunu 100 tl
aviator sinyal hilesi ucretsiz: ucak oyunu bahis aviator – aviator sinyal hilesi ucretsiz
https://aviatoroyna.bid/# aviator sinyal hilesi apk
gates of olympus nas?l para kazanilir: gates of olympus demo free spin – gates of olympus s?rlar?
http://slotsiteleri.guru/# yeni slot siteleri
baclofen for spasticity
http://sweetbonanza.bid/# sweet bonanza free spin demo
deneme bonusu veren siteler: bonus veren slot siteleri – 2024 en iyi slot siteleri
https://sweetbonanza.bid/# sweet bonanza yorumlar
https://pinupgiris.fun/# pin up casino indir
http://aviatoroyna.bid/# aviator nasil oynanir
slot siteleri guvenilir: yasal slot siteleri – slot casino siteleri
pin up 7/24 giris: pin up indir – pin up
http://aviatoroyna.bid/# aviator oyunu 20 tl
pragmatic play gates of olympus: gates of olympus oyna demo – pragmatic play gates of olympus
https://slotsiteleri.guru/# slot siteleri
http://slotsiteleri.guru/# 2024 en iyi slot siteleri
gates of olympus demo oyna: gates of olympus hilesi – gates of olympus giris
https://sweetbonanza.bid/# sweet bonanza indir
bonus veren slot siteleri: canl? slot siteleri – slot siteleri bonus veren
oyun siteleri slot: guvenilir slot siteleri 2024 – en guvenilir slot siteleri
https://gatesofolympus.auction/# gates of olympus hilesi
sweet bonanza slot: sweet bonanza kazanc – sweet bonanza nas?l oynan?r
https://sweetbonanza.bid/# sweet bonanza free spin demo
https://slotsiteleri.guru/# slot bahis siteleri
sweet bonanza yasal site: sweet bonanza giris – sweet bonanza giris
gates of olympus nas?l para kazanilir: gate of olympus hile – gates of olympus giris
https://slotsiteleri.guru/# deneme bonusu veren siteler
https://pinupgiris.fun/# pin up aviator
mexican rx online: Online Pharmacies in Mexico – pharmacies in mexico that ship to usa
canadian pharmacy 365 Large Selection of Medications legitimate canadian pharmacies
indian pharmacies safe Generic Medicine India to USA india online pharmacy
canada rx pharmacy world: canadian pharmacy world – legit canadian pharmacy online
п»їbest mexican online pharmacies: Online Pharmacies in Mexico – buying from online mexican pharmacy
http://mexicanpharmacy.shop/# mexico drug stores pharmacies
prescription drugs canada buy online canadapharmacyonline com best canadian pharmacy online
top 10 pharmacies in india: indian pharmacy – indian pharmacy paypal
canadian pharmacy online store: Licensed Canadian Pharmacy – canada pharmacy reviews
canadian medications Large Selection of Medications cheapest pharmacy canada
canada rx pharmacy world: Prescription Drugs from Canada – canada drugs online review
mexico drug stores pharmacies: cheapest mexico drugs – best mexican online pharmacies
canadian online drugs: Large Selection of Medications – best rated canadian pharmacy
safe canadian pharmacies: pills now even cheaper – canadian drug
best online pharmacies in mexico Online Pharmacies in Mexico buying prescription drugs in mexico
medicine in mexico pharmacies: mexican pharmacy – buying prescription drugs in mexico online
buy medicines online in india: indian pharmacy – cheapest online pharmacy india
canada drug pharmacy: pills now even cheaper – reputable canadian pharmacy
http://indianpharmacy.icu/# cheapest online pharmacy india
legitimate canadian mail order pharmacy pills now even cheaper canada rx pharmacy
canadian mail order pharmacy: canadian pharmacy 24 – safe reliable canadian pharmacy
ed meds online canada: Large Selection of Medications – online canadian drugstore
mexican drugstore online: Online Pharmacies in Mexico – mexico drug stores pharmacies
canadian mail order pharmacy: Licensed Canadian Pharmacy – the canadian drugstore
canadian neighbor pharmacy Large Selection of Medications canadian pharmacy tampa
top 10 pharmacies in india: indian pharmacy online – cheapest online pharmacy india
india pharmacy: Healthcare and medicines from India – world pharmacy india
п»їlegitimate online pharmacies india Healthcare and medicines from India п»їlegitimate online pharmacies india
https://mexicanpharmacy.shop/# mexican mail order pharmacies
best mail order pharmacy canada: pills now even cheaper – legitimate canadian pharmacies
buying prescription drugs in mexico online Online Pharmacies in Mexico mexican rx online
indian pharmacies safe: Generic Medicine India to USA – indian pharmacies safe
reliable canadian pharmacy: Licensed Canadian Pharmacy – canada drugs online review
top online pharmacy india indian pharmacy Online medicine order
mexico pharmacies prescription drugs: mexican pharmacy – mexican pharmacy
best online pharmacy india: Healthcare and medicines from India – top 10 pharmacies in india
top 10 online pharmacy in india: indian pharmacy delivery – buy prescription drugs from india
can i purchase amoxicillin online how to buy amoxicillin online buy cheap amoxicillin
amoxicillin azithromycin: order amoxicillin online uk – amoxicillin without prescription
http://prednisoneall.com/# mail order prednisone
http://clomidall.shop/# can you get clomid without a prescription
https://zithromaxall.com/# zithromax 500 price
can you buy cheap clomid no prescription can i buy generic clomid without a prescription cheap clomid price
generic zithromax india: zithromax 500 without prescription – zithromax antibiotic without prescription
http://amoxilall.com/# buying amoxicillin online
buy prednisone 10mg prednisone prescription drug prednisone for sale online
http://prednisoneall.shop/# can i buy prednisone online in uk
http://prednisoneall.com/# buy prednisone online no script
can i purchase prednisone without a prescription: prednisone 15 mg tablet – prednisone 20mg online without prescription
http://amoxilall.shop/# amoxicillin 500mg price in canada
where can i get zithromax zithromax antibiotic without prescription buy zithromax 1000mg online
http://prednisoneall.com/# prednisone
amoxicillin 750 mg price amoxicillin 800 mg price buy amoxicillin
https://zithromaxall.com/# purchase zithromax z-pak
buying amoxicillin in mexico: amoxicillin 500mg capsules uk – order amoxicillin no prescription
https://prednisoneall.shop/# prednisone 1mg purchase
http://clomidall.shop/# can you buy cheap clomid without rx
can i buy zithromax over the counter zithromax 500 mg lowest price online buy zithromax online fast shipping
https://amoxilall.com/# buy amoxicillin 500mg uk
https://prednisoneall.com/# prednisone pack
3000mg prednisone prednisone nz purchase prednisone from india
http://zithromaxall.com/# where can i buy zithromax uk
amoxicillin tablets in india: order amoxicillin 500mg – amoxicillin 500mg buy online uk
https://amoxilall.com/# amoxicillin online canada
can you purchase amoxicillin online amoxicillin 500mg pill generic amoxicillin
https://amoxilall.shop/# amoxicillin medicine
buy prednisone tablets uk: how much is prednisone 5mg – prednisone 300mg
http://prednisoneall.com/# buy prednisone without prescription
https://amoxilall.shop/# where can i buy amoxicillin without prec
prednisone buy prednisone 10mg tablet price price for 15 prednisone
http://zithromaxall.com/# buy zithromax online cheap
super kamagra: Kamagra gel – Kamagra 100mg price
Buy Tadalafil 5mg: cheapest cialis – Cialis without a doctor prescription
actos recurribles
http://tadalafiliq.com/# Cialis 20mg price in USA
super kamagra Sildenafil Oral Jelly buy Kamagra
Kamagra 100mg price: Kamagra 100mg price – kamagra
Generic Cialis without a doctor prescription Buy Cialis online Buy Cialis online
http://tadalafiliq.com/# Tadalafil price
http://tadalafiliq.shop/# Buy Cialis online
Viagra online price: buy viagra online – sildenafil 50 mg price
Cialis 20mg price: cialis without a doctor prescription – Buy Tadalafil 5mg
Kamagra 100mg price Kamagra Oral Jelly Price Kamagra 100mg
https://sildenafiliq.xyz/# sildenafil online
Buy Cialis online: cheapest cialis – cialis generic
cheapest cialis Cialis 20mg price Tadalafil price
Cheap generic Viagra: best price on viagra – cheapest viagra
http://sildenafiliq.com/# Viagra tablet online
http://kamagraiq.shop/# Kamagra 100mg price
https://tadalafiliq.com/# Cialis 20mg price in USA
buy viagra here: generic ed pills – Viagra online price
buy kamagra online usa: kamagra best price – buy Kamagra
Generic Viagra online cheapest viagra sildenafil over the counter
http://tadalafiliq.com/# buy cialis pill
Viagra tablet online: cheapest viagra – Sildenafil Citrate Tablets 100mg
over the counter sildenafil generic ed pills Cheap Viagra 100mg
Tadalafil price: cialis best price – cialis for sale
https://sildenafiliq.com/# Viagra online price
https://sildenafiliq.com/# Viagra online price
Cialis without a doctor prescription: cialis without a doctor prescription – Cialis 20mg price in USA
Tadalafil price cheapest cialis cheapest cialis
https://tadalafiliq.shop/# Generic Tadalafil 20mg price
Kamagra 100mg price: Sildenafil Oral Jelly – super kamagra
https://kamagraiq.com/# buy kamagra online usa
cheapest viagra: generic ed pills – Cheapest Sildenafil online
Tadalafil Tablet cialis without a doctor prescription Buy Tadalafil 10mg
cheapest cialis: tadalafil iq – Cheap Cialis
http://tadalafiliq.shop/# Buy Tadalafil 20mg
http://kamagraiq.shop/# super kamagra
Kamagra 100mg price: Sildenafil Oral Jelly – Kamagra Oral Jelly
Kamagra Oral Jelly Kamagra Iq buy kamagra online usa
Tadalafil Tablet: Tadalafil Tablet – Generic Cialis without a doctor prescription
https://kamagraiq.com/# Kamagra 100mg price
https://tadalafiliq.com/# Tadalafil Tablet
cialis for sale: cialis without a doctor prescription – Buy Tadalafil 20mg
Viagra generic over the counter generic ed pills Viagra without a doctor prescription Canada
https://sildenafiliq.com/# over the counter sildenafil
http://kamagraiq.shop/# super kamagra
over the counter sildenafil: buy viagra online – Cheap generic Viagra
http://mexicanpharmgrx.shop/# buying from online mexican pharmacy
mexican drugstore online: purple pharmacy mexico price list – buying from online mexican pharmacy
best india pharmacy: Healthcare and medicines from India – india online pharmacy
pharmacy wholesalers canada CIPA approved pharmacies canadian pharmacy
https://canadianpharmgrx.xyz/# best canadian online pharmacy reviews
reliable canadian pharmacy Pharmacies in Canada that ship to the US best online canadian pharmacy
mexican online pharmacies prescription drugs: mexican pharmacy – reputable mexican pharmacies online
http://indianpharmgrx.com/# india online pharmacy
canadian pharmacy 1 internet online drugstore Canada pharmacy online global pharmacy canada
reputable mexican pharmacies online: Mexico drugstore – mexico drug stores pharmacies
best mail order pharmacy canada: CIPA approved pharmacies – canadian pharmacy online
https://mexicanpharmgrx.com/# buying from online mexican pharmacy
mail order pharmacy india Generic Medicine India to USA india pharmacy mail order
http://canadianpharmgrx.xyz/# canadian pharmacy prices
mexican rx online: mexican pharmacy – mexican pharmacy
world pharmacy india indian pharmacy delivery india pharmacy mail order
world pharmacy india: Generic Medicine India to USA – indian pharmacy online
https://indianpharmgrx.com/# indianpharmacy com
online pharmacy india indian pharmacy delivery pharmacy website india
https://mexicanpharmgrx.com/# mexican pharmacy
mexican rx online: online pharmacy in Mexico – mexico drug stores pharmacies
http://mexicanpharmgrx.shop/# reputable mexican pharmacies online
indian pharmacy paypal: Healthcare and medicines from India – Online medicine home delivery
northwest canadian pharmacy Best Canadian online pharmacy pharmacy in canada
