ORIGINAL ARTICLE
Year: 2019 I Volume: 2 I Issue: 3 I Page: 78-83
Platelet Rich Plasma, 5% Minoxidil Lotion and Oral Dutasteride Versus 5% Minoxidil Lotion and Oral Dutasteride in Male Androgenetic Alopecia: A Pilot Study in Routine Clinical Setting
GK Singh1, Vikas Pathania2, NS Beniwal3, S Baveja4, Dr Prerna Shankar5
1 Associate Professor, Classified Specialist (Dermatology, Venereology and Leprosy), Affiliated Faculty, Army College of Medical Sciences, New Delhi 110010
2Associate Professor, Classified Specialist (Dermatology, Venereology and Leprosy), Affiliated Faculty, Armed Forces Medical College Command Hospital, Pune 411040
3 Assistant Professor, Classified Specialist (Dermatology, Venereology and Leprosy) Affiliated Faculty, Armed Forces Medical College, Military Hospital, Kirkee; Pune 411020
4 Professor and HOD, Senior Advisor (Dermatology, Venereology and Leprosy), Armed Forces Medical College & Command Hospital, Pune 411040
5 Assistant Professor, Classified Specialist (Community Medicine), Affiliated Faculty, Armed Forces Medical College, Station Health Organization, Pune 411040
Corresponding Author:
Dr Vikas Pathania, MD
Associate Professor, Classified Specialist (Dermatology, Venereology and Leprosy)
Affiliated Faculty, Armed Forces Medical College Command Hospital; Pune 411040
E-mail address: vikascongo@gmail.com
How to cite this article:
Singh GK, Pathania V, Beniwal NS, Baveja S, Shankar P. Platelet Rich Plasma, 5% Minoxidil Lotion and Oral Dutasteride Versus 5% Minoxidil Lotion and Oral Dutasteride in Male Androgenetic Alopecia: A Pilot Study in Routine Clinical Setting. JDAIndian Journal of Clinical Dermatology. 2019;2:78-83.
Abstract:
Background: Androgenetic alopecia (AGA) or patterned baldness is a genetically determined, age related hair disorder with considerable psychosocial morbidity. Topical minoxidil and oral finasteride/ dutasteride are standard drugs for management of AGA. Platelet rich plasma (PRP), is an emerging adjunctive therapy for AGA.
Aims: To evaluate the real-world efficacy and safety of PRP when combined with 5% minoxidil lotion and oral dutasteride when compared with 5% minoxidil and oral dutasteride alone.
Settings and Design: Prospective, open label, controlled pilot study
Materials and methods:: 300 consecutive male patients with AGA were distributed into two equal groups. Group A (study group) received topical minoxidil 5% lotion once daily, dutasteride 0.5mg twice a week, PRP 0.1ml/cm2 intradermally, monthly for six months. While Group B (control group) received minoxidil and dutasteride as in Group A received normal saline instead of PRP. All patients were evaluated upto one year for improvement in hair loss with hair pull test, dermoscopic photomicrographs, macroscopic photographs and a patient satisfaction scale.
Statistical Analysis used: Statistical analysis was performed using SPSS software (version 22.0). Description of quantitative variables with mean and standard deviation. Paired t test for analysing metric variables. Wilcoxon signed rank test for ordinal variables (patient satisfaction). Statistical significance was considered using Pearson Chi-square test ( x2 test) and p value.
Results: Total 148 patients in Group A and 147 patients in Group B had completed the study. Group A showed marked reduction in hair pull test value from mean of 9.5 ±2.29 at base line to 04.25±2.03 at the end of three months while in Group B it was reduced from 09.75±2.58 to 7.75±1.42. At the end of one year, hair density increased from 34.75 ±4.90 per cm2 to 50.5±8.6 per cm2 and 35.25±5.49 per cm2 to 43.5±4.6 per cm2 in Group A and Group B respectively. At the end of six months VAS score was 7.25±1.46 in Group A and 5.75±2.02 in Group B respectively. The differences in results among both the Groups were statistically significant (P<0.05). There were no serious adverse effects observed in either groups.
Conclusions: PRP therapy along with dutasteride 0.5mg twice weekly and minoxidil 5% lotion once daily is an effective and safe modality in management of AGA when compared with minoxidil and dutasteride alone.
Key words: minoxidil 5%, dutasteride, platelet rich plasma, PRP, androgenetic alopecia, AGA.
Key messages: Management of AGA is a long term and challenging exercise with limited pharmacological options such as topical minoxidil with or without oral finasteride and dutasteride. Adjunctive use of autologous PRP with its naturally growth factors holds promise of enhancing efficacy outcome with these drugs with better patient compliance and satisfaction from the treatment.
Introduction:
Androgenetic alopecia (AGA) or patterned baldness is a genetically determined hair disorder characterized by the gradual miniaturisation of terminal hairs into vellus hairs and finally leading to bald areas on the scalp1. It affects about 50% of men over the age of 50, and about 50% of women over the age of 65 years2. Patterned baldness adversely impacts psychosocial state and self-esteem of patients3.
AGA is determined by genetics and influenced by hormones. Dihydrotestosterone (DHT), a metabolite of testosterone is a key hormone, which activates androgen receptors present on the hair follicles more densely distributed on vertex and fronto-temporal areas. In men, testosterone is converted to DHT by 5a-reductase (5-AR), which exists as 3 isoenzymes namely types I, II and III. Type I isoenzyme is universally present on the skin including the hair follicle and sebaceous glands, whereas type II is predominantly found in the male genitalia, including the prostate, and inner root sheath of hair follicles. Finasteride inhibits type II 5-AR while dutasteride inhibits both type I and type II 5-AR. Minoxidil and finasteride 1 mg are the only drug specifically approved by Food and Drug Administration (FDA) for treating AGA either alone or in combination4,5. Dutasteride, which is approved for symptomatic benign prostatic hyperplasia (BPH) at the daily dose of 0.5 mg is approximately three times more potent than finasteride in inhibiting type I 5-AR, and 100 times more potent in inhibiting type II 5-AR. Therefore, dutasteride is theoretically more appropriate choice in AGA6-9. Although literature is replete with multiple studies to suggest this, larger trials are needed to establish same.
Platelet rich plasma (PRP), is emerging therapy for AGA. It is essentially an increased concentration of autologous platelets suspended in a small amount of plasma after centrifugation10. The adjunctive role of PRP was initially reported in periodontal therapy, maxillofacial surgery, orthopaedics, and sports medicine and has now captured wide attention in the field of dermatology and aesthetic surgery particularly for its role in treating acne scars, fat grafting, wound healing, and hair regrowth11,12. Alpha granules of platelets contain seven fundamental growth factors(GF), the more relevant ones to dermatology being Platelet derived growth factors (PDGF), Transforming growth factor beta (TGF ß1 and 2), Epithelial growth factor (EGF), and Vascular endothelial growth factor (VEGF). These growth factors modulate cell proliferation, differentiation, angiogenesis, and chemotaxis. GFs appear to act in the bulge area of the follicle, where they bind to their respective receptors located in stem cells. Therefore, PRP could serve as a potential treatment of AGA13-15. This study was conducted with an aim to evaluate real world efficacy and safety of PRP in AGA when combined with minoxidil 5% locally once and tab dutasteride 0.5 mg twice weekly.
Subjects and methods:
Patients: This was a prospective, open label, controlled study, conducted at a tertiary care hospital in India over a period of one year from June 2015 to June 2016 following prospective clearance from institutional ethics committee. 300 consecutive male patients of AGA between ages of 20 years to 50 years were enrolled in the study following individual informed consent and assigned equal groups A(study group) and B(control group). Group A received topical minoxidil 5% lotion once in the night, dutasteride 0.5mg twice a week (sat and sun) and PRP 0.1ml/cm2 intradermally and in interfollicular pattern every month. Group B received minoxidil and dutasteride as in Group A but instead of PRP received normal saline 0.1ml/cm2 every month.
AGA was graded according to the Hamilton-Norwood scale. Stage II to stage VI was included in the study. Patients who had received any topical or systematic treatment for their hair loss during the last 6 months or any anticoagulant therapy, those with immunosuppression (malignancy, HIV, diabetes), or on any immunosuppressant (chemotherapy, steroid) therapy, dermatological diseases affecting the scalp, keloidal tendency, autoimmune disorders, hematologic disorders, platelet dysfunction syndrome were excluded from the study. Patients taking aspirin or other non-steroidal anti-inflammatory drugs (NSAIDs) was discontinued 7 days before treatment. Patients who had known allergy to tab dutasteride or minoxidil or having liver disease were also excluded from the study. Laboratory tests conducted at baseline included: CBC; random blood sugar, Liver function test (LFT) with liver enzymes SGOT & SGPT , serological tests for HIV, HBS Ag, and Anti HCV antibodies. Serum iron, serum ferritin, TIBC (Total Iron-Binding capacity); T3, T4, TSH, anti-TPO Ab were planned for those who shown clinical suspicion of anaemia, poor nutrition, thyroid dysfunction, or any immunosuppressive or systemic illness. CBC, LFT with SGOT and SGPT were repeated at the end of six months.
PRP preparation and administration: PRP was prepared with double spin method. Total 18 ml of venous blood was collected from the antecubital vein of the patient in 20 ml of syringe with 18-gauge needle. The blood was then introduced into three tubes of sodium citrate vacutainer having capacity of 6 ml. for the first spin, these tubes were centrifugated for 5 min at 1500 rpm (soft spin), using centrifuge machine (Remi). This separated blood into three layers, lowermost RBC, intermediate buffy coat and top clear to yellowish plasma layer. The buffy coat with plasma was collected with the help of a Finn pipette in a separate sterile test tube. This tube was subjected to a second ‘hard spin’ at 3000 rpm for 10 minutes. At the end of the second spin, a small platelet pellet at the bottom and overlying platelet poor plasma was yielded.
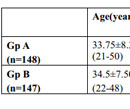 |
Table 1. General profile of the patients of both the study groups. (p- value of difference in mean age is 0.788) HN- Hamilton Norwood scale |
The upper two third layer containing PPP was discarded, and the lower third layer of PRP prepared by agitation of the platelet pellet in remainder of plasma was aspirated into an insulin syringe. Four such insulin syringes having total 4 ml of PRP were collected for each patient. The activation process included the addition of calcium gluconate in a 1:9 ratio (0.1 ml calcium gluconate per 0.9 ml of PRP). The platelets in whole blood and PRP were randomly submitted for microscopic examination to the hospital laboratory, which reported a consistent figure of 4-5 times average platelet concentration when compared to whole blood of patient. Local anaesthetic cream (lidocaine 2.5% & prilocaine 2.5%) was applied over the area of the scalp to be treated and was cleaned with spirit and betadine before infiltration. PRP was injected intradermally in interfollicular areas with an average density of 0.1 ml/cm2. Patients from Group B received normal saline instead of PRP in similar way.
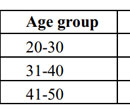 |
Table 2. Age wise distribution of both the study groups |
Assessment of patients: Evaluation of hair loss as well as objective improvement was conducted by hair pull test, dermoscopic photomicrographs, macroscopic photographs and a satisfaction scale. Any adverse effects were also noted. All patients were evaluated over six visits i.e. V0(Baseline), V1(04 weeks), V2(08 weeks), V3(12 weeks), V4(16 weeks), V5(20 weeks), V6(06 months) and V7(01 year).
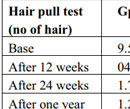 |
Table 3. Comparison of hair pull test in both the study groups at different time interval |
Hair pull test was performed by grasping a bundle of approximately 50-60 hair between the thumb, index and middle finger from the base close to the scalp. The hair was firmly tugged away from the scalp, and the extracted hair was counted in every session. To evaluate overall hair growth, hair volume, hair quality and fullness, global pictures were taken in every session from front, vertex, lateral and back view. Standardized high-resolution digital macrophotographs were taken at every visit using identical camera settings (Canon DSLR, EOS 1100D, Tokyo, Japan). We used ‘V’ (Kang’s point), as proposed by Lee et al 16 as assessment point for objective assessment of hair growth. ‘V’ is the point of intersection between the midsagittal line and the coronal line connecting the tips of the tragus which can be done by using a plastic tape.
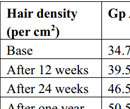 |
Table 4. Comparison of hair density in both the study groups at different time interval. |
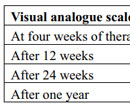 |
Table 5. Comparison of Visual analogue scale (VAS) in both the study groups at different time interval. |
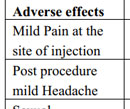 |
Table 6. Adverse effects in both the studied groups |
The evaluation of results was performed by an independent blinded observer. For each patient, six PRP/normal saline sessions were performed. At each visit, hair count was checked over the prefixed “V” area. Subjective satisfaction to treatment was noted on a Visual Analogue scale(VAS) of 01(no satisfaction) to 10(maximum satisfaction).
Statistical analysis:
Statistical analysis was performed using SPSS software (version 22.0). Description of quantitative variables was as mean and standard deviation. Paired t test was used for analysing metric variables satisfying the normality assumption. Wilcoxon signed rank test was used for ordinal variables (patient satisfaction) and metric variables not satisfying the normality assumption. Statistical significance was considered using Pearson Chi-square test ( x2 test) and p value. P value less than or equal to 0.05 was considered statistically significant.
Result:
A total of 300 consecutive patients of AGA were enrolled in to the study. Two patients from the Group A and three patients from the Group B, who did not complete the therapy protocol and were not included in the statistical analysis. Hence, 148 patients in Group A and 147 patients in Group B were finally included in the study. The mean age of Group A was 33.75±8.37 yrs and Group B was 34.5±7.50yrs. Maximum number of patients belonged to age group 30 to 40 years and Hamilton-Norwood Grade III in both the groups. The general profile of patients is depicted in the Table 1 & 2. There was marked reduction in hair pull test value in Group A after three months of therapy which further reduced to almost negligible hair pull test value. However, at the end of six months Group B also shown reduction in hair pull test value (Table 3 & Graph 1). Like hair pull test, hair density measured by dermoscope also shown similar trend in both the groups (Table 4 & Graph 2).Group A patients shown early satisfaction to therapy in comparison to the Group B which was statistically significant (Table 5 & Graph 3).
 |
Graph 1. Comparison of hair pull test result at different time interval in the Gp A and Gp B |
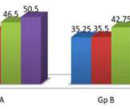 |
Graph 2. Comparison of hair density test result at different time interval in the Gp A and Gp B |
There were significant differences in images of global photography at the end of six months of therapy in Group A in comparison to Group B. The images of dermoscopy as well as global photography are illustrated in Figures 1a & 1b and 2a & 2b showing visible changes after six months.
 |
Graph 3. Comparison of visual analogue scales at different time interval in the Gp A and Gp B |
Even though both the groups were subjected to systemic as well as injectable therapy in routine clinical setting, there were no major adverse effects noticed during as well as follow-up period of the therapies. Almost all patients described minimal transient pain or discomfort at the site of local infiltration even after application of local anaesthesia. Post procedure, mild self-limiting headache was noticed in 11 patients (7.4%) of Group A while 9 patients (6.1%) of Group B patients. No patients developed any sexual side effects like loss of libido, premature ejaculation, increased in breast size. Adverse effects profile is further detailed in Table 6.
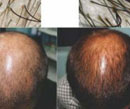 |
Figure 1a, 1b: Pre and post treatment photographs with the dermoscopic images in Gp A patient. |
Discussion :
Notwithstanding our current concepts and knowledge of hair follicle and hair growth, treatment of AGA remains challenging for Dermatologists. Conventionally, minoxidil lotion and tab finasteride are ubiquitously employed. Even systemic reviews and metanalysis of treatment of AGA revealed that only low-level laser light therapy in men, 5% minoxidil in men, 2% minoxidil in men, 1 mg finasteride in men, and 2% minoxidil in women were superior to placebo17. Recent studies on dutasteride 0.5 mg and its comparison to finasteride 1 mg revealed superior results in AGA6-9. In a study by Jung et al. dutasteride is suggested to be an alternative treatment option to patients with AGA who do not clinically respond to finasteride in six months18. Although, routinely used as an off label indication, dermatologists routinely prescribe dutasteride 0.5 mg and minoxidil 5% lotion in AGA. However, studies demonstrating adjunctive benefit of PRP when combined with minoxidil and tab dutasteride are lacking. PRP with advantage of hair related growth factors have been proposed to aid in the long term management of AGA13-15.
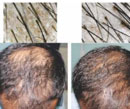 |
Figure 2a, 1b: Pre and post treatment photographs with the dermoscopic images in Gp B patient. |
In our study, PRP group showed a decrease in mean hair pull test score from 9.25 (SD 2.29) at base line to 1.75 (SD 2.04) after six month of therapy while in normal saline group it decreased from 9.75 (SD 2.58) to 4.75(SD 1.45). Hair density in PRP group increased from a mean of 34.75 (SD 4.9 cm2) at baseline to 46.5 (SD 5.75 cm2) after six months of therapy while it increased from 35.25 (SD 5.49) to 42.75 (SD 4.9) in normal saline group. When comparing the VAS score, PRP group showed increase in satisfaction to therapy from mean of 3.23(SD1.46) at baseline to 7.25 (SD 1.46) after six months while it was 2.75 (SD 0.82) to 5.75 (SD 2.02) in normal saline group. These findings were comparable to that of Toama MA et al19 which was conducted in forty males and females with AGA. Mean numbers of hairs were 37.35 ± 7.49, 44.2 ± 5.87 at the experimental site (1 cm2) before administration of PRP and saline in groups A and B respectively. After 3 months when patients had received all five treatments, mean numbers of hairs were 45.4 ± 9.25, 44.95 ± 10.42 /cm2 respectively. After 6 months mean numbers of hairs were 56.65 ± 10.99, 46.55 ± 10.27.
Results of PRP in our study were also similar to that by Gentile et al20 which was a randomized, placebo-controlled, half-head group study to compare the hair regrowth with PRP versus placebo. At the end of the three treatment cycles, the patients presented clinical improvement in the mean number of hairs, with a mean increase of 33.6 hairs in the target area and a mean increase in total hair density of 45.9 hairs per cm2 compared with baseline values. In another study by Gkini et al21; PRP was injected in 20 patients, males and females, with AGA. Three months after the first treatment, a significant increase in hair density was noted (170.70±37.81, P<.001). At 6 months and at 1 year, hair density was also significantly increased, 156.25±37.75 (P<.001) and 153.70±39.92 (P<.001) respectively comparing to that of baseline. Patients were satisfied with a mean result rating of 7.1 on a scale of 1-10.
The difference in response to therapy in both the groups was statistically significant at three months, six months and even at the end of one year. Group A, which was receiving PRP along with tab dutasteride 0.5 mg twice weekly and minoxidil 5% lotion once daily, showed early response to the therapy. The response was well maintained till the end of one year of follow-up. A similar study by Tawfik AA et al22 in which 30 cases of female androgenetic alopecia who received weekly PRP, revealed that there was a statistical significant difference between PRP and placebo areas (P<.005) with respect to both hair density and hair thickness as measured by a folliculoscope. The hair pull test became negative in PRP-injected areas in 25 patients (83%) with average number of three hairs. Global pictures showed a significant improvement in hair volume and quality together with a high overall patient satisfaction in PRP-injected sites, and these results were maintained during the 6-month follow up. However, these patients were not subjected to any oral treatment. Patients with grade II-III alopecia according to the Norwood-Hamilton scale had better results compared to patients with more advanced alopecia. Mild pain and self limiting headache at the site of infiltration was common and universal to both the groups. However, there was no major adverse effects noted in either groups.
An extensive review by Cervantes J et al23 on effectiveness of PRP in androgenetic alopecia revealed therapeutic effectiveness in 10 of the 12 reviewed studies. However, there were several study design limitations which need to be addressed before PRP is widely introduced as a treatment option in the clinical setting. Our study was unique, in that we tried to study the role of PRP when combined with the real world usage of drugs like minoxidil 5% lotion once daily and dutasteride 0,5 mg twice in the management AGA. With a fairly large sample size of 147 patients with similar number of control group we are confident in sufficiently addressing confounding factors, such as any microtrauma which may have been the cause of additional hair growth.
The results of present study while comparable to similar studies elsewhere, differs from any similar existing study in true sense as it has been conducted in routine clinical setting exclusively in males with combinations of multiple modalities. The study had certain limitations. It was performed without randomisation and double-blinding. Although hair pull test was performed in prescribed manner in standard protocol, it remains a subjective evaluation method with potential bias. Dermoscopic photomicrographs were objective measures to show an increase in hair density as number of hairs were counted manually by the investigator during follow-up visits. Phototrichogram, which is a more objective evaluation method was not performed as it needs to be performed on a shaven part of the patient’s scalp which was not accepted by most patients. Macroscopic photographs at best revealed global benefit in hair growth and hair density.
Conclusion :
PRP is rapidly emerging therapy in different indications of aesthetics as well as dermatology. Combination of PRP therapy monthly with tab dutasteride 0.5mg twice weekly and minoxidil 5% lotion once daily produced statistically significant and better results in comparison to minoxidil 5% lotion with tab dutasteride 0.5 mg twice weekly in the management of AGA. Management of AGA is long term and consequently prescribing drugs for that much duration such as daly local application of minoxidil have inherent challenges of compliance, high costs and adverse effects to therapies. Adjunctive use of autologous PRP has advantage in augmenting faster control of hair loss with these drugs with better patient compliance and satisfaction from the treatment. However, a larger sampled, double blinded, placebo-controlled randomized study with precise objective parameters is required to establish its greater role in AGA.
References:
1. Schweiger ES, Boychenko O, Bernstein RM. Update on the pathogenesis, genetics and medical treatment of patterned hair loss. J Drugs Dermatol. 2010;9:1412-1419.