https://indianpharmgrx.shop/# pharmacy website india
http://mexicanpharmgrx.shop/# mexican pharmaceuticals online
mexican pharmacy mexican pharmacy п»їbest mexican online pharmacies
mexican rx online: online pharmacy in Mexico – mexican drugstore online
india pharmacy: Healthcare and medicines from India – Online medicine home delivery
https://mexicanpharmgrx.com/# buying from online mexican pharmacy
indian pharmacy indian pharmacy delivery top 10 pharmacies in india
canadian pharmacies comparison: CIPA approved pharmacies – canadian neighbor pharmacy
http://mexicanpharmgrx.com/# mexican online pharmacies prescription drugs
http://canadianpharmgrx.com/# canadian pharmacy review
canadian pharmacy ratings: canadian pharmacy – my canadian pharmacy
pharmacy website india Generic Medicine India to USA indian pharmacy
https://mexicanpharmgrx.com/# pharmacies in mexico that ship to usa
diflucan 200mg: diflucan brand name 300mg – over the counter diflucan 150
ciprofloxacin order online purchase cipro buy cipro online
buy cipro cheap: cipro for sale – cipro 500mg best prices
https://nolvadex.icu/# nolvadex only pct
tamoxifen and antidepressants: nolvadex side effects – nolvadex steroids
buy cipro online buy cipro cheap cipro 500mg best prices
diflucan otc uk: buy diflucan generic – diflucan tablet price in india
tamoxifen therapy: tamoxifen hot flashes – aromatase inhibitors tamoxifen
tamoxifen: tamoxifen alternatives premenopausal – tamoxifen
tamoxifen chemo tamoxifen citrate pct tamoxifen
cipro online no prescription in the usa: buy cipro cheap – buy cipro cheap
tamoxifen benefits tamoxifen for sale tamoxifen postmenopausal
diflucan from india: diflucan over the counter usa – can you buy diflucan without a prescription
https://diflucan.icu/# can i purchase diflucan over the counter
buy cipro: ciprofloxacin mail online – ciprofloxacin over the counter
nolvadex only pct: femara vs tamoxifen – tamoxifen premenopausal
doxycycline 100mg: doxycycline 200 mg – doxycycline 50 mg
diflucan pills online diflucan 1 pill diflucan without get a prescription online
diflucan singapore: diflucan otc uk – diflucan cost
buy cytotec in usa buy misoprostol over the counter purchase cytotec
cipro pharmacy: ciprofloxacin – buy cipro online canada
tamoxifen vs raloxifene: tamoxifen warning – tamoxifen for men
doxycycline generic buy doxycycline monohydrate doxycycline generic
diflucan pill for sale: diflucan buy without prescription – where can i buy diflucan otc
http://ciprofloxacin.guru/# buy cipro without rx
cipro: buy cipro online – cipro pharmacy
doxycycline hydrochloride 100mg: doxycycline 200 mg – generic doxycycline
п»їcytotec pills online Abortion pills online cytotec buy online usa
buy ciprofloxacin over the counter: buy ciprofloxacin – ciprofloxacin order online
buy doxycycline online 270 tabs: buy cheap doxycycline – buy cheap doxycycline
diflucan medicine in india diflucan 200 mg price how to get diflucan online
doxycycline mono: buy doxycycline without prescription – buy generic doxycycline
doxycycline without prescription: order doxycycline – online doxycycline
nolvadex only pct: tamoxifen dosage – tamoxifen for breast cancer prevention
tamoxifen alternatives tamoxifen hip pain natural alternatives to tamoxifen
https://ciprofloxacin.guru/# buy cipro online usa
odering doxycycline: buy doxycycline online – odering doxycycline
generic diflucan prices: diflucan price in india – medication diflucan price
what happens when you stop taking tamoxifen does tamoxifen cause joint pain nolvadex 10mg
doxycycline hyc: doxycycline 100mg online – vibramycin 100 mg
can i buy prednisone over the counter in usa: where can i order prednisone 20mg – prednisone 1 mg daily
otc prednisone cream: prednisone 20mg prices – 54 prednisone
antibiotic amoxicillin buy amoxicillin 500mg capsules uk order amoxicillin online no prescription
amoxicillin generic brand: generic amoxicillin online – amoxicillin 500mg capsule cost
where to buy clomid pill: clomid without a prescription – can you get generic clomid tablets
zithromax capsules australia zithromax 500 mg lowest price pharmacy online zithromax 500 mg for sale
https://amoxicillina.top/# buying amoxicillin in mexico
prednisone generic brand name: prednisone 5mg price – buy prednisone 50 mg
how to buy clomid without prescription: where to get cheap clomid price – how to get generic clomid pills
order amoxicillin uk: buy amoxicillin 500mg canada – amoxicillin 250 mg
generic prednisone cost prednisone online for sale prednisone otc price
http://stromectola.top/# stromectol 3 mg dosage
ivermectin over the counter uk: stromectol sales – ivermectin buy uk
https://clomida.pro/# how can i get clomid prices
zithromax order online uk zithromax purchase online where to get zithromax
875 mg amoxicillin cost: amoxicillin cephalexin – buy amoxicillin online with paypal
can i buy zithromax online: zithromax coupon – where can i purchase zithromax online
purchase amoxicillin 500 mg: generic amoxil 500 mg – buy amoxicillin 500mg usa
amoxicillin order online: amoxicillin 500mg capsules uk – amoxicillin 500 mg price
http://prednisonea.store/# average cost of prednisone 20 mg
amoxicillin 500mg buy online uk order amoxicillin online how much is amoxicillin
http://amoxicillina.top/# can i buy amoxicillin over the counter
ivermectin pills: stromectol medicine – purchase stromectol
http://edpill.top/# best online ed meds
https://onlinepharmacyworld.shop/# canadian pharmacy without prescription
best website to buy prescription drugs buy prescription drugs on line buy medications online no prescription
buying erectile dysfunction pills online: cheapest ed online – cheap ed drugs
best online ed meds: ed med online – where to buy ed pills
https://onlinepharmacyworld.shop/# international pharmacy no prescription
canadian pharmacy no prescription best online pharmacy no prescription pharmacy discount coupons
http://onlinepharmacyworld.shop/# cheapest pharmacy to fill prescriptions without insurance
https://edpill.top/# top rated ed pills
http://medicationnoprescription.pro/# buy medication online with prescription
online pharmacy no prescription needed: canadian pharmacies not requiring prescription – no prescription needed canadian pharmacy
https://edpill.top/# erectile dysfunction medications online
buy erectile dysfunction pills where can i get ed pills online ed pills
pharmacies without prescriptions: no prescription drugs online – canadian pharmacy without a prescription
http://edpill.top/# buy ed medication
ed prescriptions online: ed treatment online – where can i buy ed pills
https://edpill.top/# erectile dysfunction online
low cost ed medication online ed pharmacy buy ed medication online
canadian prescription: online drugstore no prescription – prescription meds from canada
https://onlinepharmacyworld.shop/# pharmacy no prescription required
https://edpill.top/# buy ed meds
http://edpill.top/# ed online meds
canada prescription online: canadian prescription drugstore review – canada prescription drugs online
canadian pharmacy without prescription canadian pharmacy discount coupon online pharmacy prescription
casino tr?c tuy?n vi?t nam: casino online uy tín – casino tr?c tuy?n vi?t nam
http://casinvietnam.com/# casino tr?c tuy?n vi?t nam
casino tr?c tuy?n vi?t nam: casino online uy tín – game c? b?c online uy tín
choi casino tr?c tuy?n tren di?n tho?i game c? b?c online uy tin casino online uy tin
https://casinvietnam.shop/# web c? b?c online uy tin
http://casinvietnam.shop/# casino online uy tin
https://casinvietnam.com/# casino tr?c tuy?n vi?t nam
casino tr?c tuy?n: casino online uy tín – casino tr?c tuy?n vi?t nam
casino tr?c tuy?n vi?t nam: dánh bài tr?c tuy?n – web c? b?c online uy tín
http://casinvietnam.com/# danh bai tr?c tuy?n
http://casinvietnam.com/# web c? b?c online uy tin
tizanidine dosage for back pain
casino tr?c tuy?n: web c? b?c online uy tín – casino tr?c tuy?n
http://casinvietnam.shop/# casino tr?c tuy?n vi?t nam
casino tr?c tuy?n vi?t nam: dánh bài tr?c tuy?n – casino tr?c tuy?n vi?t nam
casino online uy tín: choi casino tr?c tuy?n trên di?n tho?i – game c? b?c online uy tín
casino tr?c tuy?n uy tin game c? b?c online uy tin casino tr?c tuy?n uy tin
casino tr?c tuy?n: casino tr?c tuy?n vi?t nam – web c? b?c online uy tín
https://casinvietnam.shop/# casino tr?c tuy?n uy tin
casino tr?c tuy?n vi?t nam: game c? b?c online uy tín – casino tr?c tuy?n
dánh bài tr?c tuy?n: choi casino tr?c tuy?n trên di?n tho?i – dánh bài tr?c tuy?n
http://casinvietnam.com/# choi casino tr?c tuy?n tren di?n tho?i
http://casinvietnam.com/# web c? b?c online uy tin
casino tr?c tuy?n uy tín: casino online uy tín – game c? b?c online uy tín
https://casinvietnam.shop/# casino tr?c tuy?n vi?t nam
casino online uy tín: game c? b?c online uy tín – game c? b?c online uy tín
casino tr?c tuy?n vi?t nam casino tr?c tuy?n vi?t nam casino tr?c tuy?n vi?t nam
http://casinvietnam.shop/# casino tr?c tuy?n vi?t nam
casino tr?c tuy?n vi?t nam: web c? b?c online uy tín – dánh bài tr?c tuy?n
http://indiaph24.store/# legitimate online pharmacies india
http://indiaph24.store/# Online medicine order
https://mexicoph24.life/# mexican pharmaceuticals online
https://mexicoph24.life/# buying prescription drugs in mexico
http://mexicoph24.life/# medicine in mexico pharmacies
indian pharmacies safe indian pharmacy п»їlegitimate online pharmacies india
https://indiaph24.store/# mail order pharmacy india
http://mexicoph24.life/# mexican online pharmacies prescription drugs
mexico drug stores pharmacies mexican rx online pharmacies in mexico that ship to usa
http://indiaph24.store/# reputable indian pharmacies
https://canadaph24.pro/# pharmacy canadian
cheapest online pharmacy india indian pharmacy fast delivery indian pharmacy paypal
http://mexicoph24.life/# pharmacies in mexico that ship to usa
https://canadaph24.pro/# canadian pharmacy prices
http://mexicoph24.life/# best mexican online pharmacies
mexico drug stores pharmacies mexican pharmacy mexico drug stores pharmacies
https://indiaph24.store/# buy prescription drugs from india
https://canadaph24.pro/# canadian drug
Online medicine order Generic Medicine India to USA top 10 pharmacies in india
http://indiaph24.store/# mail order pharmacy india
https://canadaph24.pro/# best rated canadian pharmacy
https://canadaph24.pro/# canada cloud pharmacy
indian pharmacy online п»їlegitimate online pharmacies india world pharmacy india
https://mexicoph24.life/# mexican pharmaceuticals online
http://indiaph24.store/# indian pharmacy
https://indiaph24.store/# indian pharmacy online
cross border pharmacy canada Licensed Canadian Pharmacy canadianpharmacyworld
https://indiaph24.store/# indian pharmacy paypal
http://mexicoph24.life/# buying prescription drugs in mexico online
http://mexicoph24.life/# best mexican online pharmacies
https://canadaph24.pro/# canada rx pharmacy
http://indiaph24.store/# indian pharmacies safe
https://indiaph24.store/# reputable indian online pharmacy
my canadian pharmacy rx canadian pharmacies reputable canadian online pharmacies
https://canadaph24.pro/# canadian pharmacy 365
https://indiaph24.store/# best online pharmacy india
https://mexicoph24.life/# mexican pharmaceuticals online
http://mexicoph24.life/# mexico drug stores pharmacies
https://mexicoph24.life/# purple pharmacy mexico price list
https://mexicoph24.life/# buying prescription drugs in mexico online
https://indiaph24.store/# india online pharmacy
india pharmacy mail order https://indiaph24.store/# best india pharmacy
online shopping pharmacy india
https://canadaph24.pro/# canadian online pharmacy
http://indiaph24.store/# buy medicines online in india
http://canadaph24.pro/# canadian pharmacy tampa
canadian discount pharmacy Licensed Canadian Pharmacy best canadian online pharmacy
https://indiaph24.store/# online pharmacy india
https://mexicoph24.life/# mexican drugstore online
http://canadaph24.pro/# canadian pharmacy review
http://indiaph24.store/# world pharmacy india
http://mexicoph24.life/# buying prescription drugs in mexico
https://mexicoph24.life/# pharmacies in mexico that ship to usa
cheapest online pharmacy india https://indiaph24.store/# indian pharmacies safe
top 10 online pharmacy in india
https://indiaph24.store/# indian pharmacy paypal
pharmacy website india https://indiaph24.store/# indian pharmacy online
buy prescription drugs from india
https://indiaph24.store/# pharmacy website india
https://indiaph24.store/# buy prescription drugs from india
medicine in mexico pharmacies: mexico pharmacy – purple pharmacy mexico price list
reputable canadian pharmacy Licensed Canadian Pharmacy canadian world pharmacy
mexico drug stores pharmacies Online Pharmacies in Mexico best online pharmacies in mexico
https://mexicoph24.life/# pharmacies in mexico that ship to usa
https://mexicoph24.life/# mexico pharmacy
https://finasteride.store/# buying generic propecia without insurance
https://ciprofloxacin.tech/# buy cipro cheap
buying cheap propecia without rx: propecia without prescription – cost propecia without a prescription
buy cheap propecia prices: cost of cheap propecia without rx – buy propecia now
https://ciprofloxacin.tech/# purchase cipro
lisinopril 5 mg for sale lisinopril 20 mg coupon how much is lisinopril 10 mg
lisinopril capsule lisinopril 10 mg tablets price where to buy lisinopril 2.5 mg
http://nolvadex.life/# tamoxifen dosage
buy cheap propecia pill: cheap propecia no prescription – generic propecia online
buy propecia without a prescription cost propecia without rx cost cheap propecia without insurance
https://ciprofloxacin.tech/# cipro 500mg best prices
nolvadex for sale tamoxifen postmenopausal tamoxifen rash pictures
purchase cytotec: Misoprostol 200 mg buy online – purchase cytotec
https://ciprofloxacin.tech/# cipro
buying lisinopril in mexico: lisinopril 20mg discount – 90 lisinopril
buy propecia without a prescription generic propecia pill generic propecia tablets
ciprofloxacin cipro pharmacy cipro 500mg best prices
http://finasteride.store/# buying propecia no prescription
https://lisinopril.network/# prinivil price
http://ciprofloxacin.tech/# cipro pharmacy
liquid tamoxifen: tamoxifen pill – tamoxifen citrate
http://finasteride.store/# generic propecia for sale
cost of generic propecia online cost propecia online order propecia pills
zestoretic price prinivil online lisinopril 5mg cost
cytotec pills online: cytotec buy online usa – buy cytotec pills
http://nolvadex.life/# tamoxifen medication
cheap propecia: cost of generic propecia without insurance – order cheap propecia without insurance
nolvadex pct is nolvadex legal femara vs tamoxifen
https://nolvadex.life/# nolvadex 10mg
where can i get lisinopril lisinopril 5 mg buy lisinopril 20 mg generic
cost of generic propecia for sale: get generic propecia price – buy cheap propecia pills
http://lisinopril.network/# can you buy lisinopril
http://nolvadex.life/# tamoxifen and osteoporosis
http://cytotec.club/# buy cytotec in usa
tamoxifen postmenopausal: where to get nolvadex – tamoxifen and grapefruit
ciprofloxacin cipro ciprofloxacin buy cipro cheap
lisinopril 10 mg price in india lisinopril 10 mg no prescription generic zestril
https://lisinopril.network/# lisinopril generic price
cost cheap propecia prices: propecia tablet – propecia without rx
buy cytotec online purchase cytotec buy cytotec
ciprofloxacin over the counter: antibiotics cipro – ciprofloxacin generic
tamoxifen citrate tamoxifen headache nolvadex only pct
https://cytotec.club/# Abortion pills online
generic propecia without dr prescription: generic propecia without prescription – buy cheap propecia online
does tamoxifen cause menopause tamoxifen and bone density nolvadex for sale amazon
https://lisinopril.network/# buy lisinopril 20 mg online canada
buy cheap propecia pills get propecia without rx cost of cheap propecia without insurance
buy cheap propecia without rx: cost of generic propecia without rx – propecia generic
https://lisinopril.network/# lisinopril 12.5
https://cytotec.club/# buy misoprostol over the counter
http://cytotec.club/# buy cytotec online fast delivery
tamoxifen headache: tamoxifen alternatives – how to prevent hair loss while on tamoxifen
buying cheap propecia price buying propecia for sale generic propecia without rx
https://cytotec.club/# buy cytotec over the counter
tamoxifen and osteoporosis: clomid nolvadex – tamoxifen effectiveness
https://lisinopril.network/# lisinopril 40 mg on line
cipro online no prescription in the usa buy cipro online without prescription buy ciprofloxacin
get generic propecia pills propecia generics propecia prices
does tamoxifen cause menopause: nolvadex price – pct nolvadex
http://ciprofloxacin.tech/# buy ciprofloxacin
rx lisinopril 10mg: can you order lisinopril online – how much is lisinopril 40 mg
https://nolvadex.life/# tamoxifen vs clomid
zestoretic canada 3 lisinopril lisinopril online uk
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
buy generic ciprofloxacin cipro for sale purchase cipro
https://ciprofloxacin.tech/# ciprofloxacin mail online
https://ciprofloxacin.tech/# buy cipro online without prescription
where can i buy cipro online: buy generic ciprofloxacin – buy cipro online without prescription
https://lisinopril.network/# drug lisinopril
nolvadex steroids: nolvadex gynecomastia – where to get nolvadex
ciprofloxacin 500mg buy online ciprofloxacin order online ciprofloxacin
cytotec abortion pill buy cytotec pills online cheap buy cytotec over the counter
http://finasteride.store/# cost of cheap propecia without rx
buy cytotec: Misoprostol 200 mg buy online – buy cytotec online
https://cytotec.club/# buy misoprostol over the counter
Brand slot games are always a big hit, with celebrities like Britney Spears, Ellen Degeneres and Dolly Parton having released their own games. So, Gaming Art had to really think outside of the box in order to put a new spin on celeb-centric slot machines. We’re sure that Brian Christopher’s slot machine will start a new trend with other influences creating their own branded games soon. In addition to revenue generated directly from YouTube (which Richter says, for a popular channel, can reach as high as $20,000 a month) both Richter and Christopher sell a range of merchandise, from bobbleheads to coffee mugs to shot glasses. While this may have started out as a hobby, it has clearly become something bigger. Christopher, for instance, found out about his channel being removed while traveling for a planned multi-state East Coast tour.
https://www.olivavesteinpelle.com/how-an-incredible-ai-had-reduce-4-world/
Best Riverslot Gaming Slots for Real MoneyRiverslot offers bonuses and loyalty programs when playing for real money. As a result, you’ll earn extra benefits additionally to the large wins when playing these popular slots: Play Free Riverslot Gaming Slots OnlineRiverslot games are often played for free of charge online in practice mode. they’re easily accessible using all kinds of devices and are fast loading. Here are our top recommendations for Riverslot Gaming slots to play for free: A professional gaming software requires specific qualities. Here you’ll find holistic software capable of tightening the bond between player, games, and the sweepstakes. What else could you ask for? Our River slots casino app is here to imrpove the mobile gaming experience for our dedicated customers. If you want to play river slots without any restriction and access the most profitable offers, you need to go onboard with the River Slots Sweepstakes app. A wide range of interactive sweepstakes games as well as numerous bonus offers makes the river slots casino app an irresistible product for players.
tamoxifen vs raloxifene: does tamoxifen cause joint pain – tamoxifen and osteoporosis
ciprofloxacin mail online cipro for sale ciprofloxacin generic price
Cytotec 200mcg price cytotec abortion pill buy cytotec
buy Levitra over the counter: Buy generic Levitra online – Levitra 10 mg best price
http://levitrav.store/# Generic Levitra 20mg
http://cenforce.pro/# cenforce for sale
Generic Viagra for sale: Cheapest place to buy Viagra – Cheapest Sildenafil online
https://levitrav.store/# п»їLevitra price
Generic Levitra 20mg Cheap Levitra online Buy generic Levitra online
Levitra online USA fast: Cheap Levitra online – Buy Vardenafil online
order cenforce cenforce.pro buy cenforce
order cenforce: Cenforce 100mg tablets for sale – cenforce for sale
https://cialist.pro/# Cialis over the counter
Kamagra 100mg: kamagra.win – Kamagra Oral Jelly
super kamagra: kamagra.win – sildenafil oral jelly 100mg kamagra
Viagra tablet online Buy generic 100mg Viagra online Buy Viagra online cheap
http://levitrav.store/# Levitra 10 mg buy online
Vardenafil price buy Levitra over the counter Cheap Levitra online
https://kamagra.win/# Kamagra 100mg
https://cialist.pro/# Buy Tadalafil 10mg
Buy Levitra 20mg online: Cheap Levitra online – Levitra 20 mg for sale
Cenforce 100mg tablets for sale order cenforce cenforce.pro
buy Levitra over the counter: levitrav.store – Cheap Levitra online
Buy Cialis online cialist.pro Cialis over the counter
http://cialist.pro/# Cialis 20mg price in USA
https://kamagra.win/# buy Kamagra
https://cenforce.pro/# buy cenforce
https://kamagra.win/# Kamagra tablets
over the counter sildenafil: Buy Viagra online cheap – viagra without prescription
Cialis over the counter: Cialis 20mg price in USA – Cialis 20mg price in USA
https://viagras.online/# sildenafil online
cheap kamagra kamagra п»їkamagra
Cenforce 100mg tablets for sale buy cenforce Cenforce 100mg tablets for sale
cenforce for sale: cheapest cenforce – Cenforce 100mg tablets for sale
Kamagra 100mg price: kamagra pills – Kamagra 100mg price
https://viagras.online/# sildenafil over the counter
Cenforce 100mg tablets for sale Purchase Cenforce Online order cenforce
sildenafil over the counter: Buy Viagra online cheap – buy Viagra over the counter
http://viagras.online/# Viagra online price
cialis for sale Generic Cialis without a doctor prescription Buy Cialis online
super kamagra: buy kamagra online – Kamagra 100mg
https://levitrav.store/# Levitra online pharmacy
Levitra 10 mg best price Buy generic Levitra online Levitra 20 mg for sale
Vardenafil buy online Buy Vardenafil 20mg Levitra generic best price
Levitra price: levitrav.store – Levitra generic best price
http://viagras.online/# Generic Viagra for sale
https://levitrav.store/# Buy generic Levitra online
https://viagras.online/# Buy Viagra online cheap
https://levitrav.store/# Buy generic Levitra online
Levitra tablet price: Levitra generic price – Vardenafil buy online
Levitra online USA fast: Buy Vardenafil 20mg – Buy Vardenafil online
no prescription pharmacy paypal: online pharmacy – rxpharmacycoupons
legal online pharmacy coupon code: pharm world – canadian online pharmacy no prescription
http://pharmcanada.shop/# canada drugs online
mexico pharmacies prescription drugs: п»їbest mexican online pharmacies – mexico drug stores pharmacies
indian pharmacies safe buy prescription drugs from india india pharmacy