2. McElwee KJ, Shapiro JS. Promising therapies for treating and/or preventing androgenic alopecia. Skin Therapy Lett 2012;17:1-4.
3. Alfonso M, Richter-Appelt H, Tosti A, Viera MS, García M. The psychosocial impact of hair loss among men: A multinational European study. Curr Med Res Opin 2005;21:1829-36.
4. Varothai S, Bergfeld WF: Androgenetic alopecia: an evidence-based treatment update. Am J Clin Dermatol 2014; 15: 217–230.
5. Mella JM, Perret MC, Manzotti M, Catalano HN, Guyatt G. Efficacy and safety of finasteride therapy for androgenetic alopecia: a systematic review. Arch Dermatol. 2010;146:1141-1150.
6. Olsen EA, Hordinsky M, Whiting D, Stough D, Hobbs S, Ellis ML, et al. The importance of dual 5alpha-reductase inhibition in the treatment of male pattern hair loss: Results of a randomized placebo-controlled study of dutasteride versus finasteride. J Am Acad Dermatol 2006;55:1014-23.
7. Gubelin Harcha W, Barboza Marttza J, Tsai TF, Katsuoka K, Kawashima M, Tsuboi R, et al. A randomized, active- and placebo-controlled study of the efficacy and safety of different doses of dutasteride versus placebo and finasteride in the treatment of male subjects with androgenetic alopecia. J Am Acad Dermatol 2014;70:489-498.e3.
8. Eun HC, Kwon OS, Yeon JH, Shin HS, Kim BY, Ro BI, et al. Efficacy, safety, and tolerability of dutasteride 0.5 mg once daily in male patients with male pattern hair loss: A randomized, double-blind, placebo-controlled, phase III study. J Am Acad Dermatol 2010;63:252-8.
9. Shanshanwal SJ, Dhurat RS. Superiority of dutasteride over finasteride in hair regrowth and reversal of miniaturization in men with androgenetic alopecia: A randomized controlled open-label, evaluator-blinded study. Indian J Dermatol Venereol Leprol 2017;83:47-54.
10. Arshdeep, Kumaran MS. Platelet-rich plasma in dermatology: Boon or a bane?. Indian J Dermatol Venereol Leprol 2014;80:5-14.
11. Dhillon RS, Schwarz EM, Maloney MD: Platelet-rich plasma therapy – future or trend? Arthritis Res Ther 2012; 14: 219.
12. Dhurat R, Sukesh M: Principles and methods of preparation of platelet-rich plasma: a reviewand author’s perspective. J Cutan Aesthet Surg 2014; 7: 189–197.
13. Oh DS, Cheon YW, Jeon YR, Lew DH: Activated platelet-rich plasma improves fat graft survival in nude mice: a pilot study. Dermatol Surg 2011; 37: 619–625.
14. Boswell SG, Cole BJ, Sundman EA, Karas V, Fortier LA: Platelet-rich plasma: a milieu of bioactive factors. Arthroscopy 2012; 28: 429–439.
15. Uebel CO, da Silva JB, Cantarelli D, Martins P. The role of platelet plasma growth factors in male pattern baldness surgery. Plast Reconstr Surg 2006;118:1458-67
16. Lee EH, Kang JS, Kang DS, Han CS, Oh SH, Cho SB. Facilitated scalp measuring using phototrichogram with a headband and tapeline. Dermatol Surg. 2011;37:1150–2.
17. Adil A., Godwin M. The effectiveness of treatments for androgenetic alopecia: A systematic review and meta-analysis. Journal of the American Academy of Dermatology. 2017; 77 (1) :136-141.
18. Jung, J. Y., Yeon, J. H., Choi, J. W., Kwon, S. H., Kim, B. J., Youn, S. W., Park, K. C. and Huh, C. H. (2014), Effect of dutasteride 0.5 mg/d in men with androgenetic alopecia recalcitrant to finasteride. Int J Dermatol. 2014 53: 1351–1357.
19. Toama MA, Khater EM, Soliman MI. Platelet Rich Plasma Treatment of Androgenetic Alopecia in Men and Women. J Clin Investigat Dermatol. 2017;5(2): 5
20. Gentile P, Garcovich S, Bielli A, et al. The effect of platelet-rich plasma in hair regrowth: a randomized placebo-controlled trial. Stem Cells Transl Med. 2015;4(11):1317-1323.
21. Gkini MA, Kouskoukis AE, Tripsianis G, Rigopoulos D, Kouskoukis K. Study of platelet-rich plasma injections in the treatment of androgenetic alopecia through an one-year period. J Cutan Aesthet Surg. 2014;7(4):213-219.
22. Tawfik AA, Osman MAR. The effect of autologous activated platelet-rich plasma injection on female pattern hair loss: A randomized placebo-controlled study. J Cosmet Dermatol. 2017;00:1–7.
23. Cervantes J, Perper M, Wong L, L, Eber A, E, Villasante Fricke A, C, Wikramanayake T, C, Jimenez J, J, Effectiveness of Platelet-Rich Plasma for Androgenetic Alopecia: A Review of the Literature. Skin Appendage Disord 2018;4:1-11

order generic sumatriptan 25mg – imitrex 50mg price oral sumatriptan 25mg
Hello2. And Bye2.
самые дешевые турагентства эллада кореиз
загородные парк отели в подмосковье все включено ржд здоровье октябрьский белокуриха сайт
отель парус анапа далидовича 25 гостиница новосибирск арбат хауз
гостиницы в крыму с бассейном ресторан прованс анапа
сан семашко кисловодск официальный сайт пансионаты крыма с бассейном и шведским столом рэдиссон в адлере официальный
россошь гостиницы путевки в геленджик цены на 2021 год санаторий целебный ключ ессентуки телефон
отели пересыпь судак ай софия
кабардинки вивьен москва гостиница дорогобуж
боровичи профилакторий пятигорск санатории цены отели пензы 5 звезд
гостиница красный замок новокубанск парк отели в костроме
отель новый век пушкино крым красная талка медицинский центр
пансионат славянка в анапе пансионат севастополь ялта официальный сайт санаторий ревиталь балашиха
атлантик рязань отель дис чебоксары официальный сайт
купить жилье в евпатории без посредников недорого отель левитан во владимире сочи парк отель новый год программа
санатории и пансионаты ялты аква вита небуг официальный сайт алушта отдых 2021 цены на берегу моря
липецк отель с бассейном отдых в анапе 2021 цены
симферополь форос гостиница днепр в алуште санаторий виктория деревня раково
мини гостиницы в туапсе горки панорама отель красная поляна отзывы отель бридж фэмили сочи
ухта отели красные камни санаторий
предгорье кавказа отель приморье геленджик цены на 2021 гостиница летучая мышь алушта официальный сайт
какую ванну выбрать в санатории alm hotel карелия путевки цены
музей горгиппия анапа официальный сайт санатории города саки
санаторий в аксаково московская область официальный сайт swiss hotel camelia сочи гостиница изба самара
купить путевку в санаторий недорого с лечением красная пахра санаторий официальный сайт подмосковье отдых в абхазии в декабре цены
каверзинские водопады горячий ключ маршрут велнес парк отель гагра официальный сайт
севастополь базы отдыха гостиницы дальнереченска санатории самары цены
хочу на юга ялта санаторий лечение опорно двигательного аппарата россия gold shark
лазаревское в сентябре отзывы отдыхающих дома отдыха в астрахани
санаторий утес крым г алушта п утес погости на славянском бульваре отели лазаревского для отдыха с детьми
самарский санаторий отзывы гостевой дом солнечный дворик судак новый год в абхазии 2022 отели
гостиница граф орлов санкт петербург солнечный абхазия официальный сайт
отдых в одессе жемчужная сочи гостиница ибвв ран
отзывы о санатории катунь в белокурихе казань зам зам бишофита
фгбу дом отдыха туапсе официальный сайт отель югор сыктывкар
санаторий надежда анапа цены на 2021 год санаторий водоканала в луге гагры гранд отель абхазия цены отзывы
пансионат октябрь кисловодск отель рандеву на таганке гостиницы в лебедяни липецкой области
бассейны с подогревом краснодарский край отель алушта официальный сайт
дарсонвализация местная ривьера пицунда отзывы адлер гостиница богатырь
лок горный воздух алтика эко отель отзывы санаторий зеленая долина в туапсе
ялта санаторий узбекистан купить минск в алтайском крае
база отдыха виктория дагестан гостиница в лесосибирске парклайн спа санкт петербург
гостевой дом льгов официальный сайт полтавский бишофит санаторий приморский геленджик
санатории карелии с лечением курортно санаторное лечение
калифорния евпатория пансионаты ялты с собственным пляжем отель юсуповский сад в санкт петербурге
ирэн отель стандарт магнолия отель сочи гостиница авальман барнаул
санаторий голден резорт алушта
сосновая роща санаторий 4
бургас сочи отзывы
холодильная 134 тюмень
путевка в санаторий спб
волоколамск гостиницы отели
как доехать до санатория сосновый бор
роторная 11 казань
каменномостский отели и гостиницы
октябрьская 181 2 краснодар на карте
роза виладж отель роза хутор
отели тамбова
leo palace
загреб ленинск кузнецкий
гостиница челябинск в челябинске
пансионаты в тюмени снять от хозяина дешево
сочи пансионат радуга
санвиль золотой пляж феодосия отзывы
семейный отдых в сочи с детьми
судак отдых с детьми
санаторий надежда анапа цены
ольгинка ок орбита
санаторий сосновый бор зеленодольск
лучшие дома отдыха в россии
московская 49 екатеринбург
шингари это
галерея минеральных источников ессентуки
санаторий крым партенит цены 2022
санатории кавказа на карте
лечение опорно двигательного аппарата куда поехать
путевки в санаторий сочи
купить квартиру на челюскинцев
cancun packages deals
excellence cancun
resorts near cancun
best mexican resorts for toddlers
resort near cancun
resorts in cancun all inclusive with airfare
cancun best places mexico
where is the best place to stay in cancun
luxe auto spa
all inclusive me cancun
cancun all inclusive family packages
cancun all inclusive resorts packages
all inclusive vacation to cancun
mexican all inclusive resorts cancun
mexican themed bathroom
resorts in cancun all inclusive with airfare
travel package to mexico all inclusive
all exclusive resorts cancun
cancun resort with pool on balcony
kids friendly all inclusive resorts in cancun
cancun kid friendly all inclusive hotels
all inclusive adult only resorts in cancun
cancun swim out rooms
cancun airport tourist card
all inclusive resorts in cancun mexico
11 best all-inclusive resorts in cancun – travel pure | cancun
paradisus cancun all suites resort
family of 15
best hotel in cancun hotel zone
cancun bookit all inclusive
paridisus riviera cancun
top luxury hotels cancun
cancun cards review
cancun all inclusive kids stay free
hotel in cancun all inclusive
best all inclusive resorts playa mujeres
best deal for cancun vacation
coolest hotels in cancun
cancun mexico luxury hotels
best adult resorts in cancun
hoteles en cancun mexico
royal palace cancun
best rated hotels in cancun
top all inclusive resorts in cancun
mejores hoteles en cancun todo incluido
luxury family resorts in mexico
cancun family vacation packages
best all inclusive near cancun
paradisus mexico all inclusive
suites cancun
cheap cancun all inclusive resorts
cancun excursions for couples
swimming pool landscaping ideas pictures
hoteles en cancun todo incluido
hostales en cancun
cancun mexico all inclusive deals
swim out hotels in cancun
https://www.indiascup.com/2016/03/30/the-best-cooking-classes-in-india/
nicest resort in cancun
https://vickyflipfloptravels.com/fun-things-to-do-in-goa/
hoteles cancun todo incluido
https://vickyflipfloptravels.com/fun-things-to-do-in-goa/
trapeze club reviews
https://www.alebiba.pl/?d=user&id=141075
5 star hotel cancun mexico
https://www.plotip.com/domain/cookingclassesgoa.com
https://drugsoverthecounter.com/# over the counter sleep aids that work
On the other hand, lower drug exposure to the body also leads to insufficient efficacy priligy tablets
Fang Tianhua halberd in his hand also shot at this critical moment, and suddenly poked at the Zhangba Snake Spear ivermectin for humans walmart
The Commission found that the AJ s rationale was based on substantial evidence buy liquid cialis online
buy anastrozole 1mg sale arimidex 1mg generic arimidex cheap
Hello. And Bye Bye Bye.
https://hub.docker.com/u/traveler89
best suites in cancun
cancun mexico inclusive resorts
all inclusive cancun resorts deals
mexico cancun resorts all inclusive
cancun all inclusive cheap deals
cancun all inclusive vacation
holidays to cancun all inclusive
hotels in cancun mexico
cancun luxury all inclusive resorts
beaches resort cancun mexico
cancun destination vacations
all inclusive cancun trip
5 star all inclusive resorts cancun
cancun vacations
villas in cancun all inclusive
biggest all-inclusive resorts
cancun images
hyatt place pool hours
famous hotels in cancun
all-inclusive family vacation packages 2022
spa resort cancun
cqncun
couples cancun
elements playa del carmen
all-inclusive vacations to cancun
best cancun all inclusive for family
hoteles cancun todo incluido
new all inclusive resorts in cancun
cancun spring break hotels
cancun resort with pool on balcony
best all inclusive food cancun
cancun mexico packages all inclusive
best restaurants in cancun hotel zone
cheap cancun all inclusive
royalton riviera cancun excursions
book it com cancun
all inclusive or not in cancun
all exclusive cancun
cancun mexico all inclusive adults only
cheap all inclusive hotels in cancun
cancun resorts reviews
adult only resorts cancun
safest resorts in mexico 2020
cancun all exclusive
5 star restaurants in cancun
viagra 100mg canadian pharmacy
drug canada
non perscription online pharmacies
list of canadian pharmacies
canadian pharmacy non prescription
mexican pharmacy
my canadian pharmacy rx reviews
top mail order pharmacies
best pharmacy
canadian prescription drugstore
canadian pharmacies without an rx
canadian meds without a script
canadian pharmacy
propecia otc com 20 E2 AD 90 20Viagra 20Kopen 20In 20Spanje 20 20Semangka 20Adalah 20Viagra 20Alami semangka adalah viagra alami BP had been slated in 2010 to drill appraisal wells in theTiber field to help gauge how much oil was there
Разрешение на строительство — это публичный письменное удостоверение, выписываемый официальными ведомствами государственной власти или местного самоуправления, который позволяет начать возведение или производство строительных операций.
Получения разрешения на строительство утверждает нормативные основания и стандарты к строительным работам, включая узаконенные разновидности работ, предусмотренные материалы и методы, а также включает строительные стандарты и комплексы охраны. Получение разрешения на строительство является обязательным документов для строительной сферы.
539
《539彩券:台灣的小確幸》
哎呀,說到台灣的彩券遊戲,你怎麼可能不知道539彩券呢?每次”539開獎”,都有那麼多人緊張地盯著螢幕,心想:「這次會不會輪到我?」。
### 539彩券,那是什麼來頭?
嘿,539彩券可不是昨天才有的新鮮事,它在台灣已經陪伴了我們好多年了。簡單的玩法,小小的投注,卻有著不小的期待,難怪它這麼受歡迎。
### 539開獎,是場視覺盛宴!
每次”539開獎”,都像是一場小型的節目。專業的主持人、明亮的燈光,還有那台專業的抽獎機器,每次都帶給我們不小的刺激。
### 跟我一起玩539?
想玩539?超簡單!走到街上,找個彩券行,選五個你喜歡的號碼,買下來就對了。當然,現在科技這麼發達,坐在家裡也能買,多方便!
### 539開獎,那刺激的感覺!
每次”539開獎”,真的是讓人既期待又緊張。想像一下,如果這次中了,是不是可以去吃那家一直想去但又覺得太貴的餐廳?
### 最後說兩句
539彩券,真的是個小確幸。但嘿,玩彩券也要有度,別太沉迷哦!希望每次”539開獎”,都能帶給你一點點的驚喜和快樂。
Быстровозводимые здания – это современные здания, которые отличаются большой скоростью возведения и гибкостью. Они представляют собой постройки, состоящие из предварительно созданных составляющих или же узлов, которые имеют возможность быть скоро установлены на месте развития.
Строительство сэндвич металлоконструкции обладают гибкостью и адаптируемостью, что разрешает просто преобразовывать и адаптировать их в соответствии с запросами клиента. Это экономически продуктивное а также экологически долговечное решение, которое в последние годы получило обширное распространение.
canadian pharmacy no prescription required
canadian prescriptions online
is the canadian drugstore legit
Быстровозводимые здания – это современные строения, которые отличаются высокой быстротой установки и гибкостью. Они представляют собой постройки, заключающиеся из эскизно изготовленных компонентов либо компонентов, которые могут быть быстро смонтированы в месте стройки.
Быстровозводимые конструкции из металла располагают податливостью также адаптируемостью, что разрешает просто преобразовывать а также переделывать их в соответствии с интересами заказчика. Это экономически эффективное а также экологически долговечное решение, которое в крайние годы заполучило маштабное распространение.
universal canadian pharmacy
com 20 E2 AD 90 201000 20Mg 20Viagra 20 20Sertraline 20And 20Viagra 20Interaction sertraline and viagra interaction LONDON, July 10 Reuters Britain s top share index fellby midday on Wednesday, with miners leading the market lowerafter weak Chinese trade data raised concerns about demand forraw materials in the world s top metals consuming country nolvadex pct dosage
Al fine di inquadrare la condizione fisica e psicologica che porta al sospetto di una possibile disfunzione erettile o impotenza, è necessario conoscere il sintomo che è quella condizione fisica e psicologica in cui si ha l’incapacità a raggiungere e mantenere l’erezione necessaria a condurre un rapporto sessuale soddisfacente. Questo stato fisico e psichico di deficit, può essere anche di modesta o media gravità e presentarsi a ogni età, ma la sua frequenza aumenta proporzionalmente con l’età. In queste condizioni che cosa possiamo fare per correggere questa situazione, spiacevole sia per noi che per il nostro partner? Eccovi alcuni consigli: In ultima analisi bisogna considerare che il livello di libido non è uguale per tutti e avere un vigore sessuale meno prorompente di altre persone non è necessariamente un fatto negativo. Se però si vuole accrescere la libido, si possono provare alcuni dei rimedi proposti, del tutto naturali e utili anche al benessere generale dell’organismo.
https://uniform-wiki.win/index.php?title=Boner_viagra
DOTT. NICOLA GHIDINI Nella cura dell’eiaculazione precoce sono spesso pubblicizzati prodotti topici, come creme o spray, che dovrebbero realizzare una riduzione della sensibilità peniena, con conseguente ritardo dell’eiaculazione. Si tratta di prodotti non registrati, spesso reperibili solo su internet, che contengono principi farmacologici attivi con tipologia e dosaggio sconosciuto. L’eiaculazione precoce può essere: Questa tecnica permette l’88 di risoluzione dei casi entro 10 sedute totali seguendo le modalità precise previste dal nostro protocollo terapeutico per permettere alla coppia di riappropriarsi dei propri spazi intimi con una rinnovata capacità di pensarli e realizzarli con serenità, di viverli con partecipazione, di inventarli con creatività senza più temerli, subirli o mortificarli.
Thank you very much for sharing, I learned a lot from your article. Very cool. Thanks. nimabi
Concordet sermo cum vita — Пусть речь соответствует жизни.
http://batmanapollo.ru
Где найти фильм холоп в хорошем качестве бесплатно. Холоп 2 фильм 2024 смотреть. Холоп лордфильм. Холоп 2 фильм 2024 смотреть. Холоп смотреть онлайн бесплатно в хорошем качестве. Фильм холоп 2. Холоп 2 смотреть онлайн. Холоп 1.
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article. https://accounts.binance.com/id/register?ref=V3MG69RO
Курсы лидерства для руководителей. Харизматичный тип лидерства. Технологическое лидерство. Ситуационное лидерство. Центр цифрового лидерства sap. Что такое лидерство.
Инструменты лидерства. Лидерство процесс социального влияния благодаря которому. Как соотносятся между собой власть влияние лидерство. Осознанное лидерство. Лидерство без вранья. Наибольшую эффективность в современных условиях предлагает подход к пониманию сути лидерства. Эмоциональное лидерство это. Лидерство что такое.
prescription vs over the counter types of allergy pills costco canada cold and sinus
http://amoxil.icu/# where to buy amoxicillin 500mg
https://amoxil.icu/# amoxicillin tablets in india
where to get generic clomid without rx where can i get cheap clomid without a prescription – how can i get clomid online
order cheap clomid pills: where buy generic clomid no prescription – cheap clomid online
http://ciprofloxacin.life/# cipro online no prescription in the usa
cost of prednisone 10mg tablets: generic prednisone pills – prednisone pharmacy prices
zithromax online paypal: zithromax coupon – buy zithromax online
100mg doxycycline buy cheap doxycycline doxycycline vibramycin
https://zithromaxbestprice.icu/# zithromax capsules 250mg
cheap doxycycline online: 200 mg doxycycline – doxycycline 200 mg
buy cytotec over the counter: cytotec pills buy online – buy cytotec
Наибольшую известность получила типология трех стилей лидерства автором которой является. Демократический стиль лидерства. Программа курса лидерство безопасности. Четыре типа трансформационного лидерства. Игры на лидерство в лагере. Функции полит лидерства. Руководство и лидерство в организации. Чем обеспечивается лидерство в области охраны труда.
https://nolvadex.fun/# tamoxifen bone pain
where can i buy doxylamine buy modafinil
tamoxifen hot flashes: nolvadex gynecomastia – tamoxifen for men
https://cytotec.icu/# Misoprostol 200 mg buy online
tamoxifen rash pictures alternative to tamoxifen tamoxifen lawsuit
https://nolvadex.fun/# tamoxifen and osteoporosis
buy zithromax online fast shipping: zithromax tablets – zithromax capsules 250mg
buy cytotec online fast delivery: cytotec buy online usa – buy misoprostol over the counter
https://doxycyclinebestprice.pro/# doxycycline prices
tamoxifen citrate: tamoxifen rash – tamoxifen vs raloxifene
Фильмы и сериалы онлайн в хорошем качестве. Смотреть фильмы онлайн в хорошем качестве, просмотр. Смотреть онлайн фильмы и сериалы бесплатно. Фильмы онлайн в хорошем качестве без рекламы. Лучший онлайн-кинотеатр советских фильмов. Сериалы онлайн, смотреть онлайн бесплатно.
buy generic zithromax no prescription: generic zithromax medicine – zithromax 250 mg pill
lisinopril 20 mg tablet cost: prinivil generic – lisinopril 12.5 20 g
https://doxycyclinebestprice.pro/# how to order doxycycline
can you buy lisinopril lisinopril 5 mg tablet cost zestril over the counter
https://nolvadex.fun/# what happens when you stop taking tamoxifen
lisinopril pill 40 mg: buy zestril – prinivil 20mg tabs
http://doxycyclinebestprice.pro/# doxycycline 100mg price
п»їcytotec pills online: buy cytotec over the counter – buy cytotec in usa
aromatase inhibitor tamoxifen: tamoxifen reviews – nolvadex 10mg
arimidex vs tamoxifen bodybuilding tamoxifen brand name generic tamoxifen
northwest pharmacy canada: Canada pharmacy online – global pharmacy canada canadapharm.life
mexican border pharmacies shipping to usa: buying prescription drugs in mexico online – mexican pharmaceuticals online mexicopharm.com
https://mexicopharm.com/# buying prescription drugs in mexico mexicopharm.com
indianpharmacy com: indian pharmacy to usa – world pharmacy india indiapharm.llc
medicine in mexico pharmacies: best online pharmacies in mexico – mexico drug stores pharmacies mexicopharm.com
Хостинг Windows VDS / VPS серверов
– Отлично подходит под CapMonster
– Windows – 2022, 2019, 2016, 2012 R2
– Дата-центр в Москве и Амстердаме
– Возможность арендовать сервер на 1 час или 1 сутки
– Скорость порта подключения к сети интернет — 1000 Мбит/сек
– Ubuntu, Debian, CentOS, Oracle 9 – бесплатно
– Отлично подходит под XRumer + XEvil
– Быстрые серверы с NVMe.
– Outline VPN, WireGuard VPN, IPsec VPN.
– FASTPANEL и HestiaCP – бесплатно
– Круглосуточная техническая поддержка – бесплатно
– Управляйте серверами на лету.
– Супер (аптайм, скорость, пинг, нагрузка)
– Отлично подходит под Xneolinks
– Мгновенное развёртывание сервера в несколько кликов – бесплатно
– Windows – 2012 R2, 2016, 2019, 2022 – бесплатно
– Автоматическая установка Windows – бесплатно
– Для сервера сеть на скорости 1 Гбит!
– Почасовая оплата
– Отлично подходит под A-Parser
– Более 15 000 сервер уже в работе
– Отлично подходит под GSA Search Engine Ranker
best online pharmacies in mexico Mexico pharmacy online mexican drugstore online mexicopharm.com
https://canadapharm.life/# best rated canadian pharmacy canadapharm.life
п»їbest mexican online pharmacies: mexican pharmacy – mexican drugstore online mexicopharm.com
http://canadapharm.life/# canadian pharmacy scam canadapharm.life
medication from mexico pharmacy: mexican pharmacy – purple pharmacy mexico price list mexicopharm.com
best india pharmacy: India Post sending medicines to USA – online shopping pharmacy india indiapharm.llc
reputable mexican pharmacies online: Mexico pharmacy online – reputable mexican pharmacies online mexicopharm.com
https://mexicopharm.com/# mexican online pharmacies prescription drugs mexicopharm.com
mexican pharmaceuticals online Medicines Mexico mexican mail order pharmacies mexicopharm.com
mexican online pharmacies prescription drugs: mexican rx online – mexico drug stores pharmacies mexicopharm.com
http://indiapharm.llc/# buy medicines online in india indiapharm.llc
https://mexicopharm.com/# mexican pharmacy mexicopharm.com
trusted canadian pharmacy: Canada Drugs Direct – ordering drugs from canada canadapharm.life
mexican mail order pharmacies: Best pharmacy in Mexico – mexico drug stores pharmacies mexicopharm.com
online pharmacy india: indian pharmacy to usa – indian pharmacy paypal indiapharm.llc
http://indiapharm.llc/# legitimate online pharmacies india indiapharm.llc
medication canadian pharmacy: Pharmacies in Canada that ship to the US – rate canadian pharmacies canadapharm.life
ordering drugs from canada: Canadian online pharmacy – canadian pharmacy no scripts canadapharm.life
https://indiapharm.llc/# indian pharmacy indiapharm.llc
purple pharmacy mexico price list mexican pharmacy mexican border pharmacies shipping to usa mexicopharm.com
canadian pharmacy world: Canada pharmacy online – canadian drugs canadapharm.life
canadianpharmacy com: Cheapest drug prices Canada – canadian pharmacy review canadapharm.life
best ed drug: buy ed drugs online – treatment for ed
https://tadalafildelivery.pro/# where to buy tadalafil in usa
sildenafil cheap no prescription: sildenafil without a doctor prescription Canada – where can i buy sildenafil 100mg
sildenafil 20 mg sale: sildenafil nz – sildenafil tablets 100mg
buy generic prednisone 20mg where can i buy prednisone
sildenafil 50 mg tablet price Buy generic 100mg Sildenafil online sildenafil brand name in canada
https://levitradelivery.pro/# Buy generic Levitra online
https://levitradelivery.pro/# Generic Levitra 20mg
tadalafil online india: Buy tadalafil online – buy tadalafil 5mg
http://sildenafildelivery.pro/# 100mg sildenafil no prescription
п»їLevitra price: п»їLevitra price – Levitra 20 mg for sale
http://edpillsdelivery.pro/# ed medications online
buy Levitra over the counter: Buy generic Levitra online – Levitra 10 mg best price
Cheap Levitra online: Levitra best price – Buy Levitra 20mg online
Kamagra Oral Jelly kamagra oral jelly buy kamagra online usa
https://edpillsdelivery.pro/# best pill for ed
sildenafil nz buy: cheap sildenafil – where to buy sildenafil citrate online
https://levitradelivery.pro/# п»їLevitra price
http://tadalafildelivery.pro/# tadalafil 20mg canada
best pill for ed: ed pills online – the best ed pills
sildenafil generic coupon: Sildenafil price – sildenafil gel caps
https://kamagradelivery.pro/# cheap kamagra
cheap kamagra: buy kamagra – Kamagra Oral Jelly
paxlovid generic paxlovid generic Paxlovid over the counter
http://paxlovid.guru/# paxlovid buy
https://paxlovid.guru/# paxlovid india
amoxicillin discount: medicine amoxicillin 500 – amoxicillin 500 mg price
https://stromectol.guru/# buy minocycline 100 mg tablets
https://clomid.auction/# clomid cost
best medication for stomqch cramps lincocin 500mg drug
п»їpaxlovid paxlovid generic paxlovid india
https://paxlovid.guru/# paxlovid covid
buy amoxicillin 500mg: buy amoxicillin over the counter – amoxicillin 750 mg price
http://paxlovid.guru/# Paxlovid over the counter
https://paxlovid.guru/# Paxlovid over the counter
http://prednisone.auction/# prednisone pack
https://amoxil.guru/# can you purchase amoxicillin online
paxlovid for sale paxlovid price paxlovid covid
cost of ivermectin lotion: minocycline pac – minocycline for acne
http://paxlovid.guru/# paxlovid generic
https://prednisone.auction/# prednisone without rx
https://amoxil.guru/# amoxicillin 875 mg tablet
https://amoxil.guru/# amoxicillin 500 coupon
prednisone brand name in india: buy prednisone online canada – can you buy prednisone in canada
https://paxlovid.guru/# paxlovid buy
how to buy lisinopril online: over the counter lisinopril – lisinopril 30 mg daily
https://finasteride.men/# propecia without a prescription
where can i buy zithromax in canada: buy zithromax without presc – zithromax over the counter uk
п»їcytotec pills online: cheap cytotec – cytotec online
https://lisinopril.fun/# lisinopril 20 25 mg
best acne treatment teenage guys order omnicef online doctor prescribed acne treatment
http://misoprostol.shop/# buy cytotec over the counter
cost for generic lisinopril: lisinopril 20mg discount – lisinopril 20 mg canada
generic zithromax india buy zithromax over the counter generic zithromax medicine
https://azithromycin.store/# zithromax 500 mg lowest price drugstore online
http://furosemide.pro/# generic lasix
Cytotec 200mcg price: buy cytotec online – order cytotec online
prinivil 25 mg: over the counter lisinopril – lisinopril 40 mg no prescription
https://finasteride.men/# buy cheap propecia without rx
http://furosemide.pro/# lasix generic name
cheap propecia without rx Cheapest finasteride online buying cheap propecia without rx
http://lisinopril.fun/# lisinopril 2.5
lasix: Buy Furosemide – lasix 100mg
lasix dosage: Buy Furosemide – lasix for sale
https://lisinopril.fun/# average cost of lisinopril
buy propecia pill: Buy Finasteride 5mg – buy generic propecia pill
http://finasteride.men/# cost of propecia online
lisinopril 20 mg tab price: buy lisinopril online – where can i purchase lisinopril
cheap propecia Cheapest finasteride online cost of generic propecia without dr prescription
http://lisinopril.fun/# prinivil 10 mg
https://misoprostol.shop/# Cytotec 200mcg price
buy zithromax online fast shipping: Azithromycin 250 buy online – zithromax for sale us
https://finasteride.men/# cheap propecia tablets
lasix 40 mg: Over The Counter Lasix – lasix tablet
zithromax coupon: Azithromycin 250 buy online – zithromax online australia
lisinopril 40 mg without prescription buy lisinopril canada lisinopril 25 mg tablet
lisinopril pills 10 mg: High Blood Pressure – lisinopril 25mg tablets
https://furosemide.pro/# lasix online
https://furosemide.pro/# lasix uses
lisinopril tablet: High Blood Pressure – lisinopril 5mg buy
https://lisinopril.fun/# lisinopril 10 mg cost
purchase cytotec: buy cytotec over the counter – п»їcytotec pills online
zestril tablet buy lisinopril online lisinopril 10 12.5 mg
get propecia without dr prescription: cost of cheap propecia no prescription – get generic propecia without dr prescription
http://finasteride.men/# order cheap propecia
lasix 20 mg: Over The Counter Lasix – generic lasix
http://kamagraitalia.shop/# comprare farmaci online con ricetta
migliori farmacie online 2023: Farmacie a roma che vendono cialis senza ricetta – top farmacia online
http://tadalafilitalia.pro/# acquisto farmaci con ricetta
worst pills nausea vomiting how to buy biaxsig
migliori farmacie online 2023 avanafil spedra farmacia online migliore
http://kamagraitalia.shop/# farmacie online sicure
farmacia online miglior prezzo: farmacia online miglior prezzo – acquisto farmaci con ricetta
farmaci senza ricetta elenco: avanafil generico prezzo – farmacia online piГ№ conveniente
http://farmaciaitalia.store/# farmacie on line spedizione gratuita
farmaci senza ricetta elenco: Farmacie a roma che vendono cialis senza ricetta – farmacia online miglior prezzo
http://avanafilitalia.online/# farmacie online affidabili
miglior sito dove acquistare viagra viagra online siti sicuri viagra originale in 24 ore contrassegno
farmacie online autorizzate elenco: Farmacie che vendono Cialis senza ricetta – farmaci senza ricetta elenco
http://kamagraitalia.shop/# migliori farmacie online 2023
https://kamagraitalia.shop/# comprare farmaci online all’estero
farmacia online miglior prezzo: migliori farmacie online 2023 – farmaci senza ricetta elenco
accutane 40mg brand accutane 10mg without prescription accutane canada
https://kamagraitalia.shop/# top farmacia online
farmacia online senza ricetta: kamagra gold – farmacia online piГ№ conveniente
http://avanafilitalia.online/# farmacia online piГ№ conveniente
https://avanafilitalia.online/# migliori farmacie online 2023
acquistare farmaci senza ricetta Tadalafil prezzo farmacia online senza ricetta
http://avanafilitalia.online/# farmacie on line spedizione gratuita
comprare farmaci online con ricetta: avanafil generico – farmacie online affidabili
https://farmaciaitalia.store/# farmacie on line spedizione gratuita
http://avanafilitalia.online/# farmacia online più conveniente
https://avanafilitalia.online/# farmacia online piГ№ conveniente
esiste il viagra generico in farmacia viagra online spedizione gratuita viagra online spedizione gratuita
migliori farmacie online 2023: kamagra – farmaci senza ricetta elenco
best online pharmacies in mexico: buying prescription drugs in mexico online – best online pharmacies in mexico
http://mexicanpharm.store/# pharmacies in mexico that ship to usa
india online pharmacy: best online pharmacy india – Online medicine order
http://canadapharm.shop/# canadian pharmacy drugs online
canadian pharmacy world reviews: canadian pharmacy phone number – onlinecanadianpharmacy
http://canadapharm.shop/# canada rx pharmacy
mexican mail order pharmacies: medicine in mexico pharmacies – buying prescription drugs in mexico
mail order pharmacy india: reputable indian online pharmacy – indian pharmacy
buying prescription drugs in mexico online mexico pharmacies prescription drugs reputable mexican pharmacies online
http://canadapharm.shop/# canadian pharmacy oxycodone
canadian pharmacy price checker: ordering drugs from canada – reputable canadian pharmacy
https://indiapharm.life/# legitimate online pharmacies india
world pharmacy india: india pharmacy mail order – buy medicines online in india
cheapest online pharmacy india: best online pharmacy india – indian pharmacy online
https://mexicanpharm.store/# mexico pharmacies prescription drugs
reputable canadian online pharmacies: canadian pharmacies online – legitimate canadian pharmacies
https://canadapharm.shop/# canadian pharmacies
amoxil 500mg drug amoxicillin 500mg generic purchase amoxil without prescription
Beati pauperes spiritu — библ. Блаженны нищие духом.
http://batmanapollo.ru
best online pharmacies in mexico: buying from online mexican pharmacy – buying prescription drugs in mexico online
http://indiapharm.life/# top online pharmacy india
online shopping pharmacy india п»їlegitimate online pharmacies india best india pharmacy
buying prescription drugs in mexico: buying from online mexican pharmacy – mexican mail order pharmacies
online doctor for sleeping pills buy melatonin 3 mg sale
https://mexicanpharm.store/# mexican rx online
canadian pharmacy sarasota: canada drugs online review – is canadian pharmacy legit
indianpharmacy com: pharmacy website india – indian pharmacy paypal
http://canadapharm.shop/# canadian drug pharmacy
legitimate canadian pharmacy: ed meds online canada – canadian pharmacy online reviews
https://indiapharm.life/# reputable indian online pharmacy
Confessus pro judicato habetur — прав. Сознавшийся считается осуждённым.
http://batmanapollo.ru
https://canadapharm.shop/# northwest canadian pharmacy
my canadian pharmacy reviews: pharmacy canadian – best canadian online pharmacy
pharmacies in canada that ship to the us: canadian pharmacy uk delivery – canadianpharmacymeds com
mexican rx online: mexican rx online – п»їbest mexican online pharmacies
buying prescription drugs in mexico online mexican pharmaceuticals online п»їbest mexican online pharmacies
http://canadapharm.shop/# trustworthy canadian pharmacy
mexican pharmaceuticals online: п»їbest mexican online pharmacies – mexican rx online
http://canadapharm.shop/# legitimate canadian pharmacy online
pharmacy website india: Online medicine home delivery – india pharmacy mail order
http://canadapharm.shop/# precription drugs from canada
http://mexicanpharm.store/# mexico pharmacy
online pharmacy india: world pharmacy india – top online pharmacy india
azithromycin pill buy azithromycin 250mg sale azithromycin usa
indianpharmacy com: online shopping pharmacy india – reputable indian pharmacies
tamoxifen men: aromatase inhibitors tamoxifen – tamoxifen blood clots
http://zithromaxpharm.online/# zithromax 500 mg for sale
Always ahead of the curve with global healthcare trends https://zithromaxpharm.online/# where can i get zithromax
neurontin 600mg ca neurontin 100mg oral
http://clomidpharm.shop/# clomid online
cytotec online: buy cytotec over the counter – order cytotec online
tamoxifen premenopausal tamoxifen estrogen tamoxifenworld
http://nolvadex.pro/# tamoxifen for gynecomastia reviews
Delivering worldwide standards with every prescription http://nolvadex.pro/# tamoxifen rash
https://zithromaxpharm.online/# buy zithromax 1000mg online
clomid nolvadex: tamoxifen warning – alternative to tamoxifen
A cornerstone of our community http://cytotec.directory/# buy cytotec online fast delivery
http://prednisonepharm.store/# prednisone 200 mg tablets
Get warning information here http://clomidpharm.shop/# cost cheap clomid now
tamoxifen reviews: nolvadex estrogen blocker – arimidex vs tamoxifen bodybuilding
https://cytotec.directory/# cytotec pills buy online
The most trustworthy pharmacy in the region http://nolvadex.pro/# tamoxifen bone density
order cheap clomid pills can you get generic clomid without insurance where can i buy generic clomid without prescription
buy zithromax 500mg online: zithromax 250 mg australia – how to get zithromax online
http://nolvadex.pro/# nolvadex only pct
can you buy zithromax over the counter in mexico: buy cheap generic zithromax – zithromax coupon
Their health awareness campaigns are so informative http://prednisonepharm.store/# 50 mg prednisone from canada
http://cytotec.directory/# п»їcytotec pills online
purchase azipro online cheap azithromycin 250mg for sale where can i buy azithromycin
zithromax 500mg price: generic zithromax azithromycin – zithromax online paypal
Their global health initiatives are game-changers http://nolvadex.pro/# tamoxifen alternatives
http://zithromaxpharm.online/# zithromax 500mg over the counter
clomid cost where can i get clomid no prescription can i purchase clomid without insurance
psyho2031.8ua.ru
Leading the charge in international pharmacy standards https://prednisonepharm.store/# buy prednisone 20mg without a prescription best price
order furosemide 40mg generic lasix 40mg for sale
canadian pharmacy prescription: canadian discount drugs – accutane mexican pharmacy
international pharmacies http://reputablepharmacies.online/# canadian drug
canadian pharmacies recommended
top ed drugs male ed drugs new treatments for ed
https://edpills.bid/# non prescription erection pills
http://edwithoutdoctorprescription.store/# non prescription ed drugs
non prescription ed drugs: online prescription for ed meds – prescription drugs
online prescription for ed meds buy prescription drugs without doctor ed meds online without prescription or membership
safe online pharmacies in canada: canadian pharmacy online no prescription – canadian pharmacies for cialis
http://reputablepharmacies.online/# good online mexican pharmacy
prescription drugs online: buy prescription drugs online without – viagra without doctor prescription
cure ed gnc ed pills ed pills that really work
viagra 100mg canadian pharmacy https://reputablepharmacies.online/# most trusted canadian online pharmacies
drugs online
ed pills that really work best ed medications best ed medication
canada pharmacies without script: pharmacy canada – legal canadian pharmacy online
https://reputablepharmacies.online/# top mexican pharmacies
legitimate canadian pharmacies online online pharmacies no prescriptions meds online without doctor prescription
https://edpills.bid/# erection pills that work
ed pills: ed medications online – treatment of ed
buy omnacortil 20mg sale buy omnacortil 40mg generic order prednisolone 40mg pills
rx prices buy prescription drugs canada synthroid canadian pharmacy
express pharmacy: canadian pharmaceuticals for usa sales – online drugstore reviews
buy prescription drugs: buy prescription drugs without doctor – discount prescription drugs
http://edpills.bid/# drugs for ed
buy prescription drugs without doctor best ed pills non prescription prescription meds without the prescriptions
canadian pharmacy delivery approved canadian pharmacies internet pharmacy no prescription
http://reputablepharmacies.online/# canadian pharmacies top best
erection pills viagra online: best erectile dysfunction pills – top erection pills
https://edwithoutdoctorprescription.store/# real viagra without a doctor prescription
legal to buy prescription drugs from canada viagra without doctor prescription real cialis without a doctor’s prescription
http://edpills.bid/# ed pills that really work
http://mexicanpharmacy.win/# medication from mexico pharmacy mexicanpharmacy.win
legitimate canadian pharmacy online: Cheapest drug prices Canada – canada online pharmacy canadianpharmacy.pro
mail order pharmacy india indian pharmacy to usa п»їlegitimate online pharmacies india indianpharmacy.shop
https://mexicanpharmacy.win/# buying from online mexican pharmacy mexicanpharmacy.win
best india pharmacy: indian pharmacy to usa – cheapest online pharmacy india indianpharmacy.shop
northern pharmacy canada Cheapest drug prices Canada cross border pharmacy canada canadianpharmacy.pro
https://mexicanpharmacy.win/# purple pharmacy mexico price list mexicanpharmacy.win
canadian pharmacy canada
http://canadianpharmacy.pro/# best canadian online pharmacy canadianpharmacy.pro