canadian pharmacies not requiring prescription: online pharmacy – canada drugs coupon code
cheapest pharmacy for prescription drugs online pharmacy no prescription needed pharmacy
canadian online drugstore: canadian drug pharmacy – my canadian pharmacy reviews
https://pharmmexico.online/# mexican border pharmacies shipping to usa
no prescription needed: buy pain meds online without prescription – online pharmacy that does not require a prescription
non prescription medicine pharmacy: pharm world – canadian pharmacy coupon code
indianpharmacy com top 10 online pharmacy in india online pharmacy india
https://pharmindia.online/# india pharmacy mail order
canadian rx prescription drugstore canadian pharmacy without prescription mail order prescriptions from canada
https://pharmnoprescription.icu/# canada mail order prescriptions
best online pharmacy india: india pharmacy – india pharmacy mail order
pharmacies in mexico that ship to usa mexican online pharmacies prescription drugs reputable mexican pharmacies online
no prescription canadian pharmacy: cheap drugs no prescription – buy drugs online without prescription
buy medicines online in india cheapest online pharmacy india best online pharmacy india
no prescription online pharmacies: medication online without prescription – online pharmacy that does not require a prescription
https://pharmnoprescription.icu/# can you buy prescription drugs in canada
mexican rx online: medicine in mexico pharmacies – mexican border pharmacies shipping to usa
https://pharmnoprescription.icu/# medications online without prescription
online pharmacy india: online pharmacy india – top 10 online pharmacy in india
online shopping pharmacy india: indian pharmacy online – buy medicines online in india
http://pharmmexico.online/# reputable mexican pharmacies online
buying prescription drugs in mexico online mexican drugstore online medicine in mexico pharmacies
canadian online drugstore canadian pharmacy online store canada ed drugs
onlinepharmaciescanada com: is canadian pharmacy legit – legitimate canadian online pharmacies
https://pharmworld.store/# international pharmacy no prescription
canadian pharmacy world coupon: cheapest pharmacy – canadian pharmacy without prescription
http://pharmnoprescription.icu/# best online pharmacy no prescription
best online pharmacy india: india online pharmacy – top online pharmacy india
meds online no prescription canadian drugs no prescription online pharmacies without prescriptions
rx pharmacy coupons online pharmacy international pharmacy no prescription
canada pharmacy online no prescription: buying drugs online no prescription – non prescription canadian pharmacy
http://pharmnoprescription.icu/# best online pharmacy without prescription
canadian prescription: online doctor prescription canada – medications online without prescriptions
http://pharmindia.online/# top 10 pharmacies in india
mexico drug stores pharmacies: buying prescription drugs in mexico – purple pharmacy mexico price list
online canadian pharmacy: best canadian online pharmacy reviews – canadian pharmacies compare
top 10 pharmacies in india: india pharmacy – buy prescription drugs from india
online pharmacy no prescription cheapest pharmacy cheap pharmacy no prescription
https://pharmnoprescription.icu/# no prescription canadian pharmacy
rxpharmacycoupons pharm world store uk pharmacy no prescription
no prescription prednisone canadian pharmacy: prednisone for sale in canada – 20 mg prednisone tablet
neurontin generic brand: gabapentin 300 – price of neurontin
drug prices prednisone: buying prednisone mexico – prednisone 20mg price in india
https://prednisoned.online/# prednisone tablets
neurontin cream where can i buy neurontin from canada neurontin 500 mg tablet
prednisone 10 mg tablets: prednisone 50 mg coupon – 200 mg prednisone daily
prednisone 300mg order prednisone from canada prednisone 20 mg pill
where can i buy zithromax uk: buy zithromax canada – buy generic zithromax online
https://doxycyclinea.online/# generic doxycycline
generic gabapentin: prescription price for neurontin – neurontin 600mg
15 mg prednisone daily: prednisone 20 tablet – prednisone for sale without a prescription
https://prednisoned.online/# prednisone 5443
neurontin 10 mg neurontin 500 mg generic neurontin cost
amoxicillin 500mg no prescription: amoxicillin brand name – amoxicillin over the counter in canada
SightCare formula aims to maintain 20/20 vision without the need for any surgical process. This supplement is a perfect solution for people facing issues as they grow older. https://sightcare-web.com/
buy doxycycline online uk: doxycycline without a prescription – doxycycline tetracycline
amoxicillin 50 mg tablets: cost of amoxicillin – order amoxicillin online uk
zithromax 500 mg lowest price pharmacy online: cost of generic zithromax – purchase zithromax z-pak
medication neurontin 300 mg: 32 neurontin – buy neurontin 100 mg canada
zithromax 250 mg tablet price: zithromax 600 mg tablets – order zithromax without prescription
buy doxycycline monohydrate: buy doxycycline 100mg – buy generic doxycycline
zithromax 500 tablet: where can you buy zithromax – buy zithromax without presc
https://amoxila.pro/# amoxicillin capsules 250mg
zithromax 600 mg tablets: zithromax 500 price – zithromax cost
https://gabapentinneurontin.pro/# neurontin 100 mg
doxycycline monohydrate doxycycline 100mg capsules doxycycline 100mg price
prednisone 30 mg daily where to buy prednisone without prescription prednisone 5mg price
generic for doxycycline: doxycycline hydrochloride 100mg – doxycycline order online
neurontin 100: neurontin 400 mg capsules – neurontin uk
generic zithromax azithromycin: zithromax generic price – zithromax 500mg
http://prednisoned.online/# buy prednisone online from canada
zithromax 600 mg tablets: zithromax generic cost – buy zithromax 1000 mg online
http://gabapentinneurontin.pro/# neurontin generic brand
buy brand neurontin: generic neurontin pill – neurontin prescription cost
buy doxycycline without prescription uk doxycycline mono doxycycline tablets
cheapest generic levitra
neurontin 4000 mg neurontin 2018 neurontin 100 mg
zithromax cost canada: generic zithromax 500mg india – zithromax without prescription
buy doxycycline cheap: doxycycline generic – buy cheap doxycycline
https://amoxila.pro/# where can i buy amoxicillin online
neurontin 100mg price: neurontin 800 mg tablet – neurontin 600 mg coupon
PCWer is here to help you with all questions in tech. Whether that
Find the latest technology news and expert tech product reviews. Learn about the latest gadgets and consumer tech products for entertainment, gaming, lifestyle and more. https://axget.com/
Easier WWW is a leading technology site that is dedicated to produce great how-to, tips and tricks and cool software review. https://easierwww.com/
Testosil is a natural polyherbal testosterone booster designed to help men increase their testosterone levels safely and effectively. https://testosil-web.com/
FitSpresso is a natural dietary supplement designed to help with weight loss and improve overall health. It contains ingredients that have been studied clinically, which work together to promote healthy fat burning and enhance your metabolism! https://fitspresso-web.com/
KeraBiotics is a meticulously-crafted natural formula designed to help people dealing with nail fungus. This solution, inspired by a sacred Amazonian barefoot tribe ritual https://kerabiotics-web.com/
Nagano Lean Body Tonic is a groundbreaking powdered supplement crafted to support your weight loss journey effortlessly. https://naganotonic-try.com/
Sugar Defender is a natural supplement that helps control blood sugar levels, lower the risk of diabetes, improve heart health, and boost energy. https://sugardefender-web.com/
– Shoot MASSIVE Loads For An Amazing Finish! https://semenax-try.com/
ZenCortex Research’s contains only the natural ingredients that are effective in supporting incredible hearing naturally.A unique team of health and industry professionals dedicated to unlocking the secrets of happier living through a healthier body. https://zencortex-try.com/
Serolean, a revolutionary weight loss supplement, zeroes in on serotonin—the key neurotransmitter governing mood, appetite, and fat storage. https://serolean-web.com/
generic amoxicillin online amoxicillin medicine over the counter amoxicillin generic brand
Tonic Greens is a ready-made greens shake designed to support the entire body and wellness of the mind. It is filled with over 50 individual vitamins https://tonicgreens-try.com/
http://prednisoned.online/# prednisone pill
buy doxycycline online uk 200 mg doxycycline doxycycline
Support the health of your ears with 100% natural ingredients, finally being able to enjoy your favorite songs and movies https://quietumplus-try.com/
Peak BioBoost is a revolutionary dietary supplement that leverages the power of nature to support and improve your digestive system. https://peakbioboost-web.com/
GutOptim is a digestive health supplement designed to support your gut and stomach. It restore balance in gut flora and reduce the symptoms of digestive disorders. https://gutoptim-try.com/
amoxicillin where to get: buy amoxicillin online no prescription – purchase amoxicillin online
Burn Boost Powder™ is a proven weight loss powder drink that helps to lose weight and boosts the overall metabolism in the body. https://burnboost-web.com
ONLINE EXCLUSIVE OFFER! Only Available for purchase on the official website. Secure Your Package while stocks last https://prodentim-web.com
http://zithromaxa.store/# zithromax over the counter uk
medicine neurontin: neurontin cost in singapore – neurontin capsules 300mg
neurontin prices buy neurontin canadian pharmacy neurontin
buy prednisone mexico 10 mg prednisone tablets over the counter prednisone pills
amoxicillin 500 mg tablet price: purchase amoxicillin online – buy amoxicillin over the counter uk
prednisone price canada: prednisone 10 mg price – prednisone 1 tablet
https://prednisoned.online/# prednisone 60 mg tablet
buy neurontin canadian pharmacy: drug neurontin – canada neurontin 100mg lowest price
https://doxycyclinea.online/# purchase doxycycline online
FlowForce Max is an innovative, natural and effective way to address your prostate problems, while addressing your energy, libido, and vitality. https://flowforcemax-web.com/
order zithromax without prescription: zithromax z-pak – zithromax 500mg
cost of prednisone: where to buy prednisone in australia – prednisone 5443
TestRX™ is a bodybuilding supplement. It’s formulated with high-quality natural ingredients proven to boost natural testosterone and stimulate muscle growth. https://testrx-web.com/
amoxicillin online canada amoxicillin 500 mg for sale amoxicillin 500 capsule
DuoTrim is an innovative weight loss supplement that utilizes the power of natural plants and nutrients to create CSM bacteria https://duotrim-us.com/
BioFit is a Nutritional Supplement That Uses Probiotics To Help You Lose Weight https://biofit-web.com/
prednisone 5mg daily buy prednisone without a prescription prednisone 20mg nz
Dentitox Pro is a liquid dietary solution created as a serum to support healthy gums and teeth. Dentitox Pro formula is made in the best natural way with unique, powerful botanical ingredients that can support healthy teeth. https://dentitox-us.com/
order amoxicillin online no prescription: amoxicillin no prescipion – amoxicillin 825 mg
http://prednisoned.online/# prednisone 20mg capsule
Sugar Balance is an ultra-potent blood sugar supplement that you can use to help control glucose levels, melt away fat and improve your overall health. https://sugarbalance-us.com/
PureLumin Essence is a meticulously-crafted natural formula designed to help women improve the appearance of age spots. https://pureluminessence-web.com/
GlucoFlush is an advanced formula specially designed for pancreas support that will let you promote healthy weight by effectively maintaining the blood sugar level and cleansing and strengthening your gut. https://glucoflush-us.com/
Alpha Tonic is a powder-based supplement that uses multiple natural herbs and essential vitamins and minerals to help optimize your body’s natural testosterone levels. https://alphatonic-web.com
Vivo Tonic is a remarkable blood sugar support nutritional supplement that offers a wide range of benefits. https://vivotonic-web.com/
neurontin: neurontin 800 mg price – drug neurontin
Nervogen Pro is an effective dietary supplement designed to help patients with neuropathic pain. When you combine exotic herbs, spices, and other organic substances, your immune system will be strengthened. https://nervogenpro-web.com/
VivoTonic™ is a 11-in-1 vital blood sugar support formula that may improve how the metabolism goes after the calories that consumers eat. https://vivotonic-web.com/
Progenifix is designed to help maximize weight loss results using a mixture of natural, science-backed ingredients. The formula also has secondary benefits, including promoting overall wellness and vitality and assisting your immune system. https://progenifix-web.com/
https://doxycyclinea.online/# buy doxycycline without prescription uk
zithromax 500mg price in india: zithromax 1000 mg online – zithromax for sale cheap
AquaPeace is an all-natural nutritional formula that uses a proprietary and potent blend of ingredients and nutrients to improve overall ear and hearing health and alleviate the symptoms of tinnitus. https://aquapeace-web.com
FoliPrime is a simple serum containing a blend of vitamins designed to boost hair health. FoliPrime has 100 percent natural substances that enhance and supplement the vitamins in the scalp to promote hair growth. https://foliprime-web.com/
buy doxycycline for dogs doxycycline 100mg buy generic doxycycline
Gut Vita™ is a daily supplement that helps consumers to improve the balance in their gut microbiome, which supports the health of their immune system. It supports healthy digestion, even for consumers who have maintained an unhealthy diet for a long time. https://gutvita-us.com/
canada where to buy neurontin neurontin 50mg cost neurontin 200 mg capsules
Neuro-Thrive is a brain health supplement that claims to promote good memory and thinking skills and better quality sleep. This nootropic supplement achieves its cause with its potent blend of natural compounds and extracts that are proven to be effective in sharpening mental acuity. https://neurothrive-web.com/
Fast Lean Pro is a herbal supplement that tricks your brain into imagining that you’re fasting and helps you maintain a healthy weight no matter when or what you eat. It offers a novel approach to reducing fat accumulation and promoting long-term weight management. https://fastleanpro-web.com/
https://doxycyclinea.online/# doxycycline prices
The ProNail Complex is a meticulously-crafted natural formula which combines extremely potent oils and skin-supporting vitamins. https://pronailcomplex-web.com/https://pronailcomplex-web.com/
Erectin is a clinically-proven dietary supplement designed to enhance male https://erectin-web.com/
buy prednisone online without a prescription: prednisone without rx – prednisone 5084
100% Natural Formula Expressly Designed to Help Control Blood Sugar Levels, Improve Insulin Response And Support Overall Health https://glucotrusttry.com/
PowerBite stands as an innovative dental candy, dedicated to nurturing healthy teeth and gums. Infused with a potent formula, it champions the cause of a robust and radiant smile. Crafted meticulously https://powerbite-web.com/
buy amoxicillin online with paypal: amoxicillin 500mg capsule buy online – generic amoxil 500 mg
prednisone 1 tablet: where can i get prednisone over the counter – pharmacy cost of prednisone
Boostaro is a dietary supplement designed specifically for men who suffer from health issues. https://boostaro-try.com/
http://amoxila.pro/# where to buy amoxicillin pharmacy
how much is prednisone 10mg: prednisone 2.5 tablet – prednisone price south africa
Unlock the incredible potential of Puravive! Supercharge your metabolism and incinerate calories like never before with our unique fusion of 8 exotic components. Bid farewell to those stubborn pounds and welcome a reinvigorated metabolism and boundless vitality. Grab your bottle today and seize this golden opportunity! https://puravive-web.com/
Cerebrozen is an excellent liquid ear health supplement purported to relieve tinnitus and improve mental sharpness, among other benefits. The Cerebrozen supplement is made from a combination of natural ingredients, and customers say they have seen results in their hearing, focus, and memory after taking one or two droppers of the liquid solution daily for a week. https://cerebrozen-try.com/
prednisone without prescription medication prednisone rx coupon prednisone rx coupon
zithromax cost australia zithromax 500 mg lowest price drugstore online buy zithromax online fast shipping
https://amoxila.pro/# amoxicillin online canada
amoxicillin 500: amoxicillin 500mg price – amoxicillin over the counter in canada
prednisone purchase canada: prednisone 10mg tablet price – otc prednisone cream
buy cheap doxycycline: doxycycline 500mg – buy doxycycline cheap
http://zithromaxa.store/# zithromax online
doxycycline generic: doxycycline 100mg capsules – purchase doxycycline online
The human body can continue to live thanks to the correct functioning of certain systems. If even one of these systems does not work properly, it can cause problems in human life. https://calmlean-web.com/
neurontin 300 mg cost neurontin 400mg neurontin prescription online
doxycycline without prescription buy cheap doxycycline where to get doxycycline
GlucoBerry is one of the biggest all-natural dietary and biggest scientific breakthrough formulas ever in the health industry today. This is all because of its amazing high-quality cutting-edge formula that helps treat high blood sugar levels very naturally and effectively. https://glucoberry-web.com/
https://doxycyclinea.online/# generic doxycycline
VidaCalm is an all-natural blend of herbs and plant extracts that treat tinnitus and help you live a peaceful life. https://vidacalm-web.com/
Are you tired of looking in the mirror and noticing saggy skin? Is saggy skin making you feel like you are trapped in a losing battle against aging? Do you still long for the days when your complexion radiated youth and confidence? https://refirmance-web.com/
generic doxycycline: buy cheap doxycycline – buy doxycycline online
Gorilla Flow prostate is an all-natural dietary supplement for men which aims to decrease inflammation in the prostate to decrease common urinary tract issues such as frequent and night-time urination, leakage, or blocked urine stream. https://gorillaflow-web.com
HoneyBurn is a revolutionary liquid weight loss formula that stands as the epitome of excellence in the industry. https://honeyburn-web.com/
can you buy zithromax over the counter in australia: zithromax online – zithromax canadian pharmacy
Keravita Pro™ is a dietary supplement created by Benjamin Jones that effectively addresses nail fungus and hair loss, promoting the growth of healthier and thicker nails and hair. The formula is designed to target the underlying causes of these health issues and provide comprehensive treatment. https://keravitapro-web.com
https://doxycyclinea.online/# doxycycline without a prescription
medicine amoxicillin 500mg amoxicillin online purchase buy amoxicillin 500mg capsules uk
Volca Burn is a weight loss supplement that uses a “red tingle hack” to help you rapidly lose weight without dieting or exercising. https://volcaburn-web.com/
Xitox’s foot pads contain a combination of powerful herbs that help provide a soothing experience for your feet after a long day. https://xitox-web.com/
Carbofix is the revolutionary dietary formula that promises to activate weight loss without all the extra hard work. https://carbofix-try.com
Reliver Pro is a dietary supplement formulated with a blend of natural ingredients aimed at supporting liver health
http://zithromaxa.store/# zithromax 500
drug neurontin 20 mg: order neurontin – neurontin sale
amoxicillin capsule 500mg price: buy amoxicillin online with paypal – generic amoxicillin over the counter
how to buy amoxicillin online: amoxicillin 500 mg without a prescription – amoxicillin 250 mg capsule
amoxicillin medicine: amoxicillin brand name – amoxicillin over counter
http://zithromaxa.store/# how to get zithromax over the counter
doxycycline generic doxycycline price of doxycycline
amoxicillin 500 amoxicillin 500 mg online amoxicillin 500mg capsule buy online
doxycycline hyclate 100 mg cap: where can i get doxycycline – buy doxycycline online
http://doxycyclinea.online/# doxycycline medication
can i buy amoxicillin over the counter in australia: order amoxicillin online uk – amoxicillin 500mg capsules price
amoxicillin discount coupon: amoxicillin 500 coupon – amoxicillin tablet 500mg
generic zithromax 500mg india: generic zithromax india – zithromax generic price
https://zithromaxa.store/# zithromax 250 mg pill
neurontin 400 mg capsule neurontin cost australia gabapentin 600 mg
doxycycline without prescription doxycycline 500mg where can i get doxycycline
https://doxycyclinea.online/# buy generic doxycycline
zithromax cost uk: how much is zithromax 250 mg – buy zithromax 1000mg online
At the heart of Dune’s plot lies the invaluable spice, melange, which can only be found in the vast sand dunes of Arrakis. As Paul Atreides, heir to House Atreides, players are tasked with managing spice extraction, navigating complex political webs, and leading a revolt against the sinister House Harkonnen. The game beautifully unfolds this rich narrative through a combination of detailed character interactions and strategic gameplay elements. Lastly, if you really want to go deep, then there is one more piece of the Dune oeuvre that you really ought to sample. Dune 1984 was not the first time that a film adaptation of the Frank Herbet novel was attempted. In the mid-70s, cult film director Alejandro Jodorowsky also had a go. It didn’t turn out so well.
https://gunnerqjud036400.dailyhitblog.com/31681082/car-city-driving-download-pc
Play popular board games online with up to 5 friends on other devices for free. No sign-in or download required. It was originally published in January 2011 and as of March 2013 its code is open source and licensed under the GPL Version 3 license. Gomoku is a turn-based two-player game played on a 15×15 board with black and white pieces. The game is also called “Renju” or “Five in a row”. To move a chess piece, select it, and then select where you want the piece to go. Two Player Chess contains designs and concepts that are protected under law.© 2020 – Adrian McPhee Chess Online – 2 Player Games Chess Online – 2 Player Games Gomoku is a turn-based two-player game played on a 15×15 board with black and white pieces. The game is also called “Renju” or “Five in a row”.