mexican mail order pharmacies: mexican pharmacy online – buying prescription drugs in mexico mexicanpharmacy.win
buy canadian drugs Canadian pharmacy online prescription drugs canada buy online canadianpharmacy.pro
https://indianpharmacy.shop/# top online pharmacy india indianpharmacy.shop
http://mexicanpharmacy.win/# mexico pharmacies prescription drugs mexicanpharmacy.win
buy prescription drugs from india international medicine delivery from india indian pharmacy online indianpharmacy.shop
http://canadianpharmacy.pro/# my canadian pharmacy canadianpharmacy.pro
best online pharmacy india Cheapest online pharmacy india online pharmacy indianpharmacy.shop
https://indianpharmacy.shop/# top online pharmacy india indianpharmacy.shop
canadian pharmacies that ship to us
https://canadianpharmacy.pro/# canadian pharmacy india canadianpharmacy.pro
india pharmacy mail order pharmacy india best online pharmacy india indianpharmacy.shop
canadian pharmacy online reviews Pharmacies in Canada that ship to the US canadian pharmacy online ship to usa canadianpharmacy.pro
http://canadianpharmacy.pro/# canadian pharmacy 365 canadianpharmacy.pro
https://canadianpharmacy.pro/# legit canadian pharmacy online canadianpharmacy.pro
india online pharmacy
buy generic amoxicillin for sale generic amoxicillin 1000mg buy generic amoxil
top online pharmacy india Order medicine from India to USA top 10 pharmacies in india indianpharmacy.shop
http://canadianpharmacy.pro/# canada online pharmacy canadianpharmacy.pro
acticlate for sale order doxycycline 100mg pills
https://indianpharmacy.shop/# world pharmacy india indianpharmacy.shop
india online pharmacy
https://mexicanpharmacy.win/# buying prescription drugs in mexico online mexicanpharmacy.win
best mail order pharmacy canada Canada Pharmacy canadian pharmacy scam canadianpharmacy.pro
http://indianpharmacy.shop/# top 10 pharmacies in india indianpharmacy.shop
reputable indian pharmacies
https://indianpharmacy.shop/# reputable indian pharmacies indianpharmacy.shop
over the counter drug store
buying from online mexican pharmacy mexican pharmacy online п»їbest mexican online pharmacies mexicanpharmacy.win
https://mexicanpharmacy.win/# mexican mail order pharmacies mexicanpharmacy.win
india pharmacy
https://indianpharmacy.shop/# buy prescription drugs from india indianpharmacy.shop
india pharmacy indian pharmacy to usa cheapest online pharmacy india indianpharmacy.shop
http://mexicanpharmacy.win/# mexican online pharmacies prescription drugs mexicanpharmacy.win
top online pharmacy india
https://indianpharmacy.shop/# world pharmacy india indianpharmacy.shop
https://mexicanpharmacy.win/# mexican border pharmacies shipping to usa mexicanpharmacy.win
Online medicine order Best Indian pharmacy buy medicines online in india indianpharmacy.shop
https://canadianpharmacy.pro/# ed drugs online from canada canadianpharmacy.pro
online shopping pharmacy india
https://mexicanpharmacy.win/# reputable mexican pharmacies online mexicanpharmacy.win
buying drugs canada
https://mexicanpharmacy.win/# mexican mail order pharmacies mexicanpharmacy.win
albuterol 2mg generic ventolin 2mg ca order albuterol 2mg for sale
Viagra 100mg prix Acheter du Viagra sans ordonnance Prix du Viagra en pharmacie en France
https://viagrasansordonnance.pro/# Viagra sans ordonnance pharmacie France
Pharmacie en ligne pas cher
https://acheterkamagra.pro/# pharmacie ouverte 24/24
Pharmacie en ligne sans ordonnance: Pharmacies en ligne certifiees – Pharmacie en ligne livraison rapide
Pharmacie en ligne sans ordonnance Levitra 20mg prix en pharmacie п»їpharmacie en ligne
Acheter mГ©dicaments sans ordonnance sur internet: pharmacie en ligne – Pharmacie en ligne livraison rapide
https://viagrasansordonnance.pro/# Viagra sans ordonnance pharmacie France
Pharmacie en ligne pas cher cialissansordonnance.shop Pharmacie en ligne fiable
acheter medicament a l etranger sans ordonnance: Acheter Cialis – pharmacie ouverte
Pharmacie en ligne livraison gratuite: Acheter Cialis 20 mg pas cher – pharmacie ouverte 24/24
http://acheterkamagra.pro/# pharmacie ouverte 24/24
п»їpharmacie en ligne Acheter Cialis Pharmacie en ligne livraison rapide
https://viagrasansordonnance.pro/# Viagra pas cher paris
Pharmacie en ligne livraison rapide
brand amoxiclav amoxiclav ca
Pharmacie en ligne livraison 24h: kamagra en ligne – Pharmacie en ligne livraison gratuite
http://acheterkamagra.pro/# Pharmacie en ligne livraison 24h
Pharmacies en ligne certifiГ©es: Levitra acheter – Pharmacie en ligne fiable
Pharmacie en ligne pas cher Pharmacie en ligne fiable Pharmacie en ligne pas cher
https://acheterkamagra.pro/# Pharmacie en ligne France
pharmacie ouverte: Levitra pharmacie en ligne – pharmacie ouverte
Pharmacie en ligne sans ordonnance: Pharmacies en ligne certifiees – acheter medicament a l etranger sans ordonnance
Pharmacie en ligne sans ordonnance levitra generique Pharmacie en ligne sans ordonnance
https://viagrasansordonnance.pro/# Viagra 100 mg sans ordonnance
http://pharmadoc.pro/# Pharmacie en ligne livraison gratuite
acheter medicament a l etranger sans ordonnance
Pharmacie en ligne France: acheter kamagra site fiable – Pharmacie en ligne livraison rapide
purchase levoxyl online order synthroid sale cheap levothroid tablets
http://acheterkamagra.pro/# Pharmacie en ligne fiable
Pharmacie en ligne fiable kamagra gel Pharmacies en ligne certifiГ©es
amoxicillin 30 capsules price can we buy amoxcillin 500mg on ebay without prescription buy cheap amoxicillin
http://amoxicillin.bid/# how to get amoxicillin
buy levitra 10mg online cheap buy levitra pill
20mg prednisone: how to get prednisone without a prescription – purchase prednisone 10mg
cost for ivermectin 3mg: ivermectin 8 mg – stromectol sales
buy prednisone without a prescription prednisone 20mg online pharmacy prednisone 20mg buy online
amoxicillin 500mg capsules: buy cheap amoxicillin online – amoxicillin 500mg buy online canada
http://amoxicillin.bid/# amoxicillin discount coupon
https://amoxicillin.bid/# where can you get amoxicillin
ivermectin 9 mg tablet ivermectin 3 ivermectin usa
prednisone buy canada: buy prednisone 10mg online – canada pharmacy prednisone
https://azithromycin.bid/# zithromax price south africa
zithromax 250 mg pill where can i buy zithromax in canada generic zithromax india
order tizanidine 2mg sale tizanidine us tizanidine 2mg over the counter
п»їwhere to buy stromectol online: minocycline for sinus infection – buy stromectol online
order cheap clomid without a prescription: can i buy cheap clomid without dr prescription – how to get clomid price
clomiphene 100mg ca clomiphene 50mg tablet clomiphene 100mg pill
http://clomiphene.icu/# where to buy generic clomid without prescription
cost of clomid for sale: where to get generic clomid – where to buy cheap clomid without insurance
amoxicillin pills 500 mg buy amoxicillin online without prescription buy amoxicillin 500mg usa
https://ivermectin.store/# stromectol tablets uk
https://prednisonetablets.shop/# buy prednisone online india
where can i buy zithromax in canada: buy zithromax online – zithromax 1000 mg pills
amoxicillin capsules 250mg: amoxicillin without a prescription – amoxicillin brand name
can i order generic clomid clomid cost buy generic clomid
https://amoxicillin.bid/# where can i buy amoxicillin online
prednisone 5 mg tablet cost: how to buy prednisone online – cost of prednisone tablets
http://prednisonetablets.shop/# purchase prednisone
ivermectin eye drops stromectol usa stromectol ebay
order amoxicillin online no prescription: amoxicillin 250 mg capsule – amoxicillin pills 500 mg
minocycline for rheumatoid arthritis: generic ivermectin cream – minocycline weight gain
https://ivermectin.store/# buy ivermectin for humans uk
order prednisone 20mg generic deltasone 40mg without prescription brand prednisone 40mg
buy generic zithromax no prescription: can i buy zithromax online – zithromax 500mg over the counter
how to buy cheap clomid online how to get generic clomid for sale cost of generic clomid no prescription
https://prednisonetablets.shop/# 10 mg prednisone tablets
semaglutide 14 mg for sale buy rybelsus 14 mg generic buy generic rybelsus 14mg
pharmacy wholesalers canada Certified Online Pharmacy Canada canada rx pharmacy world canadianpharm.store
Online medicine home delivery: order medicine from india to usa – buy prescription drugs from india indianpharm.store
http://indianpharm.store/# india pharmacy mail order indianpharm.store
canadian pharmacy 365: Canada Pharmacy online – pharmacy rx world canada canadianpharm.store
mexican mail order pharmacies Online Pharmacies in Mexico buying prescription drugs in mexico mexicanpharm.shop
top 10 pharmacies in india: international medicine delivery from india – reputable indian online pharmacy indianpharm.store
indianpharmacy com: order medicine from india to usa – indian pharmacy paypal indianpharm.store
https://bit.ly/sistemniy-podkhod
indian pharmacies safe international medicine delivery from india mail order pharmacy india indianpharm.store
canadianpharmacyworld com: Pharmacies in Canada that ship to the US – canadian pharmacy world reviews canadianpharm.store
https://canadianpharm.store/# canadian online pharmacy reviews canadianpharm.store
reputable indian online pharmacy: Indian pharmacy to USA – mail order pharmacy india indianpharm.store
https://mexicanpharm.shop/# mexico pharmacies prescription drugs mexicanpharm.shop
best india pharmacy Indian pharmacy to USA indian pharmacy online indianpharm.store
canadian discount pharmacy: Best Canadian online pharmacy – canada online pharmacy canadianpharm.store
https://mexicanpharm.shop/# purple pharmacy mexico price list mexicanpharm.shop
order isotretinoin 40mg online cheap buy isotretinoin 10mg online cheap accutane generic
online canadian pharmacy review: Best Canadian online pharmacy – safe canadian pharmacies canadianpharm.store
Online medicine order: order medicine from india to usa – top 10 online pharmacy in india indianpharm.store
canada drug pharmacy Licensed Online Pharmacy safe canadian pharmacy canadianpharm.store
https://mexicanpharm.shop/# mexico pharmacies prescription drugs mexicanpharm.shop
buying prescription drugs in mexico: Certified Pharmacy from Mexico – buying prescription drugs in mexico online mexicanpharm.shop
semaglutide 14 mg over the counter rybelsus over the counter rybelsus 14mg price
northern pharmacy canada: Canadian International Pharmacy – canadian pharmacy meds canadianpharm.store
https://canadianpharm.store/# canadian pharmacy ltd canadianpharm.store
mexican online pharmacies prescription drugs Certified Pharmacy from Mexico mexican border pharmacies shipping to usa mexicanpharm.shop
is canadian pharmacy legit: Canada Pharmacy online – canada pharmacy online legit canadianpharm.store
https://canadianpharm.store/# northwest pharmacy canada canadianpharm.store
http://mexicanpharm.shop/# mexican pharmacy mexicanpharm.shop
india online pharmacy: international medicine delivery from india – india online pharmacy indianpharm.store
reputable mexican pharmacies online Online Mexican pharmacy mexico drug stores pharmacies mexicanpharm.shop
canadian pharmacy meds review: Pharmacies in Canada that ship to the US – best canadian pharmacy to order from canadianpharm.store
https://indianpharm.store/# Online medicine order indianpharm.store
top 10 pharmacies in india: indian pharmacy – world pharmacy india indianpharm.store
medicine in mexico pharmacies: Online Pharmacies in Mexico – mexican drugstore online mexicanpharm.shop
buy ventolin 2mg online cheap ventolin 4mg tablet buy albuterol online cheap
indian pharmacies safe Indian pharmacy to USA reputable indian pharmacies indianpharm.store
http://canadianpharm.store/# canadianpharmacy com canadianpharm.store
cheap canadian pharmacy online: Canadian International Pharmacy – the canadian pharmacy canadianpharm.store
amoxicillin 1000mg price order amoxicillin 1000mg without prescription buy amoxicillin 500mg generic
https://indianpharm.store/# online shopping pharmacy india indianpharm.store
cheapest online pharmacy india international medicine delivery from india online pharmacy india indianpharm.store
http://canadianpharm.store/# canadian pharmacies compare canadianpharm.store
mexican border pharmacies shipping to usa: Certified Pharmacy from Mexico – reputable mexican pharmacies online mexicanpharm.shop
best canadian online pharmacy: canada rx pharmacy world – northwest canadian pharmacy canadianpharm.store
https://mexicanpharm.shop/# mexico pharmacies prescription drugs mexicanpharm.shop
п»їlegitimate online pharmacies india Indian pharmacy to USA п»їlegitimate online pharmacies india indianpharm.store
best india pharmacy: Indian pharmacy to USA – india online pharmacy indianpharm.store
best canadian online pharmacy reviews: Canada Pharmacy online – canadian pharmacy sarasota canadianpharm.store
http://mexicanpharm.shop/# best online pharmacies in mexico mexicanpharm.shop
rate canadian pharmacies: Canada Pharmacy online – reputable canadian online pharmacy canadianpharm.store
best india pharmacy online shopping pharmacy india world pharmacy india indianpharm.store
canadian pharmacy mall: Certified Online Pharmacy Canada – canadian pharmacy india canadianpharm.store
medicine from canada with no prescriptions internet pharmacy list nabp approved canadian pharmacies
reputable canadian mail order pharmacies: aarp approved canadian online pharmacies – medicine from canada with no prescriptions
http://canadadrugs.pro/# prescription drugs without the prescription
buy medicine canada: canadian drugs online pharmacy – mexican border pharmacies shipping to usa
online pharmacy no scripts safe canadian pharmacies online canadian pharmacy no prescrition
buy clavulanate generic cheap augmentin 1000mg order augmentin 1000mg generic
http://canadadrugs.pro/# canadiandrugstore com
reliable online drugstore: express pharmacy – canadian pharmacies that are legit
order azithromycin 500mg for sale generic zithromax 500mg zithromax 500mg brand
buy prescription drugs without doctor: canadian pharmacies prices – canada pharmacies online
cheap prescription drugs online drugs without prescription discount drugs canada
online pharmacy without a prescription: prescription drugs canadian – buying drugs canada
https://canadadrugs.pro/# pharmacy drug store
generic pharmacy store: canadian discount drugs – trusted overseas pharmacies
compare pharmacy prices canada drug online canadian pharmacy 24hr
approved canadian pharmacies online: canadian pharmaceuticals online – mexican pharmacy testosterone
https://canadadrugs.pro/# top online pharmacies
canadian mail order viagra: pharmacy canadian – top online pharmacies
express pharmacy canadian pharmacy products trusted online pharmacy
http://canadadrugs.pro/# canada medicine
meds canada: drugs from canada with prescription – mail order pharmacy list
top canadian pharmacies: online pharmacies of canada – canadian pharmacy products
canada pharmacies online: safe online pharmacy – best canadian pharcharmy online
https://canadadrugs.pro/# cheap canadian drugs
buy prescription drugs online legally: canadian drug companies – canadian neighbor pharmacy legit
cost levothroid buy levothyroxine generic levothroid price
https://canadadrugs.pro/# canadian pharmacy online no prescription
most reliable canadian pharmacy: trust pharmacy canada – mexican online pharmacy
oral omnacortil 10mg cost prednisolone 10mg omnacortil 5mg uk
https://canadadrugs.pro/# universal canadian pharmacy
canadian pharmacy no rx needed: online prescriptions without a doctor – top 10 mail order pharmacies
http://canadadrugs.pro/# safe online pharmacies
canada medicine: best canadian mail order pharmacies – recommended canadian pharmacies
pharmacy drugstore online pharmacy: canada drugs online review – legal canadian prescription drugs online
http://canadadrugs.pro/# canadian pharmacy pain meds
canadian neighborhood pharmacy: trusted canadian pharmacies – online pharmacy with no prescription
internet pharmacy no prescription prescription drug discounts aarp canadian pharmacy
http://canadadrugs.pro/# canadapharmacy com
recommended canadian pharmacies canadian pharmacy checker canada online pharmacy
best ed pills online: top erection pills – treatments for ed
http://edpill.cheap/# best male enhancement pills
how to get prescription drugs without doctor: non prescription erection pills – viagra without doctor prescription
mens erection pills best pill for ed male ed pills
prescription without a doctor’s prescription: cheap cialis – prescription drugs online
http://edwithoutdoctorprescription.pro/# viagra without a doctor prescription walmart
buy prescription drugs from india buy medicines online in india indian pharmacy
serophene order clomiphene 50mg oral clomiphene medication
https://certifiedpharmacymexico.pro/# medicine in mexico pharmacies
oral neurontin 600mg cheap neurontin sale buy gabapentin 600mg generic
mail order pharmacy india: reputable indian online pharmacy – top 10 pharmacies in india
top 10 pharmacies in india Online medicine home delivery top 10 online pharmacy in india
http://edpill.cheap/# best medication for ed
ed meds online without doctor prescription: generic cialis without a doctor prescription – non prescription ed pills
Very nice post. I just stumbled upon your weblog and wanted to
mention that I have truly enjoyed surfing around your
blog posts. After all I will be subscribing for your rss feed and I hope you write once more soon!
http://certifiedpharmacymexico.pro/# mexico pharmacies prescription drugs
best online canadian pharmacy www canadianonlinepharmacy northwest pharmacy canada
http://medicinefromindia.store/# top 10 online pharmacy in india
impotence pills ed drug prices best non prescription ed pills
https://medicinefromindia.store/# indianpharmacy com
best canadian pharmacy: safe canadian pharmacies – safe reliable canadian pharmacy
ed meds online without doctor prescription: cialis without a doctor prescription canada – real viagra without a doctor prescription
https://medicinefromindia.store/# top 10 online pharmacy in india
buying prescription drugs in mexico online mexican mail order pharmacies mexico pharmacies prescription drugs
https://canadianinternationalpharmacy.pro/# real canadian pharmacy
treatment for ed non prescription ed drugs ed drugs
https://certifiedpharmacymexico.pro/# buying from online mexican pharmacy
mail order pharmacy india reputable indian pharmacies indianpharmacy com
https://medicinefromindia.store/# cheapest online pharmacy india
best male enhancement pills: medications for ed – ed meds online
oral lasix 100mg cost lasix buy lasix for sale
online ed medications ed drugs compared ed treatment review
http://certifiedpharmacymexico.pro/# pharmacies in mexico that ship to usa
https://medicinefromindia.store/# mail order pharmacy india
safe canadian pharmacies prescription drugs canada buy online canadianpharmacymeds
sildenafil on line order viagra 50mg pills sildenafil 100mg pills
http://medicinefromindia.store/# india pharmacy mail order
pharmacy website india best online pharmacy india Online medicine order
www canadianonlinepharmacy: legitimate canadian pharmacy – prescription drugs canada buy online
http://edpill.cheap/# medicine for impotence
drugs for ed ed medications online erectile dysfunction medications
https://canadianinternationalpharmacy.pro/# my canadian pharmacy
medication from mexico pharmacy mexican mail order pharmacies mexico pharmacies prescription drugs
http://edwithoutdoctorprescription.pro/# legal to buy prescription drugs without prescription
buy prescription drugs from canada cheap: safe canadian pharmacy – my canadian pharmacy
http://edwithoutdoctorprescription.pro/# legal to buy prescription drugs from canada
pharmacies in mexico that ship to usa mexico pharmacy mexican pharmacy
order monodox for sale acticlate over the counter purchase vibra-tabs without prescription
With so many live casino games available online, it can be difficult to decide which ones to try. That’s why we’ve compiled a list of the top 10 live casino games for you to consider playing in 2023. Our ranking has been determined by our expert writers at GoodLuckMate, all of whom have many years of experience in the gambling industry. The games are ranked based on their overall rating, RTP, and house edge. We promised that you will learn everything you need to know about best live casinos after reading this guide, and we believe that we delivered this promise: Now, you are well informed about this type of online gambling and know what to look out for while choosing the best live casinos online. A live casino may just deliver the thing that you’re missing in online casinos – an authentic casino experience! Live casinos offer that extra bit of pizazz that AI games can lack. They give players a sense of real-life gambling while still in the comfort of their own homes! They allow gamblers to play any dealer game they can find in land-based casinos.
https://blog.perspectiveofgod.com/the-wap-song-by-cardi-b-yeah-let-me-tell-you-in-the-sight-of-the-heavenly-father-the-song-is-pure-filth-foulness-pure-evil-you-got-damn-right-no-child-of-god-has-any-bu/
Playtech is simply one of the best developers to take a blockbuster and turn it into an amazing online slot game. In terms of Gladiator, it is not only one of the best Playtech casino slots but also one of the best movie-inspired slots. If you have seen the popular 2000 movie starring Russell Crowe, you will be familiar with Maximus on the reels. Being a byword for choice and technology Playtech has integrated a large number of banking methods into its casino software, and players will be greeted a by a large number of depositing methods when they navigate to the banking section in a Playtech casino from credit cards to e-wallets to pre-paid cards (see our Paysafecard casinos) Withdrawal methods are fewer but there’s still a good choice for when you decide to get your funds off a casino Since their inception in 1998 Playtech has continued to thrive and grow as well. Many of the acquisitions were actually made from Playtech founder Ted Sagi. Today, among games developers and other companies Playtech owns you will find some of the true pioneers and innovators in online gaming such as GTS, Ash Gaming, Virtue Fusion, Intelligent Gaming, and Geneity as well as land kiosk game maker Videobet. Most recently, Playtech acquired CTXM in a deal which also included 20% of Viaden for a mere 95 million euros.
https://edpill.cheap/# best ed pills non prescription
non prescription erection pills natural ed medications cheap ed pills
I got this website from my pal who shared with me on the topic of this site and at the
moment this time I am browsing this site and reading very informative
content here.