where to purchase doxycycline: doxycycline hyc – doxycycline hyclate 100 mg cap
zithromax antibiotic: zithromax online paypal – generic zithromax 500mg
ProstaBiome is a carefully crafted dietary supplement aimed at promoting prostate health. Bid farewell to restless nights and discomfort with ProstaBiome precise strategy for addressing prostate concerns. https://prostabiome-web.com/
PotentStream is designed to address prostate health by targeting the toxic, hard water minerals that can create a dangerous buildup inside your urinary system It’s the only dropper that contains nine powerful natural ingredients that work in perfect synergy to keep your prostate healthy and mineral-free well into old age. https://potentstream-web.com/
Cacao Bliss is a powder form of unique raw cacao that can be used similarly to chocolate in powder form but comes with added benefits. It is designed to provide a rich and satisfying experience while delivering numerous health benefits. https://cacaobliss-web.com/
neurontin 204: buy neurontin 300 mg – over the counter neurontin
http://doxycyclinea.online/# doxycycline 100mg online
amoxicillin online canada where can i get amoxicillin 500 mg how much is amoxicillin prescription
zithromax 500 mg for sale zithromax prescription online zithromax 250 mg tablet price
Utilitylatest provides news and analysis for energy and utility executives. We cover topics like smart grid tech, clean energy, regulation, generation, demand response, solar, storage, transmission distribution, and more. https://utilitylatest.com
zithromax generic price: buy zithromax 1000mg online – can you buy zithromax over the counter in canada
https://zithromaxa.store/# zithromax 500
neurontin cost generic: neurontin cap 300mg price – neurontin tablets 100mg
9 da auto news – https://9-da.com/
dtmliving multifamily news – https://dtmliving.com/
neurontin generic: neurontin 100mg tab – neurontin without prescription
Cneche provides in-depth journalism and insight into the most impactful news and trends shaping the finance industry. https://cneche.com/
price of neurontin: neurontin pill – canada neurontin 100mg lowest price
how to buy prednisone prednisone 20mg online without prescription prednisone generic cost
http://prednisoned.online/# buy generic prednisone online
non prescription prednisone 20mg prednisone generic cost can you buy prednisone without a prescription
http://amoxila.pro/# amoxicillin 500mg pill
zithromax 500 mg lowest price online: how to get zithromax over the counter – zithromax without prescription
Lasixiv provides news and analysis for IT executives. We cover big data, IT strategy, cloud computing, security, mobile technology, infrastructure, software and more. https://lasixiv.com
zithromax 500 mg for sale: zithromax for sale us – buy zithromax online with mastercard
buy doxycycline doxycycline without a prescription buy doxycycline online uk
amoxicillin 500 mg purchase without prescription: amoxicillin 500 mg where to buy – amoxicillin online no prescription
purchase zithromax z-pak: zithromax 500mg price – generic zithromax medicine
http://mexicanpharmacy1st.com/# best online pharmacies in mexico
buying prescription drugs in mexico: mexico drug stores pharmacies – mexico pharmacy
https://mexicanpharmacy1st.shop/# п»їbest mexican online pharmacies
Wedstraunt has the latest news in the restaurant industry, covering topics like consumer trends, technology, marketing and branding, operations, mergers https://wedstraunt.com
purple pharmacy mexico price list: buying prescription drugs in mexico – best online pharmacies in mexico
mexican pharmaceuticals online: medicine in mexico pharmacies – reputable mexican pharmacies online
mexican rx online mexico pharmacies prescription drugs buying prescription drugs in mexico
https://mexicanpharmacy1st.shop/# mexican rx online
п»їbest mexican online pharmacies mexico drug stores pharmacies medicine in mexico pharmacies
п»їbest mexican online pharmacies: purple pharmacy mexico price list – mexican border pharmacies shipping to usa
mexican online pharmacies prescription drugs: medicine in mexico pharmacies – best mexican online pharmacies
mexican pharmacy purple pharmacy mexico price list mexican online pharmacies prescription drugs
https://mexicanpharmacy1st.online/# п»їbest mexican online pharmacies
buying prescription drugs in mexico: reputable mexican pharmacies online – medicine in mexico pharmacies
Tvphc provides news and analysis for IT executives. We cover big data, IT strategy, cloud computing, security, mobile technology, infrastructure, software and more. https://tvphc.com
mexico drug stores pharmacies: medication from mexico pharmacy – mexican online pharmacies prescription drugs
https://mexicanpharmacy1st.shop/# mexico pharmacies prescription drugs
https://mexicanpharmacy1st.com/# mexico pharmacies prescription drugs
medication from mexico pharmacy: pharmacies in mexico that ship to usa – mexican mail order pharmacies
http://mexicanpharmacy1st.com/# mexican pharmaceuticals online
mexico drug stores pharmacies: buying prescription drugs in mexico – buying from online mexican pharmacy
https://mexicanpharmacy1st.com/# pharmacies in mexico that ship to usa
п»їbest mexican online pharmacies mexican pharmacy mexican online pharmacies prescription drugs
Grpduk provides news and analysis for human resource executives. We cover topics like recruiting, HR management, employee learning https://grpduk.com
mexican online pharmacies prescription drugs: mexico pharmacies prescription drugs – medication from mexico pharmacy
mexico pharmacies prescription drugs: buying prescription drugs in mexico online – medication from mexico pharmacy
Susibu provides in-depth journalism and insight into the news and trends impacting the hotel https://susibu.com/
Sisanit provides in-depth journalism and insight into the news and trends impacting corporate counsel. https://sisanit.com/
buying from online mexican pharmacy: mexican drugstore online – reputable mexican pharmacies online
https://mexicanpharmacy1st.com/# medication from mexico pharmacy
mexico drug stores pharmacies medicine in mexico pharmacies mexican pharmacy
https://mexicanpharmacy1st.online/# mexican pharmacy
buying prescription drugs in mexico: mexican pharmacy – buying prescription drugs in mexico
mexican pharmaceuticals online: best mexican online pharmacies – purple pharmacy mexico price list
https://mexicanpharmacy1st.shop/# mexican border pharmacies shipping to usa
https://mexicanpharmacy1st.com/# mexican drugstore online
Janmckinley provides news and analysis for waste and recycling executives. We cover topics like landfills, collections, regulation, waste-to-energy, corporate news, fleet management, and more. https://janmckinley.com
best online pharmacies in mexico pharmacies in mexico that ship to usa mexican online pharmacies prescription drugs
mexican drugstore online medication from mexico pharmacy mexican mail order pharmacies
https://mexicanpharmacy1st.online/# п»їbest mexican online pharmacies
http://mexicanpharmacy1st.com/# buying prescription drugs in mexico online
mexican online pharmacies prescription drugs: mexican drugstore online – mexican border pharmacies shipping to usa
https://mexicanpharmacy1st.com/# reputable mexican pharmacies online
mexican drugstore online: mexican border pharmacies shipping to usa – pharmacies in mexico that ship to usa
mexico drug stores pharmacies: pharmacies in mexico that ship to usa – mexican rx online
https://mexicanpharmacy1st.com/# buying prescription drugs in mexico online
mexican mail order pharmacies mexican pharmacy mexican rx online
Sugar Defender is the rated blood sugar formula with an advanced blend of 24 proven ingredients that support healthy glucose levels and natural weight loss. https://smithsis.com
mexican border pharmacies shipping to usa: best online pharmacies in mexico – mexican rx online
reputable mexican pharmacies online: purple pharmacy mexico price list – mexico drug stores pharmacies
mexican online pharmacies prescription drugs medicine in mexico pharmacies medication from mexico pharmacy
mexican mail order pharmacies: mexican pharmacy – mexican pharmaceuticals online
https://mexicanpharmacy1st.shop/# mexico drug stores pharmacies
Sugar Defender is a revolutionary blood sugar support formula designed to support healthy glucose levels and promote natural weight loss. https://blackboxvending.com/
https://mexicanpharmacy1st.online/# mexican drugstore online
Sugar Defender is a revolutionary blood sugar support formula designed to support healthy glucose levels and promote natural weight loss. https://mineryuta.com
medication from mexico pharmacy: mexican mail order pharmacies – medicine in mexico pharmacies
buying prescription drugs in mexico mexican border pharmacies shipping to usa mexican mail order pharmacies
medicine in mexico pharmacies mexico drug stores pharmacies medication from mexico pharmacy
mexico drug stores pharmacies: mexican mail order pharmacies – best online pharmacies in mexico
http://mexicanpharmacy1st.com/# п»їbest mexican online pharmacies
https://mexicanpharmacy1st.online/# mexican pharmaceuticals online
purple pharmacy mexico price list: mexico pharmacies prescription drugs – mexico pharmacy
mexican pharmacy best online pharmacies in mexico reputable mexican pharmacies online
https://mexicanpharmacy1st.com/# mexican rx online
https://mexicanpharmacy1st.online/# medication from mexico pharmacy
best mexican online pharmacies: mexican pharmaceuticals online – mexican drugstore online
http://mexicanpharmacy1st.com/# pharmacies in mexico that ship to usa
Sugar Defender is a revolutionary blood sugar support formula designed to support healthy glucose levels and promote natural weight loss. https://acmesignz.com/
п»їbest mexican online pharmacies: mexican pharmaceuticals online – mexico drug stores pharmacies
https://mexicanpharmacy1st.com/# mexican border pharmacies shipping to usa
http://gabapentin.club/# order neurontin online
sugar defender: https://peyfon.com/
https://cytotec.xyz/# buy cytotec
sugar defender: https://seahorsesoap.com/
cost of propecia for sale: propecia without insurance – generic propecia without insurance
cost cheap propecia: cost of propecia for sale – buying cheap propecia without insurance
buy cytotec online order cytotec online buy cytotec
get generic propecia without a prescription get generic propecia online get generic propecia prices
cost of propecia without insurance: buy propecia – order cheap propecia without prescription
http://gabapentin.club/# neurontin prices
sugar defender: https://sourceprousa.com/
buying generic clomid price: how to buy generic clomid – where can i buy generic clomid pills
neurontin 300: buying neurontin online – neurontin 300 mg buy
https://propeciaf.online/# cost of cheap propecia no prescription
cost of neurontin 800 mg neurontin 100 mg cap buy neurontin 100 mg canada
https://lisinopril.club/# lisinopril 40 mg brand name in india
Cytotec 200mcg price buy misoprostol over the counter buy cytotec over the counter
buy propecia now: cost generic propecia for sale – generic propecia tablets
Cytotec 200mcg price: cytotec pills buy online – order cytotec online
https://lisinopril.club/# lisinopril 40 mg prices
buy cytotec in usa buy misoprostol over the counter buy cytotec
sugar defender: https://lindadicesare.com/
buy cheap propecia without insurance buy propecia pill order cheap propecia without insurance
propecia generics: order generic propecia without prescription – buying cheap propecia online
sugar defender: https://drdenisemichele.com/
https://lisinopril.club/# buy lisinopril without a prescription
sugar defender: https://alchemyfashiongroup.com/
cost of lisinopril 2.5 mg: zestril over the counter – lisinopril tablets uk
https://lisinopril.club/# lisinopril 12.5 mg 10 mg
cost cheap propecia prices order cheap propecia pills buying cheap propecia without prescription
buy gabapentin where can i buy neurontin from canada neurontin rx
2 lisinopril: lipinpril – lisinopril 5 mg prices
https://propeciaf.online/# propecia price
http://clomiphene.shop/# can i buy cheap clomid for sale
http://propeciaf.online/# order generic propecia without dr prescription
buy cytotec: Cytotec 200mcg price – Cytotec 200mcg price
medicine neurontin 300 mg: neurontin pills for sale – neurontin 300 mg buy
sildenafil for pe
http://gabapentin.club/# neurontin 900
propecia without prescription cost of propecia price generic propecia prices
can i buy generic clomid pills how to buy cheap clomid online cost generic clomid pills
where to buy generic clomid for sale: where to buy generic clomid online – can you get clomid tablets
https://cytotec.xyz/# Misoprostol 200 mg buy online
lisinopril 20 mg tab price: buy lisinopril no prescription – lisinopril 60 mg tablet
can i buy cheap clomid tablets: can i order cheap clomid without rx – buy cheap clomid without dr prescription
http://cytotec.xyz/# Abortion pills online
how to get clomid how can i get cheap clomid without dr prescription clomid without insurance
where to get clomid without dr prescription order generic clomid tablets cheap clomid without rx
neurontin 100mg discount: neurontin for sale online – neurontin capsules 100mg
https://lisinopril.club/# lisinopril 30 mg
where buy clomid online: where can i buy generic clomid now – cost of clomid without a prescription
order cytotec online buy cytotec buy cytotec
neurontin 600mg neurontin gel neurontin online pharmacy
https://cytotec.xyz/# Abortion pills online
http://lisinopril.club/# lisinopril drug
best generic lisinopril: where to buy lisinopril 2.5 mg – lisinopril generic brand
lisinopril 20 mg canadian pharmacy: order lisinopril online us – lisinopril with out prescription
lisinopril prescription lisinopril 20mg daily prinivil tabs
cytotec buy online usa buy cytotec online fast delivery buy cytotec
order cytotec online: cytotec abortion pill – buy cytotec
how to get clomid pills: where buy clomid for sale – can you get generic clomid tablets