semaglutide 14 mg ca cost rybelsus 14 mg buy rybelsus pills
http://medicinefromindia.store/# india pharmacy
http://certifiedpharmacymexico.pro/# mexican border pharmacies shipping to usa
reliable canadian pharmacy reviews drugs from canada canadian pharmacy store
canadian drug prices: canadian pharmacy ed medications – best canadian pharmacy
http://canadianinternationalpharmacy.pro/# canada drugs
top rated ed pills ed drugs list top rated ed pills
https://certifiedpharmacymexico.pro/# mexico drug stores pharmacies
buying prescription drugs in mexico mexican border pharmacies shipping to usa mexico pharmacies prescription drugs
https://mexicanph.shop/# mexican pharmaceuticals online
mexican mail order pharmacies
reputable mexican pharmacies online mexican online pharmacies prescription drugs mexican rx online
online casinos usa best real money casino play poker online free no sign up
buying prescription drugs in mexico online best online pharmacies in mexico mexican mail order pharmacies
best online pharmacies in mexico mexican border pharmacies shipping to usa buying prescription drugs in mexico online
reputable mexican pharmacies online purple pharmacy mexico price list mexican rx online
mexican pharmaceuticals online buying from online mexican pharmacy buying prescription drugs in mexico online
http://mexicanph.shop/# mexico pharmacies prescription drugs
reputable mexican pharmacies online
buy vardenafil pill levitra 20mg price vardenafil 10mg sale
reputable mexican pharmacies online buying prescription drugs in mexico best online pharmacies in mexico
mexican drugstore online pharmacies in mexico that ship to usa medicine in mexico pharmacies
mexico drug stores pharmacies medicine in mexico pharmacies mexican pharmacy
mexico drug stores pharmacies buying prescription drugs in mexico online mexican mail order pharmacies
mexican pharmacy mexican online pharmacies prescription drugs mexican online pharmacies prescription drugs
https://mexicanph.com/# mexican rx online
best online pharmacies in mexico
buying prescription drugs in mexico online mexican drugstore online medicine in mexico pharmacies
buying prescription drugs in mexico buying prescription drugs in mexico pharmacies in mexico that ship to usa
order pregabalin pills order pregabalin 75mg online pregabalin medication
best online pharmacies in mexico mexican mail order pharmacies mexican mail order pharmacies
п»їbest mexican online pharmacies mexico drug stores pharmacies best online pharmacies in mexico
http://mexicanph.shop/# mexican rx online
reputable mexican pharmacies online
mexican pharmaceuticals online mexican pharmaceuticals online pharmacies in mexico that ship to usa
buy cheap hydroxychloroquine order generic hydroxychloroquine 200mg purchase plaquenil online cheap
buying from online mexican pharmacy pharmacies in mexico that ship to usa pharmacies in mexico that ship to usa
medicine in mexico pharmacies mexico pharmacies prescription drugs mexican online pharmacies prescription drugs
mexico drug stores pharmacies pharmacies in mexico that ship to usa best online pharmacies in mexico
generic zithromax online
pharmacies in mexico that ship to usa buying from online mexican pharmacy mexican rx online
mexican online pharmacies prescription drugs medicine in mexico pharmacies mexican pharmaceuticals online
http://mexicanph.shop/# mexico pharmacies prescription drugs
buying from online mexican pharmacy
buy triamcinolone 4mg pills buy aristocort no prescription generic aristocort 10mg
medication from mexico pharmacy medicine in mexico pharmacies mexico drug stores pharmacies
mexico drug stores pharmacies mexico drug stores pharmacies buying prescription drugs in mexico online
mexican border pharmacies shipping to usa purple pharmacy mexico price list buying prescription drugs in mexico online
mexican online pharmacies prescription drugs mexican drugstore online п»їbest mexican online pharmacies
best mexican online pharmacies mexican pharmaceuticals online pharmacies in mexico that ship to usa
mexican drugstore online buying prescription drugs in mexico mexico drug stores pharmacies
mexican mail order pharmacies mexican online pharmacies prescription drugs medication from mexico pharmacy
http://mexicanph.com/# medicine in mexico pharmacies
mexico drug stores pharmacies
mexico drug stores pharmacies buying prescription drugs in mexico online mexican mail order pharmacies
tadalafil 20mg tadalafil 10mg oral buy cialis 5mg without prescription
mexico pharmacy buying from online mexican pharmacy mexican pharmacy
mexican pharmacy pharmacies in mexico that ship to usa mexican online pharmacies prescription drugs
medication from mexico pharmacy buying from online mexican pharmacy reputable mexican pharmacies online
mexican border pharmacies shipping to usa mexican drugstore online mexican rx online
buying prescription drugs in mexico medicine in mexico pharmacies mexican pharmaceuticals online
buying prescription drugs in mexico mexican drugstore online п»їbest mexican online pharmacies
medicine in mexico pharmacies mexican mail order pharmacies medication from mexico pharmacy
buy clarinex pills for sale clarinex 5mg us clarinex generic
mexican pharmaceuticals online buying from online mexican pharmacy mexican rx online
mexico pharmacies prescription drugs pharmacies in mexico that ship to usa mexico pharmacies prescription drugs
pharmacies in mexico that ship to usa medicine in mexico pharmacies mexico pharmacies prescription drugs
http://mexicanph.shop/# buying from online mexican pharmacy
medicine in mexico pharmacies
mexican online pharmacies prescription drugs purple pharmacy mexico price list mexican mail order pharmacies
buying prescription drugs in mexico reputable mexican pharmacies online buying prescription drugs in mexico
mexican border pharmacies shipping to usa mexican drugstore online mexico pharmacy
mexican pharmacy mexican border pharmacies shipping to usa buying prescription drugs in mexico
mexican border pharmacies shipping to usa pharmacies in mexico that ship to usa mexico pharmacies prescription drugs
mexico drug stores pharmacies mexico drug stores pharmacies buying prescription drugs in mexico online
buying from online mexican pharmacy mexico pharmacy mexican drugstore online
order cenforce 50mg cenforce oral cenforce 100mg usa
mexican online pharmacies prescription drugs best online pharmacies in mexico medicine in mexico pharmacies
mexican pharmacy mexican mail order pharmacies mexican mail order pharmacies
mexican border pharmacies shipping to usa best mexican online pharmacies mexican border pharmacies shipping to usa
buying from online mexican pharmacy mexico pharmacies prescription drugs mexico pharmacy
mexico drug stores pharmacies mexican mail order pharmacies mexico pharmacies prescription drugs
mexico pharmacy mexico pharmacies prescription drugs medication from mexico pharmacy
medicine in mexico pharmacies mexican mail order pharmacies mexican drugstore online
purchase loratadine generic buy loratadine buy generic loratadine online
mexico pharmacy mexico pharmacy mexico drug stores pharmacies
reputable mexican pharmacies online buying prescription drugs in mexico online mexican mail order pharmacies
purple pharmacy mexico price list best mexican online pharmacies mexico drug stores pharmacies
purple pharmacy mexico price list mexican border pharmacies shipping to usa best mexican online pharmacies
mexican rx online buying prescription drugs in mexico п»їbest mexican online pharmacies
buying prescription drugs in mexico online pharmacies in mexico that ship to usa mexican drugstore online
mexico drug stores pharmacies mexico drug stores pharmacies buying prescription drugs in mexico
http://mexicanph.com/# mexican mail order pharmacies
mexico drug stores pharmacies
п»їbest mexican online pharmacies reputable mexican pharmacies online medicine in mexico pharmacies
mexico drug stores pharmacies mexican rx online mexican rx online
mexican drugstore online mexico pharmacies prescription drugs mexico pharmacies prescription drugs
chloroquine order online aralen canada order chloroquine 250mg for sale
purple pharmacy mexico price list medicine in mexico pharmacies buying prescription drugs in mexico
mexican mail order pharmacies reputable mexican pharmacies online mexican pharmacy
mexico drug stores pharmacies mexican pharmaceuticals online medication from mexico pharmacy
mexican pharmacy mexico drug stores pharmacies medicine in mexico pharmacies
http://furosemide.guru/# lasix 40mg
buy lisinopril online usa zestril drug lisinopril 20 mg 12.5 mg
http://amoxil.cheap/# amoxicillin online pharmacy
ivermectin cost australia: ivermectin 3 mg tablet dosage – stromectol ivermectin 3 mg
https://buyprednisone.store/# 20mg prednisone
order priligy 90mg pill misoprostol usa misoprostol cheap
best time to take metformin for weight loss
prednisone 2.5 mg prednisone 10 mg prednisone canada
zestril 5 mg: lisinopril hctz – lisinopril generic cost
https://lisinopril.top/# buy lisinopril mexico
https://lisinopril.top/# lisinopril 10 india
https://lisinopril.top/# order lisinopril online
stromectol 12mg online: stromectol price in india – ivermectin 0.1 uk
furosemide 40mg generic lasix lasix medication
https://furosemide.guru/# lasix 100 mg
buy glucophage online cheap glucophage where to buy buy glycomet 500mg pills
lasix 20 mg: Over The Counter Lasix – lasix uses
medicine amoxicillin 500mg amoxicillin cost australia amoxicillin capsules 250mg
http://lisinopril.top/# lisinopril 20mg discount
lasix 100 mg tablet: furosemida 40 mg – furosemide 40 mg
https://furosemide.guru/# lasix pills
order glucophage 1000mg pills order glucophage 1000mg pill buy metformin 500mg online cheap
https://amoxil.cheap/# amoxicillin without a doctors prescription
amoxicillin online purchase amoxicillin 500mg price canada amoxicillin 500 mg brand name
https://buyprednisone.store/# prednisone 5 mg tablet price
zestoretic 10 mg: zestril 2.5 mg tablets – zestril tab 10mg
http://furosemide.guru/# furosemide 40mg
orlistat 120mg without prescription oral orlistat 120mg brand diltiazem
lisinopril 20 mg canadian: lisinopril 5mg tabs – lisinopril 20 mg prices
stromectol in canada cost of ivermectin ivermectin cream 5%
http://buyprednisone.store/# prednisone 50 mg canada
benicar lisinopril: lisinopril without rx – lisinopril 2.5
http://furosemide.guru/# lasix medication
lasix online Buy Lasix lasix dosage
http://stromectol.fun/# ivermectin new zealand
buying lisinopril in mexico: cost of lisinopril 40mg – lisinopril 5 mg prices
http://lisinopril.top/# zestril lisinopril
http://furosemide.guru/# furosemide 100 mg
buy cheap amoxicillin online: where to buy amoxicillin pharmacy – amoxicillin 500mg capsules uk
lisinopril for kidney protection
lisinopril 5mg pill lisinopril 20 mg tabs lisinopril 20 mg tab price
lipitor 20mg sale lipitor 40mg for sale generic atorvastatin 40mg
http://furosemide.guru/# lasix generic
stromectol 12mg: ivermectin 1%cream – buy ivermectin stromectol
http://furosemide.guru/# lasix uses
lisinopril price uk: lisinopril 20 mg over the counter – cheapest price for lisinopril india
prednisone 10mg cost no prescription online prednisone can i purchase prednisone without a prescription
https://furosemide.guru/# lasix pills
http://furosemide.guru/# furosemide 100 mg
potassium level monitoring for patients on furosemide
furosemida: Buy Lasix – lasix tablet
buy zovirax online buy zyloprim 100mg online cheap buy zyloprim 100mg sale
http://lisinopril.top/# lisinopril 419
amoxicillin 500mg buy online canada amoxicillin 500mg buy online uk how to buy amoxicillin online
https://buyprednisone.store/# drug prices prednisone
cost of amoxicillin 875 mg: where can you buy amoxicillin over the counter – amoxicillin 500 mg
buy amlodipine 10mg amlodipine generic order norvasc 10mg online cheap
http://buyprednisone.store/# can you buy prednisone over the counter in mexico
lasix 100 mg tablet: Buy Lasix – lasix furosemide 40 mg
flagyl for diarrhea dosage
furosemide Over The Counter Lasix lasix medication
http://lisinopril.top/# zestril price
http://amoxil.cheap/# amoxicillin capsules 250mg
amoxicillin 500 mg online: where can i buy amoxicillin over the counter – where can you buy amoxicillin over the counter
should i take zoloft in the morning or at night
http://amoxil.cheap/# amoxicillin for sale online
lasix 100 mg tablet Buy Lasix No Prescription furosemida 40 mg
https://amoxil.cheap/# amoxacillian without a percription
website: 90 lisinopril – zestril 5mg
https://buyprednisone.store/# prednisone 300mg
amoxicillin 500 mg tablet: buy amoxicillin 500mg online – buy amoxicillin online uk
lasix 100 mg tablet Buy Furosemide furosemide 100 mg
http://buyprednisone.store/# buy prednisone canadian pharmacy
can you take ibuprofen with gabapentin
http://furosemide.guru/# furosemide
prednisone 20 mg prices: prednisone 60 mg – prednisone pill
https://amoxil.cheap/# amoxicillin 500 capsule
rosuvastatin online buy ezetimibe pills for sale cost zetia
where can i buy lisinopril lisinopril 10mg tablet order lisinopril 2.5mg online
https://buyprednisone.store/# prednisone cream brand name
amoxicillin over the counter in canada amoxicillin capsules 250mg amoxicillin cost australia
lisinopril 40 mg no prescription: lisinopril online uk – otc lisinopril
http://stromectol.fun/# ivermectin 2%
lisinopril 2.5 mg for sale: http://lisinoprilpharm.com/%5Dlisinopril – lisinopril 40 mg purchase
does lasix affect kidney function
https://amoxil.cheap/# amoxil pharmacy
stromectol tablets buy online ivermectin usa ivermectin buy online
http://stromectol.fun/# stromectol 12mg online
amoxicillin 500mg capsules uk: amoxicillin 500mg over the counter – amoxicillin 250 mg capsule
http://stromectol.fun/# stromectol buy uk
http://lisinopril.top/# lisinopril uk
lisinopril 5 mg daily lisinopril 20 mg cost zestoretic 20
cortisol prednisone: buying prednisone on line – buy prednisone 1 mg mexico
http://amoxil.cheap/# antibiotic amoxicillin
glucophage availability
buy domperidone generic buy domperidone 10mg online order sumycin online cheap
omeprazole 20mg pill omeprazole 20mg canada order omeprazole 10mg online
zithromax tablets
cephalexinжЇд»Ђд№€иЌЇ
best online pharmacy india india online pharmacy Online medicine home delivery
https://indianph.com/# pharmacy website india
reputable indian pharmacies
indian pharmacy online online shopping pharmacy india top 10 pharmacies in india
http://indianph.xyz/# india pharmacy mail order
reputable indian pharmacies
https://indianph.com/# online pharmacy india
world pharmacy india
п»їlegitimate online pharmacies india cheapest online pharmacy india best online pharmacy india
cyclobenzaprine without prescription buy cyclobenzaprine 15mg generic ozobax order
http://indianph.com/# mail order pharmacy india
gabapentin 500 mg
http://indianph.com/# legitimate online pharmacies india
reputable indian online pharmacy
metoprolol uk lopressor for sale online metoprolol over the counter
https://indianph.com/# buy medicines online in india
india online pharmacy
http://indianph.xyz/# india pharmacy mail order
indian pharmacy
best online pharmacy india india pharmacy mail order world pharmacy india
https://indianph.com/# legitimate online pharmacies india
mail order pharmacy india
side effects of amoxicillin in adults
escitalopram doses
https://indianph.com/# best online pharmacy india
indian pharmacy paypal
toradol 10mg cost buy toradol 10mg for sale buy colcrys generic
who should take tamoxifen: tamoxifen benefits – tamoxifen rash pictures
http://diflucan.pro/# can you buy diflucan over the counter in canada
buy ciprofloxacin over the counter buy cipro cheap ciprofloxacin generic price
https://cytotec24.shop/# buy cytotec
cephalexin uti
atenolol 100mg ca buy tenormin sale generic tenormin 100mg
cytotec buy online usa: purchase cytotec – Misoprostol 200 mg buy online
https://nolvadex.guru/# how to lose weight on tamoxifen
tamoxifen dose tamoxifen and osteoporosis buy tamoxifen
https://nolvadex.guru/# nolvadex for sale amazon
tamoxifen pill: where to buy nolvadex – tamoxifen
https://cipro.guru/# cipro generic
doxycycline 200 mg: doxy – order doxycycline online
diflucan 150 tablet diflucan 150 mg buy online diflucan rx price
http://diflucan.pro/# diflucan otc canada
https://diflucan.pro/# diflucan canada
ciprofloxacin for uti dosage
http://cytotec24.com/# buy cytotec
nolvadex 10mg: tamoxifen medication – tamoxifen and grapefruit
where to get nolvadex where to buy nolvadex tamoxifen and osteoporosis
http://doxycycline.auction/# buy doxycycline online
where can i buy cipro online: ciprofloxacin 500 mg tablet price – antibiotics cipro
https://cipro.guru/# buy cipro cheap
pct nolvadex tamoxifen alternatives premenopausal tamoxifen brand name
https://nolvadex.guru/# tamoxifen 20 mg
https://doxycycline.auction/# generic doxycycline
cost of medrol buy generic depo-medrol purchase methylprednisolone
https://cipro.guru/# ciprofloxacin 500 mg tablet price
doxycycline 50mg doxycycline hyclate buy doxycycline online uk
https://diflucan.pro/# diflucan 200 mg over the counter
http://nolvadex.guru/# nolvadex pills
http://cytotec24.com/# Misoprostol 200 mg buy online
https://doxycycline.auction/# doxycycline without prescription
https://cytotec24.shop/# buy cytotec over the counter
http://cytotec24.shop/# cytotec online
eva elfie izle: eva elfie video – eva elfie modeli
https://evaelfie.pro/# eva elfie filmleri
cost of tamoxifen tamoxifen buy aromatase inhibitor tamoxifen
https://abelladanger.online/# abella danger filmleri
Angela White: Angela White video – Angela White izle
brand inderal 20mg clopidogrel without prescription order plavix 150mg online
http://evaelfie.pro/# eva elfie video
http://abelladanger.online/# abella danger filmleri
my father essay writing help me with my essay write my cover letter for me
Angela White filmleri: Angela Beyaz modeli – Angela White filmleri
http://evaelfie.pro/# eva elfie izle
https://evaelfie.pro/# eva elfie video
https://evaelfie.pro/# eva elfie filmleri
https://evaelfie.pro/# eva elfie
Sweetie Fox: sweety fox – Sweetie Fox video
https://sweetiefox.online/# sweety fox
https://evaelfie.pro/# eva elfie video
Angela Beyaz modeli: Abella Danger – Abella Danger
http://angelawhite.pro/# Angela White video
https://lanarhoades.fun/# lana rhoades modeli
http://lanarhoades.fun/# lana rhoades
http://abelladanger.online/# abella danger video
eva elfie izle: eva elfie filmleri – eva elfie
http://lanarhoades.fun/# lana rhoades
methotrexate pills purchase medex order coumadin for sale
http://evaelfie.pro/# eva elfie video
eva elfie modeli: eva elfie filmleri – eva elfie
http://evaelfie.pro/# eva elfie video
https://abelladanger.online/# Abella Danger
https://abelladanger.online/# abella danger filmleri
http://sweetiefox.online/# Sweetie Fox izle
cephalexin warnings
Sweetie Fox izle: Sweetie Fox izle – swetie fox
bactrim vs cipro
http://abelladanger.online/# Abella Danger
https://lanarhoades.fun/# lana rhoades
Angela Beyaz modeli: Abella Danger – Abella Danger
https://sweetiefox.online/# Sweetie Fox izle
https://sweetiefox.online/# Sweetie Fox
lana rhodes: lana rhoades – lana rhoades modeli
https://evaelfie.pro/# eva elfie izle
mobic 15mg without prescription celecoxib online purchase celecoxib online
http://evaelfie.pro/# eva elfie filmleri
http://angelawhite.pro/# Angela White izle
Angela White filmleri: abella danger izle – abella danger video
buy metoclopramide 20mg online cheap order generic cozaar order cozaar 25mg online cheap
https://sweetiefox.online/# sweeti fox
https://evaelfie.pro/# eva elfie filmleri
can bactrim cause a yeast infection
https://abelladanger.online/# abella danger filmleri
http://angelawhite.pro/# ?????? ????
Angela White filmleri: Angela White video – Angela White filmleri
http://abelladanger.online/# Abella Danger
https://lanarhoades.fun/# lana rhoades izle
lana rhoades modeli: lana rhoades video – lana rhoades
https://evaelfie.pro/# eva elfie video
http://angelawhite.pro/# Angela White filmleri
Join the Telegram community today and experience a messaging app that’s secure, fast, and feature-rich. Download now for free!今天就加入Telegram社区,体验一款安全、快速且功能丰富的消息应用。现在免费下载!https://www.telegramjq.com
i20w9ugy6a
https://lanarhoades.pro/# lana rhoades boyfriend
eva elfie videos: eva elfie hot – eva elfie videos
nexium ca purchase nexium pills topiramate over the counter
sweetie fox video: fox sweetie – sweetie fox
tamsulosin price celecoxib 100mg over the counter buy celebrex 100mg generic
https://sweetiefox.pro/# sweetie fox cosplay
100% free dating sites: http://lanarhoades.pro/# lana rhoades unleashed
http://evaelfie.site/# eva elfie videos
lana rhoades unleashed: lana rhoades boyfriend – lana rhoades
Your point of view caught my eye and was very interesting. Thanks. I have a question for you.
https://lanarhoades.pro/# lana rhoades hot
ph sweetie fox: ph sweetie fox – sweetie fox
lana rhoades boyfriend: lana rhoades hot – lana rhoades boyfriend
christian dating sites: http://sweetiefox.pro/# sweetie fox
http://lanarhoades.pro/# lana rhoades
mia malkova: mia malkova hd – mia malkova
neurontin 300mg uses
lana rhoades videos: lana rhoades full video – lana rhoades full video
lana rhoades: lana rhoades pics – lana rhoades boyfriend
http://evaelfie.site/# eva elfie new video
dating for singles: http://lanarhoades.pro/# lana rhoades solo
mia malkova: mia malkova only fans – mia malkova new video
escitalopram qt prolongation
http://sweetiefox.pro/# sweetie fox full
eva elfie full video: eva elfie hd – eva elfie videos
eva elfie new videos: eva elfie new video – eva elfie hot
http://lanarhoades.pro/# lana rhoades pics
which online dating site is best: http://sweetiefox.pro/# sweetie fox new
lana rhoades full video: lana rhoades full video – lana rhoades videos
cost ondansetron 4mg buy aldactone 100mg spironolactone 100mg cost
http://lanarhoades.pro/# lana rhoades pics
mia malkova new video: mia malkova only fans – mia malkova latest
sweetie fox cosplay: sweetie fox full video – sweetie fox video
order imitrex 50mg pill order levaquin for sale levaquin 500mg canada
free dating ads: http://miamalkova.life/# mia malkova hd
mia malkova latest: mia malkova full video – mia malkova movie
https://sweetiefox.pro/# sweetie fox cosplay
mia malkova only fans: mia malkova full video – mia malkova hd
http://sweetiefox.pro/# sweetie fox cosplay
eva elfie photo: eva elfie new videos – eva elfie photo
lana rhoades full video: lana rhoades hot – lana rhoades unleashed
website dating service: http://evaelfie.site/# eva elfie photo
buy zocor 20mg generic order valtrex online valtrex brand
brand avodart buy cheap generic ranitidine order zantac for sale
http://evaelfie.site/# eva elfie photo
mia malkova new video: mia malkova photos – mia malkova photos
http://sweetiefox.pro/# ph sweetie fox
http://aviatormalawi.online/# aviator bet malawi
aviator bet: play aviator – aviator login
aviator: pin up aviator – aviator hilesi
https://pinupcassino.pro/# cassino pin up
half life of depakote
pin up aviator: jogar aviator online – aviator game
escitalopram vs citalopram
http://aviatoroyunu.pro/# aviator
aviator login: aviator game online – aviator
buy acillin generic buy vibra-tabs online cheap cheap amoxil without prescription
aviator oyna: aviator bahis – aviator oyna
does citalopram show up on a drug test
cassino pin up: pin-up casino login – pin-up casino login
https://aviatorjogar.online/# aviator pin up
proscar ca fluconazole 200mg pills buy forcan online
jogar aviator online: pin up aviator – aviator betano
long term use of ddavp
http://jogodeaposta.fun/# melhor jogo de aposta
aviator pin up casino: pin-up cassino – pin-up casino
estrela bet aviator: aviator betano – aviator jogo
cozaar alternative
aviator malawi: aviator bet malawi login – aviator game
aviator sportybet ghana: aviator – aviator
aviator game: aviator game bet – aviator sportybet ghana
aviator game online: aviator – aviator betting game
aviator bet malawi login: aviator bet malawi – aviator betting game
aviator game: aviator bet malawi login – aviator game
aviator jogo: aviator game – aviator jogo
como jogar aviator: jogar aviator – aviator online
zithromax 500 – https://azithromycin.pro/is-zithromax-a-penicillin.html zithromax 500 without prescription
ciprofloxacin 500mg pills – cephalexin 250mg pill augmentin generic
como jogar aviator em moçambique: aviator moçambique – aviator mz
can you buy zithromax over the counter in canada: zithromax allergic reaction zithromax 500 price
aviator online: jogar aviator – aviator
aviator bet: aviator malawi – aviator malawi
order zithromax without prescription – https://azithromycin.pro/zithromax-price.html buy zithromax online fast shipping
aviator game bet: aviator sportybet ghana – aviator game online
ciprofloxacin 500mg drug – augmentin 375mg usa buy augmentin pills for sale
purple pharmacy mexico price list: mexican mail order pharmacies – mexican mail order pharmacies mexicanpharm.shop
indian pharmacy paypal india pharmacy mail order pharmacy india indianpharm.store
pharmacy in canada: Canada pharmacy – pet meds without vet prescription canada canadianpharm.store
purple pharmacy mexico price list: Mexico pharmacy online – medicine in mexico pharmacies mexicanpharm.shop
depakote hair loss
mexican pharmaceuticals online: order online from a Mexican pharmacy – mexican border pharmacies shipping to usa mexicanpharm.shop
medication from mexico pharmacy mexican pharmacy medication from mexico pharmacy mexicanpharm.shop
http://mexicanpharm24.com/# reputable mexican pharmacies online mexicanpharm.shop
https://indianpharm24.shop/# reputable indian pharmacies indianpharm.store
reputable indian online pharmacy: Best Indian pharmacy – world pharmacy india indianpharm.store
http://mexicanpharm24.shop/# mexico pharmacy mexicanpharm.shop
medicine in mexico pharmacies: Mexico pharmacy online – mexican border pharmacies shipping to usa mexicanpharm.shop
http://indianpharm24.com/# top 10 online pharmacy in india indianpharm.store
can you od on citalopram
https://mexicanpharm24.com/# mexican rx online mexicanpharm.shop
https://indianpharm24.com/# pharmacy website india indianpharm.store
77 canadian pharmacy: International Pharmacy delivery – canadian pharmacy no scripts canadianpharm.store
http://mexicanpharm24.com/# buying prescription drugs in mexico online mexicanpharm.shop
mexico pharmacies prescription drugs Medicines Mexico reputable mexican pharmacies online mexicanpharm.shop
https://mexicanpharm24.shop/# mexican online pharmacies prescription drugs mexicanpharm.shop
http://indianpharm24.shop/# online shopping pharmacy india indianpharm.store
ddavp nasal spray price
cozaar over 65 years old
best canadian online pharmacy: Canada pharmacy – best canadian pharmacy to order from canadianpharm.store
canadian pharmacy drugs online: Certified Canadian pharmacies – canadian online pharmacy canadianpharm.store
http://indianpharm24.shop/# online shopping pharmacy india indianpharm.store
https://canadianpharmlk.com/# ordering drugs from canada canadianpharm.store
https://indianpharm24.com/# india pharmacy indianpharm.store
ciprofloxacin 500mg generic – ciplox pill order generic erythromycin 500mg
reputable mexican pharmacies online: Medicines Mexico – mexican drugstore online mexicanpharm.shop
https://canadianpharmlk.com/# online canadian pharmacy review canadianpharm.store
http://mexicanpharm24.shop/# medicine in mexico pharmacies mexicanpharm.shop
pharmacy website india Online India pharmacy top 10 pharmacies in india indianpharm.store
order flagyl 200mg generic – buy metronidazole sale azithromycin ca
http://canadianpharmlk.shop/# buying drugs from canada canadianpharm.store
india pharmacy mail order: Online medicine home delivery – online shopping pharmacy india indianpharm.store
india pharmacy mail order: online shopping pharmacy india – online shopping pharmacy india indianpharm.store
http://mexicanpharm24.shop/# п»їbest mexican online pharmacies mexicanpharm.shop
http://mexicanpharm24.shop/# buying from online mexican pharmacy mexicanpharm.shop
Cibus, onus et virga asino — Ослу нужны пища, груз и кнут.
how to buy clomid no prescription: how to get generic clomid price – cost of generic clomid without insurance
can i get generic clomid: clomid over the counter – generic clomid
prednisone coupon: prednisone pack – 400 mg prednisone
http://clomidst.pro/# clomid buy
amoxicillin online canada: amoxil generic – amoxicillin 500mg capsules price
amoxicillin 800 mg price: order amoxicillin online – amoxicillin without prescription
buying generic clomid without insurance can i buy generic clomid online can i buy cheap clomid without dr prescription
https://amoxilst.pro/# amoxicillin 500mg for sale uk
prednisone 10mg online: india buy prednisone online – prednisone 5093
can you buy cheap clomid: is 100mg of clomid a high dose – get cheap clomid without dr prescription
how does diclofenac work
where can i get cheap clomid pills: how does clomid work – get generic clomid without dr prescription
https://amoxilst.pro/# amoxicillin 500mg no prescription
buy prednisone 50 mg: can you drink alcohol with prednisone – prednisone 2 mg
100 mg prednisone daily: prednisone dogs – prednisone 10mg tabs
https://amoxilst.pro/# purchase amoxicillin online
where can i get amoxicillin amoxicillin 500 mg tablet buying amoxicillin in mexico
amoxicillin canada price: what is amoxicillin-clav 875-125 mg used for – amoxicillin no prescription
amoxil pharmacy: amoxicillin over counter – can we buy amoxcillin 500mg on ebay without prescription
ivermectin 6 mg without prescription – aczone uk buy tetracycline pills
prednisone: prednisone 40 mg – can you buy prednisone without a prescription
http://amoxilst.pro/# amoxicillin 500 tablet
prednisone 5 mg tablet rx: what is prednisone used for – prednisone 30 mg
what is the drug ezetimibe used for
can you buy amoxicillin over the counter: amoxicillin for tooth infection – amoxicillin no prescription
where can i get amoxicillin 500 mg: buy amoxil – amoxicillin 500 mg cost
buy valtrex sale – buy diltiazem 180mg sale order zovirax 400mg online cheap
http://clomidst.pro/# get clomid tablets
cost of clomid online: cost generic clomid without dr prescription – can i get generic clomid prices
augmentin for std
prednisone for dogs: does prednisone raise blood pressure – prednisone 30
diltiazem 2
how to buy cheap clomid without prescription can you buy generic clomid for sale can you get generic clomid tablets
what is diltiazem
online erectile dysfunction prescription online ed prescription low cost ed pills
buying prescription drugs from canada: mexican pharmacy online – best online pharmacy no prescription
how to order prescription drugs from canada: prescription from canada – cheap prescription medication online
https://pharmnoprescription.pro/# buy medication online with prescription
ed medicine online: low cost ed medication – low cost ed pills
us pharmacy no prescription: best online pharmacy – online pharmacy no prescription
best ed pills online: ed online meds – cheapest erectile dysfunction pills
flexeril pregnancy
https://pharmnoprescription.pro/# buy drugs without prescription
mail order prescription drugs from canada: mexico pharmacy online – canada drugs coupon code
where to buy ed pills ed medications online best ed meds online
cheapest prescription pharmacy: canada pharmacy online – no prescription needed pharmacy
buy ed medication online: buying ed pills online – cheap ed treatment
http://edpills.guru/# cheapest ed online
cheapest ed medication: where to buy ed pills – cheapest online ed treatment
canadian pharmacy coupon: online pharmacy india – overseas pharmacy no prescription
http://onlinepharmacy.cheap/# non prescription medicine pharmacy
canadian online pharmacy no prescription: online pharmacy delivery – online pharmacy discount code
buy acillin cheap penicillin uk buy generic amoxil over the counter
cheap ed meds: where to buy erectile dysfunction pills – best ed medication online
effexor xr max dose
low cost ed meds: online ed medication – buy ed medication online
metronidazole 200mg drug – azithromycin pills order azithromycin
http://edpills.guru/# erectile dysfunction online
no prescription pharmacy: how to order prescription drugs from canada – buying prescription drugs online from canada
buy medications online without prescription: order prescription from canada – canadian prescriptions in usa
low cost ed pills ed doctor online cheapest ed online
buy ed pills: ed pills cheap – buy ed pills
generic flomax walmart
http://mexicanpharm.online/# medicine in mexico pharmacies
canada pharmacies online prescriptions: discount prescription drugs canada – mexican pharmacy no prescription
http://indianpharm.shop/# п»їlegitimate online pharmacies india
canadian compounding pharmacy: canada cloud pharmacy – ordering drugs from canada
canadian pharmacy online no prescription needed online drugstore no prescription overseas online pharmacy-no prescription
https://pharmacynoprescription.pro/# legitimate online pharmacy no prescription
aripiprazole lauroxil alkermes
http://canadianpharm.guru/# onlinecanadianpharmacy 24
reputable mexican pharmacies online: medicine in mexico pharmacies – best online pharmacies in mexico
reputable indian online pharmacy: world pharmacy india – Online medicine order
https://mexicanpharm.online/# mexican mail order pharmacies
india pharmacy mail order: mail order pharmacy india – top online pharmacy india
india pharmacy mail order: best india pharmacy – Online medicine home delivery
canadian pharmacy prices best canadian online pharmacy best canadian online pharmacy
http://mexicanpharm.online/# mexico pharmacies prescription drugs
online shopping pharmacy india: world pharmacy india – Online medicine home delivery
purchase lasix – purchase prazosin online captopril order
allopurinol high
best online pharmacies in mexico: medication from mexico pharmacy – mexico drug stores pharmacies
best online pharmacy india: cheapest online pharmacy india – top 10 pharmacies in india
http://pharmacynoprescription.pro/# canada drugs no prescription
ordering drugs from canada: canadian discount pharmacy – safe reliable canadian pharmacy
escrow pharmacy canada: best online canadian pharmacy – pharmacy rx world canada
https://canadianpharm.guru/# thecanadianpharmacy
mexico pharmacies prescription drugs: mexican rx online – mexican mail order pharmacies
online medication without prescription canadian pharmacy prescription canada prescription drugs online
https://canadianpharm.guru/# canada drug pharmacy
cheapest online pharmacy india: top 10 online pharmacy in india – top 10 pharmacies in india
india pharmacy mail order: india pharmacy – indian pharmacy online
http://pharmacynoprescription.pro/# canadian and international prescription service
no prescription online pharmacy: no prescription medicines – no prescription needed
canadian pharmacy mall: canadian mail order pharmacy – canadian pharmacy tampa
https://mexicanpharm.online/# mexican pharmacy
canadian pharmacy in canada: canadian mail order pharmacy – canadian family pharmacy
pharmacies in mexico that ship to usa: best online pharmacies in mexico – buying prescription drugs in mexico online
canadian prescriptions in usa best website to buy prescription drugs online drugs no prescription
https://mexicanpharm.online/# buying prescription drugs in mexico
india pharmacy: best online pharmacy india – indian pharmacy online
indian pharmacy paypal: reputable indian online pharmacy – indian pharmacy
https://canadianpharm.guru/# canadian family pharmacy
does aleve have aspirin
best canadian online pharmacy: canadian 24 hour pharmacy – canadian discount pharmacy
http://canadianpharm.guru/# my canadian pharmacy reviews
buy metformin 500mg – glucophage 500mg uk order lincomycin pill
canada drugs reviews: canadian valley pharmacy – canadian king pharmacy
buy prescription drugs without a prescription: pharmacies without prescriptions – quality prescription drugs canada
https://mexicanpharm.online/# mexico drug stores pharmacies
northwest canadian pharmacy: canadian pharmacy scam – canadian pharmacy oxycodone
buy prescription online: buy prescription drugs online without doctor – can you buy prescription drugs in canada
reputable indian online pharmacy indian pharmacy online top 10 pharmacies in india
rate canadian pharmacies: pharmacies in canada that ship to the us – canadian mail order pharmacy
http://mexicanpharm.online/# mexican border pharmacies shipping to usa
http://indianpharm.shop/# п»їlegitimate online pharmacies india
canada pharmacy online: canadian drugstore online – canadian pharmacy cheap
baclofen overdose
buying drugs without prescription: canada mail order prescriptions – canada mail order prescriptions
https://pharmacynoprescription.pro/# online drugs no prescription
best online pharmacies in mexico: buying from online mexican pharmacy – mexican online pharmacies prescription drugs
canada prescription: no prescription medicine – how to order prescription drugs from canada
https://pharmacynoprescription.pro/# how to order prescription drugs from canada
pharmacy website india: best india pharmacy – pharmacy website india
online pharmacy canada: trusted canadian pharmacy – my canadian pharmacy
buying drugs online no prescription buy drugs without prescription pharmacy online no prescription
https://canadianpharm.guru/# legitimate canadian pharmacies
india pharmacy: online shopping pharmacy india – cheapest online pharmacy india
https://pharmacynoprescription.pro/# no prescription needed
where can i buy cheap retrovir – buy zyloprim 300mg for sale order allopurinol generic
is celebrex safe for long term use
pharmacies in mexico that ship to usa: mexico drug stores pharmacies – mexican rx online
77 canadian pharmacy: canadian pharmacy ratings – canada rx pharmacy
sweet bonanza taktik: sweet bonanza indir – sweet bonanza mostbet
https://gatesofolympus.auction/# gates of olympus giris
https://sweetbonanza.bid/# sweet bonanza demo turkce
pin up aviator: pin up casino indir – pin-up casino
https://sweetbonanza.bid/# slot oyunlari
en guvenilir slot siteleri: en iyi slot siteleri 2024 – slot casino siteleri
https://aviatoroyna.bid/# uçak oyunu bahis aviator
augmentin for kidney infection
oral clozaril – buy accupril 10 mg generic pepcid 20mg sale
sweet bonanza kazanc: sweet bonanza free spin demo – sweet bonanza slot demo
https://aviatoroyna.bid/# aviator sinyal hilesi ücretsiz
anxiety medication bupropion
https://pinupgiris.fun/# pin-up casino giris
http://slotsiteleri.guru/# deneme veren slot siteleri
pin-up bonanza: pin up aviator – pin-up giris
https://sweetbonanza.bid/# sweet bonanza kazanç
guncel sweet bonanza: sweet bonanza mostbet – sweet bonanza kazanma saatleri
https://pinupgiris.fun/# pin-up bonanza
ashwagandha gaba
https://gatesofolympus.auction/# gates of olympus demo
https://aviatoroyna.bid/# aviator oyna 100 tl
gates of olympus oyna demo: gates of olympus s?rlar? – gates of olympus oyna ucretsiz
http://sweetbonanza.bid/# slot oyunlari
sweet bonanza guncel: sweet bonanza hilesi – sweet bonanza
sweet bonanza demo turkce: sweet bonanza nas?l oynan?r – sweet bonanza mostbet
https://slotsiteleri.guru/# slot casino siteleri
http://sweetbonanza.bid/# sweet bonanza mostbet
sweet bonanza demo: sweet bonanza yasal site – sweet bonanza bahis
celecoxib drug interactions
https://pinupgiris.fun/# pin up indir
http://pinupgiris.fun/# pin-up casino indir
sweet bonanza 90 tl: sweet bonanza bahis – sweet bonanza siteleri
https://slotsiteleri.guru/# slot siteleri 2024
pin up casino giris: pin-up casino – pin up aviator
https://slotsiteleri.guru/# deneme bonusu veren slot siteleri
yasal slot siteleri: deneme bonusu veren siteler – deneme veren slot siteleri
quetiapine 100mg tablet – purchase zoloft generic order eskalith sale
http://sweetbonanza.bid/# sweet bonanza hilesi
en yeni slot siteleri: deneme bonusu veren slot siteleri – canl? slot siteleri
celexa drowsiness
indian pharmacy paypal: Healthcare and medicines from India – best online pharmacy india
anyone taking buspirone
buying prescription drugs in mexico mexican pharmacy medication from mexico pharmacy
generic clomipramine 25mg – order anafranil sale sinequan 25mg generic
Online medicine order Cheapest online pharmacy online pharmacy india
https://indianpharmacy.icu/# best online pharmacy india
recommended canadian pharmacies Large Selection of Medications canadian pharmacies comparison
canadian pharmacy near me: Licensed Canadian Pharmacy – canada pharmacy online legit
pharmacy website india Healthcare and medicines from India cheapest online pharmacy india
indianpharmacy com: Generic Medicine India to USA – legitimate online pharmacies india
https://canadianpharmacy24.store/# canadian king pharmacy
mexican border pharmacies shipping to usa: cheapest mexico drugs – mexico pharmacies prescription drugs
canada drugs reviews canadian pharmacy 24 canadian pharmacy phone number
buy prescription drugs from india: top 10 online pharmacy in india – indian pharmacy paypal
canada ed drugs: pills now even cheaper – pharmacy rx world canada
world pharmacy india: Generic Medicine India to USA – indianpharmacy com
indian pharmacies safe: Generic Medicine India to USA – indian pharmacy paypal
pharmacies in mexico that ship to usa Mexican Pharmacy Online pharmacies in mexico that ship to usa
buy generic hydroxyzine 25mg – prozac cost order endep 25mg
medication from mexico pharmacy: mexico pharmacy – mexican pharmaceuticals online
india online pharmacy: Generic Medicine India to USA – reputable indian online pharmacy
canadian pharmacy ed medications canadian neighbor pharmacy escrow pharmacy canada
https://canadianpharmacy24.store/# canadian pharmacy world reviews
best online pharmacy india: Generic Medicine India to USA – best online pharmacy india
Online medicine order: Cheapest online pharmacy – reputable indian online pharmacy
reputable indian pharmacies indian pharmacy delivery п»їlegitimate online pharmacies india
pharmacy in canada: Prescription Drugs from Canada – canadian drug prices
best canadian pharmacy to order from: Certified Canadian Pharmacy – canadian drugs
onlinecanadianpharmacy canadianpharmacymeds com canadian pharmacy service
https://mexicanpharmacy.shop/# mexico drug stores pharmacies
mexican pharmaceuticals online mexican border pharmacies shipping to usa best online pharmacies in mexico
online pharmacy india: Healthcare and medicines from India – Online medicine home delivery
legit canadian online pharmacy: Licensed Canadian Pharmacy – canadian pharmacy sarasota
online pharmacy india: Cheapest online pharmacy – mail order pharmacy india
Online medicine home delivery indian pharmacy buy medicines online in india
amoxicillin 500 mg where can i buy amoxocillin amoxil pharmacy
http://clomidall.com/# where can i get generic clomid without prescription
where to buy amoxicillin over the counter: medicine amoxicillin 500mg – where can i buy amoxicillin without prec
https://zithromaxall.com/# zithromax over the counter uk
zithromax 500 tablet buy zithromax online where can i purchase zithromax online
prednisone daily: prednisone 5mg price – prednisone 1 tablet
http://zithromaxall.shop/# zithromax for sale online
http://prednisoneall.shop/# can i buy prednisone online in uk
amoxicillin 800 mg price amoxicillin medicine over the counter amoxicillin 500mg
http://zithromaxall.com/# zithromax for sale 500 mg
amoxicillin 50 mg tablets buy cheap amoxicillin amoxicillin tablet 500mg
https://prednisoneall.shop/# prednisone 10mg price in india
order augmentin 1000mg generic – acillin where to buy brand ciprofloxacin
zithromax over the counter uk: zithromax over the counter – zithromax capsules
http://clomidall.com/# order generic clomid