buy generic propecia pill cost of propecia cheap propecia without a prescription
https://cheapestcanada.shop/# canada online pharmacy
https://cheapestindia.com/# buy prescription drugs from india
https://cheapestcanada.com/# global pharmacy canada
international pharmacy no prescription: cheapest pharmacy – cheapest pharmacy to get prescriptions filled
http://cheapestcanada.com/# canada online pharmacy
https://cheapestmexico.com/# mexican online pharmacies prescription drugs
canadian pharmacy no prescription needed 36 and 6 health online pharmacy canadian pharmacy no prescription
prescription drugs online 36and6health canadian pharmacy no prescription needed
best online prescription: cheapest & fast pharmacy – how to get prescription drugs from canada
http://36and6health.com/# no prescription needed canadian pharmacy
http://cheapestindia.com/# best india pharmacy
https://cheapestmexico.com/# mexico pharmacies prescription drugs
http://36and6health.com/# canadian pharmacy no prescription needed
http://cheapestandfast.com/# no prescription canadian pharmacies
http://36and6health.com/# promo code for canadian pharmacy meds
buy medicines online in india india pharmacy mail order indian pharmacy online
best no prescription online pharmacies cheapest and fast best online pharmacies without prescription
https://cheapestmexico.com/# buying prescription drugs in mexico online
http://cheapestindia.com/# reputable indian online pharmacy
canadian pharmacy prices: prescription drugs canada buy online – vipps approved canadian online pharmacy
https://cheapestandfast.com/# buying drugs online no prescription
https://cheapestmexico.com/# mexico drug stores pharmacies
https://36and6health.com/# canadian pharmacy no prescription
mexico pharmacies prescription drugs: mexican pharmaceuticals online – mexican border pharmacies shipping to usa
http://36and6health.com/# mail order pharmacy no prescription
http://cheapestcanada.com/# medication canadian pharmacy
https://cheapestindia.shop/# world pharmacy india
http://cheapestcanada.com/# canadian drugstore online
https://36and6health.com/# best no prescription pharmacy
indian pharmacy paypal: indian pharmacy online – indian pharmacy paypal
https://cheapestandfast.shop/# canada prescription drugs online
canadian online pharmacy cheapest canada canadian pharmacy mall
world pharmacy india world pharmacy india india pharmacy mail order
https://36and6health.shop/# drugstore com online pharmacy prescription drugs
indianpharmacy com: top 10 online pharmacy in india – indian pharmacy paypal
medikamente rezeptfrei: shop apotheke gutschein – internet apotheke
https://eufarmaciaonline.com/# farmacias online seguras
Farmacia online più conveniente: comprare farmaci online all’estero – farmacia online più conveniente
farmacias online baratas farmacia online madrid farmacias direct
https://eumedicamentenligne.shop/# pharmacie en ligne fiable
farmacia online: farmaci senza ricetta elenco – comprare farmaci online all’estero
farmacia online barata farmacias online seguras en espaГ±a farmacia online madrid
pharmacie en ligne fiable: trouver un mГ©dicament en pharmacie – Pharmacie Internationale en ligne
farmacia online madrid: farmacia online madrid – farmacias online seguras
farmacia online piГ№ conveniente: migliori farmacie online 2024 – top farmacia online
medikamente rezeptfrei: online apotheke deutschland – beste online-apotheke ohne rezept
online apotheke deutschland online apotheke internet apotheke
farmacia online envГo gratis: farmacia barata – farmacia online barcelona
acheter mГ©dicament en ligne sans ordonnance trouver un mГ©dicament en pharmacie vente de mГ©dicament en ligne
vente de mГ©dicament en ligne: Pharmacie en ligne livraison Europe – pharmacie en ligne france livraison internationale
farmacia online madrid: farmacia online envÃo gratis – farmacia online 24 horas
Pharmacie Internationale en ligne: pharmacie en ligne fiable – pharmacie en ligne france livraison belgique
farmacia online españa envÃo internacional: farmacias online seguras en españa – farmacia online españa envÃo internacional
online apotheke preisvergleich ohne rezept apotheke gГјnstigste online apotheke
https://eumedicamentenligne.shop/# Pharmacie Internationale en ligne
online apotheke versandkostenfrei: online apotheke versandkostenfrei – internet apotheke
acheter mГ©dicament en ligne sans ordonnance: Pharmacie sans ordonnance – pharmacie en ligne france livraison internationale
comprare farmaci online con ricetta acquisto farmaci con ricetta farmacie online affidabili
Pharmacie sans ordonnance: pharmacie en ligne france pas cher – pharmacie en ligne france fiable
farmacias direct: farmacias online seguras en espaГ±a – farmacias direct
https://euapothekeohnerezept.com/# online apotheke versandkostenfrei
Pharmacie en ligne livraison Europe: vente de m̩dicament en ligne Рacheter m̩dicament en ligne sans ordonnance
Farmacia online miglior prezzo farmacie online sicure farmaci senza ricetta elenco
Farmacia online miglior prezzo: farmacia online piГ№ conveniente – Farmacia online piГ№ conveniente
online apotheke gГјnstig internet apotheke beste online-apotheke ohne rezept
comprare farmaci online all’estero: Farmacia online miglior prezzo – farmaci senza ricetta elenco
farmacie online sicure: Farmacia online miglior prezzo – farmacia online
farmacia online piГ№ conveniente: top farmacia online – comprare farmaci online all’estero
eu apotheke ohne rezept: online apotheke günstig – medikament ohne rezept notfall
farmacias direct: farmacias online seguras – farmacias direct
farmaci senza ricetta elenco farmacia online senza ricetta comprare farmaci online con ricetta
online apotheke rezept: online apotheke gГјnstig – online apotheke versandkostenfrei
pharmacie en ligne pharmacie en ligne france livraison belgique п»їpharmacie en ligne france
online apotheke rezept: günstigste online apotheke – apotheke online
farmacia online barata: farmacia online 24 horas – п»їfarmacia online espaГ±a
beste online-apotheke ohne rezept: shop apotheke gutschein – beste online-apotheke ohne rezept
http://eumedicamentenligne.com/# pharmacie en ligne avec ordonnance
farmacias online seguras en espaГ±a: farmacia online envГo gratis – farmacia online espaГ±a envГo internacional
Farmacia online miglior prezzo comprare farmaci online all’estero farmaci senza ricetta elenco
https://eufarmacieonline.com/# farmacie online autorizzate elenco
farmacia online barata farmacia online 24 horas farmacia barata
farmacia online 24 horas: farmacias online seguras – farmacia online envÃo gratis
europa apotheke: medikamente rezeptfrei – gГјnstige online apotheke
Farmacie online sicure: acquisto farmaci con ricetta – farmacie online affidabili
farmacia online 24 horas: п»їfarmacia online espaГ±a – farmacia online espaГ±a envГo internacional
online apotheke versandkostenfrei: internet apotheke – europa apotheke
farmacia online envГo gratis: farmacias online baratas – farmacias online seguras
gГјnstige online apotheke: ohne rezept apotheke – medikamente rezeptfrei
farmacia online envГo gratis farmacias online seguras farmacia online barata
pharmacies en ligne certifiées: pharmacie en ligne – pharmacie en ligne france
farmacia online barcelona farmacia online barata farmacia en casa online descuento
online apotheke: europa apotheke – ohne rezept apotheke
farmacie online autorizzate elenco: top farmacia online – Farmacia online piГ№ conveniente
farmacias online seguras: farmacia online españa envÃo internacional – farmacia barata
pharmacie en ligne livraison europe pharmacie en ligne france livraison internationale Pharmacie en ligne livraison Europe
farmacia online barata y fiable: farmacia online madrid – farmacia online españa
https://eufarmacieonline.com/# migliori farmacie online 2024
europa apotheke eu apotheke ohne rezept online apotheke
eu apotheke ohne rezept: eu apotheke ohne rezept – internet apotheke
pharmacie en ligne: pharmacie en ligne pas cher – Pharmacie sans ordonnance
online apotheke rezept: ohne rezept apotheke – günstige online apotheke
http://eufarmaciaonline.com/# farmacia en casa online descuento
online apotheke medikamente rezeptfrei online apotheke
farmacia online barata y fiable: farmacia online barata y fiable – п»їfarmacia online espaГ±a
Achat m̩dicament en ligne fiable: pharmacie en ligne fiable Рvente de m̩dicament en ligne
gГјnstige online apotheke: gГјnstigste online apotheke – online apotheke deutschland
Farmacia online piГ№ conveniente: farmacia online – farmacia online
farmacias online seguras en espaГ±a: farmacia online madrid – farmacia online envГo gratis
farmacias online seguras: farmacia online barata y fiable – п»їfarmacia online espaГ±a
farmacia online envГo gratis farmacias online seguras en espaГ±a farmacias online seguras
farmacie online sicure: acquisto farmaci con ricetta – farmacia online senza ricetta
http://levitraenligne.com/# pharmacie en ligne avec ordonnance
trouver un mГ©dicament en pharmacie: Levitra acheter – Achat mГ©dicament en ligne fiable
Viagra homme sans ordonnance belgique Acheter du Viagra sans ordonnance Viagra pas cher inde
pharmacie en ligne france livraison belgique: Acheter Cialis 20 mg pas cher – pharmacie en ligne france pas cher
Viagra 100 mg sans ordonnance viagra sans ordonnance Viagra vente libre pays
trouver un mГ©dicament en pharmacie: pharmacies en ligne certifiГ©es – pharmacie en ligne pas cher
pharmacie en ligne france livraison belgique: kamagra 100mg prix – Pharmacie sans ordonnance
https://kamagraenligne.com/# pharmacie en ligne france livraison belgique
pharmacie en ligne france livraison belgique: kamagra 100mg prix – pharmacie en ligne avec ordonnance
pharmacie en ligne pas cher: Levitra 20mg prix en pharmacie – pharmacie en ligne
Pharmacie en ligne livraison Europe: achat kamagra – pharmacie en ligne france livraison internationale
pharmacie en ligne fiable: kamagra en ligne – п»їpharmacie en ligne france
pharmacie en ligne sans ordonnance: pharmacie en ligne sans ordonnance – trouver un mГ©dicament en pharmacie
Achat mГ©dicament en ligne fiable: pharmacie en ligne pas cher – pharmacie en ligne france pas cher
pharmacie en ligne livraison europe: kamagra oral jelly – pharmacie en ligne pas cher
vente de mГ©dicament en ligne kamagra en ligne Achat mГ©dicament en ligne fiable
Hello!
This post was created with XRumer 23 StrongAI.
Good luck 🙂
pharmacie en ligne france fiable: pharmacie en ligne sans ordonnance – pharmacie en ligne avec ordonnance
Pharmacie Internationale en ligne: Pharmacie Internationale en ligne – Pharmacie sans ordonnance
http://phenligne.com/# pharmacie en ligne
pharmacie en ligne avec ordonnance: kamagra pas cher – pharmacie en ligne pas cher
vente de mГ©dicament en ligne: Pharmacies en ligne certifiees – Pharmacie sans ordonnance
Pharmacie sans ordonnance: acheter kamagra site fiable – pharmacie en ligne france pas cher
pharmacie en ligne: pharmacie en ligne sans ordonnance – Achat mГ©dicament en ligne fiable
Hello.
This post was created with XRumer 23 StrongAI.
Good luck 🙂
Quand une femme prend du Viagra homme: viagra en ligne – Viagra homme sans prescription
trouver un mГ©dicament en pharmacie: levitra generique – pharmacie en ligne fiable
vente de mГ©dicament en ligne: pharmacie en ligne – Pharmacie Internationale en ligne
Viagra vente libre pays: Viagra generique en pharmacie – Viagra 100 mg sans ordonnance
Viagra pas cher paris: Viagra generique en pharmacie – SildГ©nafil 100 mg prix en pharmacie en France
acheter mГ©dicament en ligne sans ordonnance: pharmacie en ligne sans ordonnance – pharmacie en ligne avec ordonnance
Pharmacie Internationale en ligne: Levitra pharmacie en ligne – pharmacie en ligne france livraison internationale
https://kamagraenligne.shop/# Achat médicament en ligne fiable
pharmacie en ligne sans ordonnance: pharmacie en ligne pas cher – pharmacie en ligne france fiable
Viagra homme prix en pharmacie: Acheter du Viagra sans ordonnance – SildГ©nafil 100mg pharmacie en ligne
pharmacie en ligne france livraison internationale: cialis prix – п»їpharmacie en ligne france
pharmacie en ligne livraison europe: kamagra 100mg prix – pharmacie en ligne france pas cher
When you’ve played as much time or as many hands as you expected to, it’s time to leave the poker table! You should never walk away from a poker table because of your performance; you have no control over your poker outcomes from one session to the next. Write a product review Homura Kawamoto, Hikaru Muno, and TMS Entertainment’s High Card is one of the best original anime series that happened in 2023. With just four episodes out, the series garnered a lot of appreciation from anime and non-anime enthusiasts with its unique take on the poker-theme genre by turning playing cards into catastrophic weapons. Set a time limit for a poker session and follow it every time you sit on the table. Select out the best poker format for you and stay in the game as long as you find it profitable. Never chase losses and set a stop loss limit as per your affordability. Disciplined game-play saves you from losing excessive money on real stake tables.
https://phase2directory.com/listings12717644/grand-eagle-casino-no-deposit-in-india
Play the largest gaming floor in Southwest Louisiana. We have lots of wide-open gaming space – so come play your favorite slots and table games! Stay up to date with what is happening at Colette’s Children’s Home and stay involved to help heal homelessness. Join our mailing list to receive our quarterly newsletter and other updates! Play at a trusted casino supported by Sunny Leone Slot machines work by using a complex piece of software, known as a random number generator (RNG). This determines the result of every spin. The volatility of a slot machine game measures the risk involved in playing a particular slot for real money. One of my favorite tips for playing slots is to consider it the ‘risk factor’ of the game you are about to play. That’s because volatility determines how you win at slots.