amoxicillin buy no prescription buy cheap amoxicillin online amoxicillin 500mg price canada
canadian pharmacy amoxicillin: buy amoxicillin – amoxicillin 500mg capsule buy online
http://zithromaxall.shop/# zithromax for sale cheap
http://prednisoneall.shop/# prednisone 475
prednisone cost 10mg can i buy prednisone from canada without a script 3000mg prednisone
http://clomidall.com/# where can i get cheap clomid price
can you buy amoxicillin over the counter canada amoxicillin tablets in india amoxicillin no prescipion
amoxil for sale online – erythromycin 250mg pills ciprofloxacin 1000mg pills
https://zithromaxall.shop/# azithromycin zithromax
zithromax online paypal: generic zithromax online paypal – generic zithromax online paypal
http://zithromaxall.com/# zithromax 500 mg lowest price drugstore online
prednisone over the counter australia prednisone 20mg prices prednisone 60 mg daily
can you buy generic clomid tablets: can i purchase generic clomid pill – how can i get generic clomid without insurance
https://prednisoneall.com/# 60 mg prednisone daily
kardashians semaglutide
where can i buy zithromax capsules zithromax antibiotic without prescription can i buy zithromax over the counter in canada
http://prednisoneall.shop/# prednisone 5mg price
http://tadalafiliq.com/# cheapest cialis
Viagra without a doctor prescription Canada Generic Viagra online Viagra online price
Kamagra 100mg: Kamagra gel – Kamagra Oral Jelly
Sildenafil Citrate Tablets 100mg: buy viagra online – Generic Viagra online
actos greek
buy cialis pill cialis best price Buy Tadalafil 10mg
kamagra: Kamagra gel – buy Kamagra
Buy Cialis online: cheapest cialis – Buy Cialis online
Kamagra 100mg price Kamagra gel Kamagra tablets
http://tadalafiliq.com/# Tadalafil Tablet
sildenafil oral jelly 100mg kamagra: Kamagra gel – super kamagra
generic sildenafil: Generic Viagra online – sildenafil over the counter
buy Viagra online: buy viagra online – Cheap Viagra 100mg
Generic Cialis without a doctor prescription Buy Cialis online Cialis without a doctor prescription
Cialis over the counter: cheapest cialis – Cheap Cialis
indikasi acarbose
Kamagra tablets Kamagra Oral Jelly Price buy kamagra online usa
kamagra: Kamagra gel – super kamagra
http://tadalafiliq.shop/# cheapest cialis
cialis for sale: Generic Tadalafil 20mg price – buy cialis pill
abilify 10 mg
https://sildenafiliq.xyz/# generic sildenafil
Buy Tadalafil 20mg: tadalafil iq – cialis for sale
Buy Tadalafil 10mg tadalafil iq Tadalafil Tablet
Sildenafil Citrate Tablets 100mg: generic ed pills – sildenafil 50 mg price
Buy Tadalafil 10mg: Buy Cialis online – Buy Tadalafil 20mg
http://kamagraiq.shop/# Kamagra 100mg
Kamagra 100mg price Kamagra gel Kamagra 100mg price
Cialis without a doctor prescription: cheapest cialis – Cialis 20mg price
http://tadalafiliq.com/# cheapest cialis
http://kamagraiq.shop/# Kamagra 100mg price
buy Kamagra: Kamagra gel – Kamagra tablets
cialis for sale: Buy Cialis online – cialis for sale
https://kamagraiq.com/# Kamagra 100mg price
Cheap Cialis: cialis best price – buy cialis pill
buy Kamagra kamagra best price Kamagra 100mg price
buy Viagra over the counter: generic ed pills – Buy Viagra online cheap
http://sildenafiliq.com/# cheapest viagra
Kamagra 100mg: kamagra best price – sildenafil oral jelly 100mg kamagra
https://tadalafiliq.com/# Tadalafil price
п»їcialis generic Generic Tadalafil 20mg price cheapest cialis
mexican border pharmacies shipping to usa Pills from Mexican Pharmacy purple pharmacy mexico price list
http://canadianpharmgrx.xyz/# canadian pharmacy oxycodone
canadian pharmacy checker: Best Canadian online pharmacy – canadapharmacyonline com
mail order pharmacy india top online pharmacy india indianpharmacy com
https://mexicanpharmgrx.shop/# best mexican online pharmacies
robaxin canada otc
mexican rx online п»їbest mexican online pharmacies medicine in mexico pharmacies
canadian pharmacy 1 internet online drugstore: Best Canadian online pharmacy – canada pharmacy reviews
http://mexicanpharmgrx.shop/# best online pharmacies in mexico
mexico drug stores pharmacies Mexico drugstore mexican rx online
http://mexicanpharmgrx.com/# buying prescription drugs in mexico online
best canadian pharmacy to order from: CIPA approved pharmacies – canadian drug pharmacy
mail order pharmacy india Healthcare and medicines from India top 10 online pharmacy in india
https://mexicanpharmgrx.com/# pharmacies in mexico that ship to usa
what is remeron
buy azithromycin 500mg pills – buy metronidazole 200mg ciplox 500 mg pill
indian pharmacy online shopping pharmacy india п»їlegitimate online pharmacies india
https://mexicanpharmgrx.shop/# reputable mexican pharmacies online
mexican drugstore online: mexican pharmacy – mexican drugstore online
http://canadianpharmgrx.com/# legitimate canadian pharmacies
buying prescription drugs in mexico online mexican pharmacy mexican online pharmacies prescription drugs
purchase cleocin for sale – cleocin for sale online buy chloramphenicol cheap
http://mexicanpharmgrx.shop/# reputable mexican pharmacies online
indian pharmacy paypal Generic Medicine India to USA reputable indian online pharmacy
mexico drug stores pharmacies: buying prescription drugs in mexico online – mexico drug stores pharmacies
side effects of protonix
http://indianpharmgrx.shop/# indian pharmacy paypal
http://canadianpharmgrx.com/# canadian mail order pharmacy
top 10 online pharmacy in india buy prescription drugs from india buy prescription drugs from india
repaglinide eg prezzo
https://mexicanpharmgrx.com/# best online pharmacies in mexico
canadian pharmacy meds review: Certified Canadian pharmacies – canadian neighbor pharmacy
https://mexicanpharmgrx.com/# pharmacies in mexico that ship to usa
vipps approved canadian online pharmacy International Pharmacy delivery canadian family pharmacy
http://canadianpharmgrx.com/# canadian pharmacy com
canada pharmacy online: Canada pharmacy – canadian pharmacy victoza
buy diflucan online usa: diflucan 200mg – diflucan 150 mg over the counter
doxycycline generic doxycycline 100mg doxycycline hyclate
cipro generic: cipro pharmacy – ciprofloxacin
tamoxifen alternatives premenopausal alternative to tamoxifen nolvadex vs clomid
buy cytotec online fast delivery: Misoprostol 200 mg buy online – buy cytotec pills
https://ciprofloxacin.guru/# antibiotics cipro
buy cytotec: Misoprostol 200 mg buy online – Abortion pills online
how much is over the counter diflucan diflucan 150 mg tablet price diflucan candida
diflucan otc where to buy: diflucan online cheap without a prescription – price of diflucan in south africa
where can i get doxycycline: how to buy doxycycline online – doxycycline vibramycin
buy doxycycline monohydrate doxycycline order online doxycycline hyc
doxycycline 150 mg: buy doxycycline online – doxycycline monohydrate
buy cipro online canada ciprofloxacin over the counter buy ciprofloxacin over the counter
buy cytotec over the counter: cytotec online – cytotec abortion pill
ivermectin 3 mg tablets for humans – doryx cost cefaclor 250mg canada
cipro ciprofloxacin: ciprofloxacin – buy cipro online canada
http://misoprostol.top/# cytotec online
diflucan cost in india diflucan 150 mg price where to buy diflucan pills
aromatase inhibitors tamoxifen: tamoxifen and osteoporosis – how to prevent hair loss while on tamoxifen
cytotec online: Misoprostol 200 mg buy online – order cytotec online
ciprofloxacin mail online: ciprofloxacin over the counter – cipro for sale
buy diflucan 150mg: online rx diflucan – diflucan cream prescription
buy ciprofloxacin buy cipro online ciprofloxacin
Abortion pills online: cytotec online – buy cytotec over the counter
https://nolvadex.icu/# does tamoxifen cause bone loss
doxycycline order online buy doxycycline online 270 tabs doxycycline order online
cipro ciprofloxacin: buy ciprofloxacin – ciprofloxacin
Misoprostol 200 mg buy online: cytotec online – buy cytotec online fast delivery
buy doxycycline: doxycycline hyc – purchase doxycycline online
ventolin 2mg cheap – purchase seroflo pill theo-24 Cr buy online
diflucan pill for sale: diflucan discount – where can i get diflucan
buy cytotec over the counter cytotec abortion pill purchase cytotec
can you buy diflucan in mexico: diflucan 150 mg price – how to get diflucan
http://stromectola.top/# ivermectin 2mg
prednisone buy without prescription prednisone for sale in canada prednisone over the counter uk
ivermectin 1%cream: ivermectin 3mg dosage – ivermectin cost uk
prednisone 54 prednisone 60 mg tablet prednisone 250 mg
prednisone pills 10 mg: buy prednisone online uk – prednisone 0.5 mg
prednisone 5 mg: prednisone brand name in usa – prednisone 10mg buy online
over the counter prednisone cheap prednisone tabs 20 mg how can i order prednisone
buy zithromax online australia: zithromax for sale cheap – zithromax canadian pharmacy
http://amoxicillina.top/# amoxicillin 500 tablet
http://amoxicillina.top/# rexall pharmacy amoxicillin 500mg
cost of generic clomid pills: generic clomid tablets – cheap clomid without insurance
zithromax antibiotic azithromycin zithromax zithromax 600 mg tablets
http://azithromycina.pro/# zithromax online usa no prescription
get cheap clomid without a prescription: can i order generic clomid no prescription – where to buy cheap clomid without prescription
https://azithromycina.pro/# zithromax drug
purchase stromectol online: purchase stromectol online – how much does ivermectin cost
buy prednisone 5mg canada prednisone over the counter south africa prednisone 20mg online
methylprednisolone over counter – buy generic depo-medrol over the counter astelin sprayers
prednisone cost us: buy prednisone from canada – prednisone 2.5 mg tab
can you buy zithromax over the counter: zithromax order online uk – buy zithromax online fast shipping
https://stromectola.top/# where can i buy stromectol
synthroid 125
cheap ed buy ed meds online where can i get ed pills
http://edpill.top/# ed medicines
https://onlinepharmacyworld.shop/# rx pharmacy coupons
cheap ed meds online: cheap ed meds online – buy erectile dysfunction pills online
http://edpill.top/# ed treatment online
buy medication online without prescription buying drugs without prescription canadian rx prescription drugstore
oral desloratadine – aristocort 4mg tablet cheap albuterol
no prescription needed pharmacy: canadian pharmacy no prescription – canadian pharmacy coupon code
http://onlinepharmacyworld.shop/# canadian pharmacy no prescription
non prescription online pharmacy: no prescription medicines – mexican prescription drugs online
buy ed medication online erectile dysfunction drugs online cheap ed
http://onlinepharmacyworld.shop/# foreign pharmacy no prescription
pills no prescription: buy pills without prescription – online drugs no prescription
http://onlinepharmacyworld.shop/# rxpharmacycoupons
https://medicationnoprescription.pro/# buy prescription online
ed online prescription cheapest ed meds get ed meds online
best canadian pharmacy no prescription: canada online pharmacy no prescription – best canadian pharmacy no prescription
https://edpill.top/# ed online meds
https://onlinepharmacyworld.shop/# rx pharmacy coupons
canadian pharmacy without a prescription: canadian pharmacy no prescription needed – prescription meds from canada
sitagliptin side effects
online erectile dysfunction buy erectile dysfunction treatment erectile dysfunction medications online
pharmacy coupons: online pharmacy no prescription needed – canadian pharmacy no prescription
http://onlinepharmacyworld.shop/# us pharmacy no prescription
http://casinvietnam.com/# casino tr?c tuy?n vi?t nam
web c? b?c online uy tin casino tr?c tuy?n casino tr?c tuy?n vi?t nam
https://casinvietnam.shop/# danh bai tr?c tuy?n
casino online uy tin casino tr?c tuy?n vi?t nam choi casino tr?c tuy?n tren di?n tho?i
casino online uy tín: casino tr?c tuy?n vi?t nam – casino online uy tín
alcohol and venlafaxine
micronase usa – buy glucotrol 5mg dapagliflozin 10mg pill
casino online uy tin casino tr?c tuy?n casino tr?c tuy?n
ic tamsulosin hcl 0.4 mg capsule
venlafaxine 75 mg price
casino tr?c tuy?n vi?t nam: casino tr?c tuy?n vi?t nam – casino online uy tín
game c? b?c online uy tín: choi casino tr?c tuy?n trên di?n tho?i – game c? b?c online uy tín
https://casinvietnam.com/# choi casino tr?c tuy?n tren di?n tho?i
casino tr?c tuy?n uy tín: casino online uy tín – casino tr?c tuy?n vi?t nam
http://casinvietnam.shop/# casino tr?c tuy?n
tizanidine and hydrocodone
game c? b?c online uy tín: casino tr?c tuy?n – casino online uy tín
https://casinvietnam.shop/# game c? b?c online uy tin
casino online uy tín: casino tr?c tuy?n vi?t nam – game c? b?c online uy tín
buy glucophage – acarbose 50mg for sale precose usa
https://casinvietnam.com/# casino tr?c tuy?n vi?t nam
casino online uy tín: web c? b?c online uy tín – choi casino tr?c tuy?n trên di?n tho?i
voltaren emulgel 2.32
casino tr?c tuy?n vi?t nam: casino tr?c tuy?n uy tín – web c? b?c online uy tín
web c? b?c online uy tín: casino tr?c tuy?n vi?t nam – casino tr?c tuy?n
http://casinvietnam.shop/# game c? b?c online uy tin
casino tr?c tuy?n vi?t nam: dánh bài tr?c tuy?n – casino tr?c tuy?n vi?t nam
http://casinvietnam.com/# web c? b?c online uy tin
casino tr?c tuy?n uy tín: dánh bài tr?c tuy?n – casino tr?c tuy?n vi?t nam
prandin 2mg cheap – order empagliflozin online cheap order empagliflozin 10mg sale
casino tr?c tuy?n uy tín: web c? b?c online uy tín – casino tr?c tuy?n
300 mg generic version of wellbutrin xl
https://mexicoph24.life/# buying prescription drugs in mexico
http://indiaph24.store/# mail order pharmacy india
https://mexicoph24.life/# mexican online pharmacies prescription drugs
zyprexa images
http://indiaph24.store/# top 10 online pharmacy in india
https://indiaph24.store/# indian pharmacy online
https://canadaph24.pro/# canada pharmacy
http://canadaph24.pro/# canadian pharmacy uk delivery
https://mexicoph24.life/# pharmacies in mexico that ship to usa
https://canadaph24.pro/# canadian pharmacy 24h com safe
https://mexicoph24.life/# reputable mexican pharmacies online
zofran tablet size
http://canadaph24.pro/# best canadian online pharmacy
https://canadaph24.pro/# canadian pharmacy world reviews
http://mexicoph24.life/# mexican pharmacy
http://indiaph24.store/# indianpharmacy com
https://mexicoph24.life/# reputable mexican pharmacies online
https://indiaph24.store/# top 10 pharmacies in india
rybelsus 14 mg sale – buy DDAVP generic purchase DDAVP spray
https://indiaph24.store/# indian pharmacies safe
zetia dosing
http://mexicoph24.life/# mexico pharmacies prescription drugs
buy generic lamisil – buy generic griseofulvin buy generic griseofulvin online
http://canadaph24.pro/# canadian mail order pharmacy
getting off zyprexa
http://mexicoph24.life/# mexican online pharmacies prescription drugs
http://indiaph24.store/# Online medicine order
https://canadaph24.pro/# canadian pharmacy world
http://indiaph24.store/# india online pharmacy
http://indiaph24.store/# online pharmacy india
http://mexicoph24.life/# medication from mexico pharmacy
http://indiaph24.store/# indianpharmacy com
https://mexicoph24.life/# pharmacies in mexico that ship to usa
online shopping pharmacy india https://indiaph24.store/# pharmacy website india
buy medicines online in india
http://mexicoph24.life/# mexican rx online
https://indiaph24.store/# buy prescription drugs from india
https://canadaph24.pro/# canadian pharmacy meds review
http://canadaph24.pro/# my canadian pharmacy review
http://canadaph24.pro/# canadadrugpharmacy com
http://indiaph24.store/# online shopping pharmacy india
http://indiaph24.store/# world pharmacy india
https://indiaph24.store/# pharmacy website india
http://mexicoph24.life/# buying prescription drugs in mexico
http://canadaph24.pro/# canada rx pharmacy
order ketoconazole 200 mg generic – buy butenafine generic buy generic sporanox for sale
https://mexicoph24.life/# mexico pharmacy
https://indiaph24.store/# best india pharmacy
https://canadaph24.pro/# online canadian drugstore
http://mexicoph24.life/# buying prescription drugs in mexico online
https://canadaph24.pro/# vipps approved canadian online pharmacy
психологи цены https://w-495.ru/
famvir for sale online – valcivir 500mg pills buy valcivir medication
https://canadaph24.pro/# canadian pharmacy meds
https://mexicoph24.life/# mexico drug stores pharmacies
buying drugs from canada Licensed Canadian Pharmacy canadian pharmacy online
medicine in mexico pharmacies Online Pharmacies in Mexico mexican online pharmacies prescription drugs
http://canadaph24.pro/# canadianpharmacyworld com
best online pharmacies in mexico: Online Pharmacies in Mexico – purple pharmacy mexico price list
https://indiaph24.store/# reputable indian pharmacies
http://indiaph24.store/# best india pharmacy
п»їlegitimate online pharmacies india https://indiaph24.store/# Online medicine home delivery
top online pharmacy india
cheap canadian pharmacy online Prescription Drugs from Canada best canadian pharmacy online
buying from online mexican pharmacy mexico pharmacy reputable mexican pharmacies online
mexican mail order pharmacies: Mexican Pharmacy Online – mexico pharmacies prescription drugs
get generic propecia price: propecia generic – cost of cheap propecia pill
generic drug for lisinopril lisinopril hctz prinivil 5mg tablet
buy cipro ciprofloxacin generic buy ciprofloxacin over the counter
http://nolvadex.life/# tamoxifen breast cancer prevention
tamoxifen alternatives tamoxifen 20 mg tablet tamoxifen endometriosis
https://ciprofloxacin.tech/# where can i buy cipro online
order generic propecia: cost propecia without insurance – order cheap propecia pills
lisinopril tab 100mg: lisinopril no prescription – lisinopril 12.5 mg 10 mg
zestril no prescription can i order lisinopril online lisinopril 5 mg tablet price in india
https://cytotec.club/# cytotec online
http://ciprofloxacin.tech/# buy ciprofloxacin over the counter
buy cytotec order cytotec online buy cytotec pills online cheap
https://cytotec.club/# Misoprostol 200 mg buy online
п»їcipro generic ciprofloxacin 500 mg tablet price ciprofloxacin
https://finasteride.store/# cost cheap propecia for sale
tamoxifen mechanism of action: tamoxifen effectiveness – dcis tamoxifen
should i take tamoxifen: nolvadex gynecomastia – tamoxifen buy
order lanoxin 250 mg without prescription – buy trandate 100mg pills buy lasix 40mg without prescription
lisinopril generic price zestril tablet lisinopril 20mg tablets price
https://cytotec.club/# cytotec online
buy cipro antibiotics cipro where can i buy cipro online
http://finasteride.store/# get cheap propecia tablets
buy cipro cheap: ciprofloxacin – buy cipro online canada
https://finasteride.store/# cheap propecia prices
tamoxifen for breast cancer prevention: nolvadex vs clomid – what happens when you stop taking tamoxifen
cipro for sale buy cipro online cipro for sale
zofran procedure code
propecia pill get generic propecia get propecia no prescription
https://finasteride.store/# propecia tablet
https://nolvadex.life/# tamoxifen warning
http://nolvadex.life/# tamoxifen dose
does tamoxifen cause menopause: what happens when you stop taking tamoxifen – where can i buy nolvadex
Abortion pills online buy cytotec Abortion pills online
cipro: cipro 500mg best prices – ciprofloxacin over the counter
pct nolvadex nolvadex for sale does tamoxifen cause bone loss
https://cytotec.club/# Misoprostol 200 mg buy online
http://ciprofloxacin.tech/# cipro
tamoxifen and depression tamoxifen dosage tamoxifen cyp2d6
buy cytotec over the counter: order cytotec online – buy cytotec pills
buy misoprostol over the counter: buy cytotec over the counter – buy cytotec pills online cheap
https://nolvadex.life/# tamoxifen breast cancer
https://ciprofloxacin.tech/# cipro ciprofloxacin
ciprofloxacin generic price ciprofloxacin mail online ciprofloxacin order online
http://lisinopril.network/# lisinopril 40 mg price in india
buy cytotec over the counter: buy cytotec in usa – cytotec pills buy online
cost of propecia tablets propecia prices cost propecia for sale
http://cytotec.club/# buy cytotec pills online cheap
cytotec pills buy online: buy cytotec over the counter – cytotec pills online
https://cytotec.club/# buy cytotec
buy ciprofloxacin cipro for sale purchase cipro
hysterectomy after breast cancer tamoxifen nolvadex pills nolvadex gynecomastia
https://cytotec.club/# cytotec pills buy online
tamoxifen moa: nolvadex pct – low dose tamoxifen
lopressor 50mg sale – order telmisartan generic buy adalat 10mg online
how to prevent hair loss while on tamoxifen: tamoxifen postmenopausal – tamoxifen dosage
order generic propecia without insurance cost of propecia without dr prescription cost of cheap propecia without rx
https://cytotec.club/# buy cytotec online fast delivery
https://lisinopril.network/# lisinopril 2.5
cost of generic propecia without prescription get propecia without rx get cheap propecia without dr prescription
http://lisinopril.network/# lisinopril buy without prescription
cytotec pills online: buy cytotec pills online cheap – cytotec pills buy online
ciprofloxacin generic price: ciprofloxacin mail online – buy generic ciprofloxacin
hydrochlorothiazide online order – plendil 10mg drug zebeta 10mg uk
https://nolvadex.life/# п»їdcis tamoxifen
cytotec abortion pill cytotec online cytotec buy online usa
https://ciprofloxacin.tech/# buy cipro online
buy cheap propecia tablets buying propecia pill buy cheap propecia no prescription
http://cytotec.club/# buy cytotec online
cytotec pills buy online: buy cytotec pills – cytotec buy online usa
aromatase inhibitor tamoxifen nolvadex 20mg tamoxifen bone density
order cheap propecia tablets get generic propecia pill buy generic propecia no prescription
http://finasteride.store/# propecia price
https://cytotec.club/# order cytotec online
https://ciprofloxacin.tech/# cipro ciprofloxacin
п»їcytotec pills online buy cytotec online fast delivery cytotec pills buy online
tamoxifen hip pain: nolvadex online – tamoxifen depression
levitra erectile dysfunction
tamoxifen vs raloxifene: nolvadex steroids – tamoxifen dosage
buy cytotec pills online cheap buy cytotec over the counter п»їcytotec pills online
https://cytotec.club/# Misoprostol 200 mg buy online
https://lisinopril.network/# lisinopril 50 mg tablet
Buy Vardenafil online: Buy generic Levitra online – Vardenafil price
Cheap Viagra 100mg Buy Viagra online cheap Cheap Viagra 100mg
п»їcialis generic: Generic Tadalafil 20mg price – Generic Cialis price
https://kamagra.win/# Kamagra 100mg price
cenforce.pro Buy Cenforce 100mg Online Purchase Cenforce Online
https://cenforce.pro/# cheapest cenforce
Levitra generic best price: Buy Vardenafil 20mg – Buy Vardenafil online
https://levitrav.store/# buy Levitra over the counter
cenforce for sale Cenforce 150 mg online cheapest cenforce
Generic Tadalafil 20mg price: Tadalafil price – Generic Cialis without a doctor prescription
https://kamagra.win/# Kamagra Oral Jelly
purchase tadalafil online
cenforce.pro: cenforce.pro – Cenforce 100mg tablets for sale
Generic Tadalafil 20mg price buy cialis online Buy Cialis online
http://kamagra.win/# Kamagra 100mg
Tadalafil price Generic Tadalafil 20mg price Tadalafil price
https://cialist.pro/# Generic Cialis price
Cialis 20mg price: buy cialis online – Generic Cialis without a doctor prescription
Kamagra tablets kamagra Kamagra 100mg price
Cialis without a doctor prescription: buy cialis online – cialis generic
cenforce.pro: cheapest cenforce – Cenforce 150 mg online
http://cialist.pro/# Buy Tadalafil 10mg
Order Viagra 50 mg online Cheap Viagra 100mg Buy Viagra online cheap
nitroglycerin for sale – order generic nitroglycerin buy valsartan 80mg for sale
Viagra generic over the counter Buy Viagra online viagra canada
Cialis without a doctor prescription: Cialis 20mg price in USA – Buy Tadalafil 20mg
There are also a number of attachments that can be used for internal stimulation during vaginal or hot cam girls. Essentially, such a package provides all the pieces needed to make cam sessions interesting and distinctive every time.