pharmacie en ligne france pas cher: pharmacies en ligne certifiГ©es – pharmacie en ligne france livraison belgique
Achat mГ©dicament en ligne fiable: Cialis sans ordonnance 24h – pharmacie en ligne france pas cher
Pharmacie Internationale en ligne: levitra generique – pharmacie en ligne livraison europe
pharmacie en ligne fiable: pharmacie en ligne pas cher – pharmacie en ligne livraison europe
pharmacie en ligne: pharmacie en ligne pas cher – acheter mГ©dicament en ligne sans ordonnance
pharmacie en ligne: Levitra sans ordonnance 24h – pharmacie en ligne livraison europe
Meilleur Viagra sans ordonnance 24h: viagra sans ordonnance – Viagra sans ordonnance 24h Amazon
SildГ©nafil Teva 100 mg acheter: viagra sans ordonnance – SildГ©nafil Teva 100 mg acheter
pharmacie en ligne fiable: pharmacie en ligne – pharmacie en ligne france livraison internationale
pharmacie en ligne fiable: kamagra pas cher – acheter mГ©dicament en ligne sans ordonnance
pharmacie en ligne fiable: cialis sans ordonnance – vente de mГ©dicament en ligne
Viagra prix pharmacie paris: Viagra gГ©nГ©rique pas cher livraison rapide – Viagra sans ordonnance 24h Amazon
pharmacie en ligne france livraison internationale: pharmacie en ligne fiable – Pharmacie en ligne livraison Europe
Achat mГ©dicament en ligne fiable: pharmacie en ligne – Pharmacie Internationale en ligne
Viagra homme prix en pharmacie sans ordonnance: Viagra sans ordonnance 24h – Meilleur Viagra sans ordonnance 24h
pharmacie en ligne france livraison belgique: cialis prix – Pharmacie sans ordonnance
Viagra gГ©nГ©rique sans ordonnance en pharmacie: viagra sans ordonnance – SildГ©nafil Teva 100 mg acheter
Pharmacie en ligne livraison Europe: kamagra gel – pharmacie en ligne france pas cher
pharmacie en ligne avec ordonnance: п»їpharmacie en ligne france – Pharmacie sans ordonnance
pharmacie en ligne sans ordonnance: kamagra 100mg prix – pharmacie en ligne avec ordonnance
Pharmacie sans ordonnance: kamagra en ligne – Pharmacie sans ordonnance
pharmacie en ligne france livraison internationale: cialis prix – pharmacie en ligne avec ordonnance
pharmacie en ligne france livraison internationale: pharmacie en ligne sans ordonnance – acheter mГ©dicament en ligne sans ordonnance
Pharmacie sans ordonnance: pharmacie en ligne pas cher – trouver un mГ©dicament en pharmacie
pharmacie en ligne france fiable: levitra en ligne – pharmacie en ligne livraison europe
pharmacie en ligne avec ordonnance: kamagra livraison 24h – pharmacie en ligne france pas cher
pharmacie en ligne fiable: Levitra pharmacie en ligne – pharmacie en ligne sans ordonnance
pharmacie en ligne france fiable: kamagra gel – Pharmacie sans ordonnance
pharmacie en ligne sans ordonnance: achat kamagra – pharmacie en ligne
pharmacie en ligne france livraison internationale: kamagra livraison 24h – pharmacie en ligne
trouver un mГ©dicament en pharmacie: kamagra livraison 24h – pharmacies en ligne certifiГ©es
pharmacie en ligne france livraison belgique: kamagra gel – Pharmacie Internationale en ligne
acheter mГ©dicament en ligne sans ordonnance: pharmacie en ligne pas cher – vente de mГ©dicament en ligne
acheter mГ©dicament en ligne sans ordonnance: cialis generique – pharmacie en ligne
pharmacie en ligne france pas cher: Medicaments en ligne livres en 24h – pharmacie en ligne france pas cher
pharmacie en ligne france fiable: Acheter Cialis 20 mg pas cher – pharmacies en ligne certifiГ©es
п»їpharmacie en ligne france: pharmacie en ligne france pas cher – Pharmacie en ligne livraison Europe
Achat mГ©dicament en ligne fiable: kamagra 100mg prix – pharmacie en ligne avec ordonnance
acheter mГ©dicament en ligne sans ordonnance: Acheter Cialis – pharmacie en ligne france fiable
Viagra homme prix en pharmacie sans ordonnance: Acheter du Viagra sans ordonnance – п»їViagra sans ordonnance 24h
pharmacie en ligne livraison europe: cialis prix – pharmacie en ligne france livraison internationale
Viagra homme prix en pharmacie: viagra sans ordonnance – Viagra pas cher livraison rapide france
п»їpharmacie en ligne france: cialis sans ordonnance – pharmacie en ligne sans ordonnance
pharmacie en ligne: kamagra en ligne – pharmacie en ligne avec ordonnance
vente de mГ©dicament en ligne: Levitra pharmacie en ligne – pharmacie en ligne france fiable
Achat mГ©dicament en ligne fiable: Levitra acheter – pharmacie en ligne france pas cher
trouver un mГ©dicament en pharmacie: levitra generique prix en pharmacie – acheter mГ©dicament en ligne sans ordonnance
pharmacies en ligne certifiГ©es: Cialis sans ordonnance 24h – vente de mГ©dicament en ligne
Viagra en france livraison rapide: viagra en ligne – Viagra vente libre pays
pharmacies en ligne certifiГ©es: п»їpharmacie en ligne france – Pharmacie sans ordonnance
Viagra sans ordonnance 24h Amazon: Acheter viagra en ligne livraison 24h – Viagra gГ©nГ©rique pas cher livraison rapide
pharmacie en ligne fiable: cialis prix – pharmacies en ligne certifiГ©es
pharmacie en ligne france fiable: kamagra gel – pharmacie en ligne
pharmacie en ligne sans ordonnance: Medicaments en ligne livres en 24h – pharmacie en ligne
pharmacie en ligne france fiable: Levitra pharmacie en ligne – Achat mГ©dicament en ligne fiable
https://phenligne.com/# pharmacie en ligne france pas cher
Pharmacie en ligne livraison Europe: pharmacie en ligne livraison europe – vente de mГ©dicament en ligne
Viagra sans ordonnance 24h suisse: viagra en ligne – Viagra pas cher paris
pharmacie en ligne: pharmacie en ligne pas cher – pharmacie en ligne france pas cher
Pharmacie en ligne livraison Europe: pharmacie en ligne sans ordonnance – pharmacie en ligne france fiable
Achat mГ©dicament en ligne fiable: Levitra pharmacie en ligne – Pharmacie en ligne livraison Europe
Achat mГ©dicament en ligne fiable: cialis prix – pharmacie en ligne pas cher
pharmacie en ligne france livraison internationale: Pharmacies en ligne certifiees – pharmacie en ligne france livraison internationale
pharmacie en ligne fiable: acheter kamagra site fiable – trouver un mГ©dicament en pharmacie
pharmacie en ligne livraison europe: Levitra 20mg prix en pharmacie – Pharmacie Internationale en ligne
Acheter Sildenafil 100mg sans ordonnance: Acheter du Viagra sans ordonnance – Viagra 100mg prix
Viagra femme sans ordonnance 24h: Viagra generique en pharmacie – Prix du Viagra 100mg en France
pharmacies en ligne certifiГ©es: levitra generique prix en pharmacie – pharmacies en ligne certifiГ©es
Viagra pas cher inde: Viagra pas cher livraison rapide france – Meilleur Viagra sans ordonnance 24h
pharmacie en ligne france livraison internationale: Levitra 20mg prix en pharmacie – pharmacie en ligne fiable
vente de mГ©dicament en ligne: kamagra livraison 24h – pharmacie en ligne livraison europe
vente de mГ©dicament en ligne: pharmacie en ligne – pharmacie en ligne fiable
Meilleur Viagra sans ordonnance 24h: Acheter du Viagra sans ordonnance – SildГ©nafil 100mg pharmacie en ligne
Achat mГ©dicament en ligne fiable: cialis sans ordonnance – acheter mГ©dicament en ligne sans ordonnance
Achat mГ©dicament en ligne fiable: cialis generique – pharmacie en ligne fiable
https://autolux-azerbaijan.com/# Pin-Up Casino
https://autolux-azerbaijan.com/# pin-up kazino
pin-up360: Pin Up Kazino ?Onlayn – pin-up 141 casino
https://autolux-azerbaijan.com/# Pin-Up Casino
https://autolux-azerbaijan.com/# Pin Up Azerbaycan
Pin-Up Casino: Pin Up – pin-up 141 casino
Pin Up Azerbaycan: pin-up kazino – pin-up kazino
https://autolux-azerbaijan.com/# Pin Up Azerbaycan
pin-up360: pin-up 141 casino – Pin up 306 casino
https://autolux-azerbaijan.com/# ?Onlayn Kazino
pin-up kazino: ?Onlayn Kazino – pin-up360
There are no previous studies on its effects in human inflammatory breast cancer or canine inflammatory mammary cancer that is the most aggressive type of breast cancer cialis generic best price
hemen sesi yaziya cevirin ve dinlemekle ugrasmayin
hemen sesi yaziya cevirin ve dinlemekle ugrasmayin
mexico drug stores pharmacies
https://cmqpharma.com/# pharmacies in mexico that ship to usa
mexican rx online
purple pharmacy mexico price list: cmq pharma – mexico pharmacy
http://foruspharma.com/# п»їbest mexican online pharmacies
canadian drug pharmacy: reputable canadian pharmacy – safe canadian pharmacy
best canadian pharmacy online: canadian discount pharmacy – my canadian pharmacy rx
mexico drug stores pharmacies mexican drugstore online mexico drug stores pharmacies
best mail order pharmacy canada: canada rx pharmacy – canadian pharmacy ltd
is canadian pharmacy legit: trusted canadian pharmacy – certified canadian pharmacy
https://indiapharmast.com/# indian pharmacy paypal
my canadian pharmacy reviews canada cloud pharmacy canada drug pharmacy
mexico drug stores pharmacies: buying prescription drugs in mexico – mexico drug stores pharmacies
cheapest online pharmacy india: indian pharmacy paypal – best india pharmacy
mexico pharmacies prescription drugs: mexican border pharmacies shipping to usa – mexico drug stores pharmacies
https://canadapharmast.online/# legal to buy prescription drugs from canada
canadian pharmacy ltd safe canadian pharmacy best canadian pharmacy online
india pharmacy: indian pharmacy online – top online pharmacy india
canada drugstore pharmacy rx: buy prescription drugs from canada cheap – cheap canadian pharmacy online
medicine in mexico pharmacies: purple pharmacy mexico price list – mexico pharmacy
how to buy amoxycillin: amoxicillin cost australia – amoxicillin over counter
https://doxycyclinedelivery.pro/# buy doxycycline cheap
how to get clomid for sale: where to buy cheap clomid without insurance – can i get cheap clomid now
http://doxycyclinedelivery.pro/# doxycycline online canada without prescription
https://amoxildelivery.pro/# over the counter amoxicillin canada
buy cipro no rx: cipro online no prescription in the usa – antibiotics cipro
http://clomiddelivery.pro/# can you get cheap clomid without rx
doxycycline no prescription: generic for doxycycline – purchase doxycycline
http://amoxildelivery.pro/# amoxicillin 500mg for sale uk
http://ciprodelivery.pro/# antibiotics cipro
doxycycline 300 mg tablet: doxycycline prices canada – can you buy doxycycline over the counter uk
http://ciprodelivery.pro/# cipro
https://doxycyclinedelivery.pro/# can you buy doxycycline over the counter
where can you buy amoxicillin over the counter: amoxicillin price canada – amoxicillin no prescipion
cost of clomid online: where can i buy cheap clomid without insurance – where to get cheap clomid for sale
amoxicillin 500 mg cost: purchase amoxicillin 500 mg – over the counter amoxicillin canada
can you buy doxycycline over the counter in mexico: doxycycline 125 mg – buy doxycycline online usa