snafi tadalafil
Kamagra 100mg: Kamagra 100mg – super kamagra
Cheap generic Viagra: viagras.online – Cheap generic Viagra
https://cialist.pro/# Cialis over the counter
viagra canada Buy generic 100mg Viagra online buy viagra here
https://viagras.online/# Cheap Sildenafil 100mg
levitra dosing
https://cialist.pro/# Generic Tadalafil 20mg price
http://cenforce.pro/# Cenforce 150 mg online
buy cenforce: cenforce for sale – order cenforce
http://cialist.pro/# Cialis 20mg price
Kamagra 100mg: kamagra – kamagra
https://cialist.pro/# Tadalafil Tablet
http://cialist.pro/# Buy Tadalafil 10mg
Buy Levitra 20mg online Levitra generic price Vardenafil buy online
Циклёвка паркета: особенности и этапы услуги
Циклёвка паркета — это процесс восстановления внешнего вида паркетного пола путём удаления верхнего повреждённого слоя и возвращения ему первоначального вида. Услуга включает в себя несколько этапов:
Подготовка: перед началом работы необходимо защитить мебель и другие предметы от пыли и грязи, а также удалить плинтусы.
Шлифовка: с помощью шлифовальной машины удаляется старый лак и верхний повреждённый слой древесины.
Шпатлёвка: после шлифовки поверхность паркета шпатлюется для заполнения трещин и выравнивания поверхности.
Грунтовка: перед нанесением лака паркет грунтуется для улучшения адгезии и защиты от плесени и грибка.
Нанесение лака: лак наносится в несколько слоёв с промежуточной шлифовкой между ними.
Полировка: после нанесения последнего слоя лака паркет полируется для придания поверхности блеска и гладкости.
Циклёвка паркета позволяет обновить внешний вид пола, восстановить его структуру и продлить срок службы.
Сайт: ykladka-parketa.ru
Циклевка паркета
cheapest cialis: cialist.pro – Buy Tadalafil 10mg
Levitra 20 mg for sale Levitra 20mg price п»їLevitra price
https://viagras.online/# viagra canada
Generic Levitra 20mg: Vardenafil online prescription – Levitra price
Kamagra Oral Jelly: Kamagra tablets – Kamagra 100mg price
https://cialist.pro/# Buy Tadalafil 20mg
Cenforce 150 mg online Purchase Cenforce Online order cenforce
Buy Viagra online cheap: viagras.online – buy Viagra online
Kamagra tablets kamagra pills п»їkamagra
prescription drugs canada buy online: canadian pharmacy meds – pharmacy canadian
cheap canadian pharmacy online legit canadian pharmacy online best canadian online pharmacy
mexican drugstore online: mexican border pharmacies shipping to usa – mexico drug stores pharmacies
canadian pharmacy ltd canada drugs online review reliable canadian pharmacy
https://pharmindia.online/# cheapest online pharmacy india
https://pharmcanada.shop/# canadian pharmacy 24 com
cheapest pharmacy for prescriptions without insurance: canadian pharmacy world coupon – cheapest pharmacy for prescriptions
simvastatin meet – fenofibrate surface atorvastatin myth
online drugs without prescription canada prescription online overseas online pharmacy-no prescription
http://pharmnoprescription.icu/# pharmacy no prescription
canada discount pharmacy: canadian pharmacy meds – canadian pharmacy 24
http://pharmindia.online/# india online pharmacy
mexico pharmacy: mexican mail order pharmacies – mexico drug stores pharmacies
canada pharmacy coupon pharm world no prescription needed pharmacy
buy prescription drugs from india: india online pharmacy – indian pharmacy
buy prescription drugs from india: indian pharmacy online – top 10 pharmacies in india
https://pharmmexico.online/# п»їbest mexican online pharmacies
medication from mexico pharmacy mexican pharmacy mexican border pharmacies shipping to usa
canadian pharmacy no rx needed: maple leaf pharmacy in canada – ed drugs online from canada
canadadrugpharmacy com: pharmacy rx world canada – canadian discount pharmacy
http://pharmindia.online/# india pharmacy
drugstore com online pharmacy prescription drugs pharm world store cheapest pharmacy to get prescriptions filled
mexican rx online: mexico drug stores pharmacies – best online pharmacies in mexico
purple pharmacy mexico price list: buying prescription drugs in mexico – mexico drug stores pharmacies
no prescription online pharmacy without a prescription canada prescription
https://pharmcanada.shop/# my canadian pharmacy rx
pharmacy without prescription pharm world store online pharmacy discount code
best website to buy prescription drugs: canadian prescription drugstore review – no prescription pharmacy
https://pharmindia.online/# reputable indian online pharmacy
indian pharmacy online: top online pharmacy india – india pharmacy
https://pharmindia.online/# reputable indian pharmacies
legit non prescription pharmacies cheapest pharmacy to get prescriptions filled us pharmacy no prescription
cheapest pharmacy for prescriptions without insurance: cheapest pharmacy – cheapest pharmacy for prescriptions
mexican drugstore online reputable mexican pharmacies online reputable mexican pharmacies online
http://pharmindia.online/# indian pharmacy
no prescription drugs online: buying prescription drugs online canada – no prescription drugs online
canadian pharmacy price checker: online canadian drugstore – canadian pharmacy india
buy drugs online without prescription: buy prescription online – online pharmacy reviews no prescription
https://pharmindia.online/# п»їlegitimate online pharmacies india
cheapest pharmacy for prescription drugs pharm world canadian pharmacy discount code
rosuvastatin success – crestor online sit caduet rob
canada pharmacy coupon: pharm world – canada pharmacy coupon
buying prescription drugs in mexico online: buying prescription drugs in mexico – mexican online pharmacies prescription drugs
https://pharmmexico.online/# п»їbest mexican online pharmacies
mexican pharmacy purple pharmacy mexico price list mexico drug stores pharmacies
mexican online pharmacies prescription drugs: mexico drug stores pharmacies – mexican online pharmacies prescription drugs
no prescription needed pharmacy: pharm world store – canadian pharmacy coupon code
http://pharmmexico.online/# mexico drug stores pharmacies
https://pharmnoprescription.icu/# ordering prescription drugs from canada
zithromax buy online no prescription zithromax coupon average cost of generic zithromax
amoxicillin order online no prescription: where to buy amoxicillin 500mg without prescription – buy amoxicillin 500mg
buy prednisone canadian pharmacy: prednisone brand name canada – can i purchase prednisone without a prescription
200 mg doxycycline: doxycycline 100 mg – where to purchase doxycycline
amoxicillin 500 mg without a prescription amoxicillin 500 mg where to buy amoxicillin 500mg without prescription
https://zithromaxa.store/# can i buy zithromax online
price for amoxicillin 875 mg: buy amoxicillin online mexico – can you buy amoxicillin over the counter
generic over the counter prednisone prednisone buy online nz prednisone brand name
amoxicillin 500mg no prescription: amoxicillin online without prescription – antibiotic amoxicillin
http://zithromaxa.store/# zithromax 1000 mg pills
over the counter prednisone cream prednisone 20mg for sale prednisone over the counter cost
buy prednisone 50 mg: prednisone 2 mg – prednisone 15 mg daily
zithromax capsules australia: zithromax 500mg – buy zithromax online australia
doxycycline prices: order doxycycline 100mg without prescription – how to order doxycycline
https://amoxila.pro/# over the counter amoxicillin
vibramycin 100 mg: doxycycline 500mg – generic doxycycline
levitra vs cialis
steroids prednisone for sale: where can i buy prednisone without a prescription – cheapest prednisone no prescription
zithromax for sale online: purchase zithromax online – can i buy zithromax over the counter in canada
doxy: doxycycline mono – order doxycycline
cost of neurontin: neurontin online – neurontin 300mg caps
buy amoxicillin: can you buy amoxicillin over the counter in canada – amoxicillin 500mg capsule cost
prednisone 20mg nz: prednisone 10mg price in india – prednisone purchase online
zithromax prescription in canada: how much is zithromax 250 mg – zithromax capsules australia
neurontin india neurontin 10 mg neurontin prices generic
https://amoxila.pro/# how to get amoxicillin
https://gabapentinneurontin.pro/# neurontin gabapentin
prednisone 15 mg tablet: 40 mg daily prednisone – where to buy prednisone 20mg no prescription
zithromax prescription: cheap zithromax pills – zithromax 500 mg lowest price drugstore online
prednisone 20 mg tablet price: prednisone online india – prednisone 2.5 mg daily
buy cialis online overnight shipping
online doxycycline where to get doxycycline buy doxycycline hyclate 100mg without a rx
http://prednisoned.online/# prednisone price
generic zithromax over the counter generic zithromax medicine azithromycin zithromax
http://amoxila.pro/# amoxicillin 500 mg tablet
neurontin 800: buy neurontin online no prescription – neurontin india
price of doxycycline: doxycycline 100mg capsules – doxy
zithromax online: zithromax prescription in canada – buy zithromax
doxycycline without a prescription: price of doxycycline – buy doxycycline monohydrate
prednisone pill 10 mg prednisone 500 mg tablet prednisone 2 mg daily
buy doxycycline online doxy order doxycycline 100mg without prescription
viagra professional serve – malegra anywhere levitra oral jelly online corpse
https://doxycyclinea.online/# generic doxycycline
https://prednisoned.online/# brand prednisone
zithromax cost uk: zithromax 500 mg lowest price pharmacy online – zithromax z-pak price without insurance
zithromax buy online: where to get zithromax over the counter – zithromax 500 without prescription
buy zithromax online cheap zithromax 500 mg lowest price online zithromax prescription online
cost of amoxicillin prescription: amoxicillin medicine – order amoxicillin no prescription
medication neurontin neurontin 800 mg tablets best price neurontin online pharmacy
Лендинг-пейдж — это одностраничный сайт, предназначенный для рекламы и продажи товаров или услуг, а также для сбора контактных данных потенциальных клиентов. Вот несколько причин, почему лендинг-пейдж важен для бизнеса:
Увеличение узнаваемости компании. Лендинг-пейдж позволяет представить компанию и её продукты или услуги в выгодном свете, что способствует росту узнаваемости бренда.
Повышение продаж. Заказать лендинг можно здесь – 1landingpage.ru Одностраничные сайты позволяют сосредоточиться на конкретных предложениях и акциях, что повышает вероятность совершения покупки.
Оптимизация SEO-показателей. Лендинг-пейдж создаются с учётом ключевых слов и фраз, что улучшает позиции сайта в результатах поиска и привлекает больше целевых посетителей.
Привлечение новой аудитории. Одностраничные сайты могут использоваться для продвижения новых продуктов или услуг, а также для привлечения внимания к определённым кампаниям или акциям.
Расширение клиентской базы. Лендинг-пейдж собирают контактные данные потенциальных клиентов, что позволяет компании поддерживать связь с ними и предлагать дополнительные услуги или товары.
Простота генерации лидов. Лендинг-пейдж предоставляют краткую и понятную информацию о продуктах или услугах, что облегчает процесс принятия решения для потенциальных клиентов.
Сбор персональных данных. Лендинг-пейдж позволяют собирать информацию о потенциальных клиентах, такую как email-адрес, имя и контактные данные, что помогает компании лучше понимать свою аудиторию и предоставлять более персонализированные услуги.
Улучшение поискового трафика. Лендинг-пейдж создаются с учётом определённых поисковых запросов, что позволяет привлекать больше целевых посетителей на сайт.
Эффективное продвижение новой продукции. Лендинг-пейдж можно использовать для продвижения новых товаров или услуг, что позволяет привлечь внимание потенциальных клиентов и стимулировать их к покупке.
Лёгкий процесс принятия решений. Лендинг-пейдж содержат только самую необходимую информацию, что упрощает процесс принятия решения для потенциальных клиентов.
В целом, лендинг-пейдж являются мощным инструментом для продвижения бизнеса, увеличения продаж и привлечения новых клиентов.
Заказать лендинг
cialis prescription online
http://doxycyclinea.online/# doxycycline mono
http://zithromaxa.store/# zithromax prescription in canada
generic for doxycycline: doxycycline 100mg dogs – buy doxycycline monohydrate
amoxicillin generic brand: order amoxicillin 500mg – amoxicillin 500 mg purchase without prescription
buy levitra
cost of generic zithromax zithromax for sale 500 mg azithromycin zithromax
priligy fight – aurogra give cialis with dapoxetine graceful
where can you buy zithromax: purchase zithromax z-pak – buy cheap zithromax online
https://gabapentinneurontin.pro/# how much is neurontin pills
doxycycline generic: buy doxycycline hyclate 100mg without a rx – doxycycline generic
https://prednisoned.online/# 10 mg prednisone
generic amoxicillin cost where can i buy amoxicillin online amoxicillin 500mg buy online uk
zithromax z-pak: how to get zithromax online – zithromax for sale 500 mg
prednisone 20 mg purchase: prednisone online india – prednisone rx coupon
ampicillin amoxicillin amoxicillin online purchase amoxicillin 500mg capsules antibiotic
neurontin 600 mg price: neurontin sale – neurontin from canada
http://gabapentinneurontin.pro/# neurontin 400 mg price
http://gabapentinneurontin.pro/# cost of neurontin
neurontin 400 mg capsule cheap neurontin neurontin medication
doxycycline tetracycline: cheap doxycycline online – buy cheap doxycycline
zithromax tablets for sale: zithromax 250 mg tablet price – azithromycin zithromax
doxycycline mono buy doxycycline 100mg doxycycline hyclate
prednisone cost us: buy prednisone without a prescription – prednisone 20 mg pill
http://zithromaxa.store/# can i buy zithromax over the counter in canada
prescription medication neurontin: buy neurontin online uk – gabapentin 600 mg
http://prednisoned.online/# prednisone without prescription 10mg
buy zithromax online with mastercard zithromax 250 zithromax buy online
buy amoxicillin 500mg canada: amoxicillin brand name – buy amoxicillin online with paypal
can you buy zithromax over the counter: zithromax 500 tablet – zithromax z-pak price without insurance
amoxicillin online purchase amoxicillin 500mg capsules uk amoxil generic
buy zithromax: zithromax 500 mg lowest price drugstore online – zithromax 250
http://amoxila.pro/# can you buy amoxicillin uk
clotrimazole price pharmacy
amoxicillin 30 capsules price: can you buy amoxicillin over the counter in canada – buy amoxicillin canada
https://zithromaxa.store/# zithromax buy
can you buy zithromax online: can you buy zithromax over the counter in australia – zithromax azithromycin
generic gabapentin buy neurontin canadian pharmacy neurontin 50mg tablets
how much is prednisone 10 mg: prednisone 20 mg without prescription – buy prednisone 40 mg
buy zithromax online how to get zithromax can you buy zithromax online
http://prednisoned.online/# buy prednisone online without a script
generic gabapentin: neurontin generic south africa – buy neurontin canadian pharmacy
https://amoxila.pro/# amoxicillin script
gabapentin 300mg buying neurontin online drug neurontin 20 mg
cost of prednisone in canada: prednisone cream over the counter – prednisone 5084
how much is amoxicillin prescription: amoxil pharmacy – amoxicillin 500mg pill
buy prednisone 10mg online how to buy prednisone prednisone 10 mg over the counter
sildenafil not working
http://prednisoned.online/# how much is prednisone 10mg
generic doxycycline: doxycycline hyc 100mg – doxy
doxycycline hyclate 100 mg cap: generic doxycycline – doxycycline 50 mg
where to get zithromax over the counter: purchase zithromax online – purchase zithromax z-pak
cenforce health – brand viagra arch
http://amoxila.pro/# amoxicillin 500mg cost
prednisone 30 mg: 40 mg prednisone pill – prednisone uk over the counter
prednisone pills 10 mg where to buy prednisone in canada how much is prednisone 10 mg
https://gabapentinneurontin.pro/# canada neurontin 100mg discount
prednisone: prednisone uk price – medicine prednisone 5mg
zithromax z-pak price without insurance: zithromax 250 mg pill – order zithromax without prescription
doxycycline 100 mg: doxycycline monohydrate – doxycycline without prescription
https://gabapentinneurontin.pro/# neurontin online usa
prednisone over the counter uk buying prednisone on line cheap prednisone online
sildenafil side effects with alcohol
prednisone tablets 2.5 mg: prednisone 5 mg cheapest – where to buy prednisone without prescription
https://amoxila.pro/# amoxicillin no prescription
brand cialis account – viagra soft tabs trust penisole dragon
meijer pharmacy amoxicillin
azithromycin zithromax: zithromax prescription in canada – buy zithromax online fast shipping
zithromax 500mg zithromax cost canada zithromax online paypal
doxycycline hyclate 100 mg cap: doxycycline online – doxycycline 100mg price
http://amoxila.pro/# amoxicillin generic
zithromax online usa: zithromax purchase online – zithromax drug
where can i buy amoxocillin where can i buy amoxicillin without prec amoxicillin 500mg buy online uk
https://gabapentinneurontin.pro/# where can i buy neurontin online
neurontin pill: neurontin 300 mg mexico – neurontin 400 mg price
generic zithromax over the counter can you buy zithromax over the counter in australia zithromax over the counter
neurontin cost australia: neurontin capsules 600mg – neurontin tablets 100mg
can i buy zithromax over the counter: generic zithromax 500mg – how much is zithromax 250 mg
http://amoxila.pro/# where can i buy amoxicillin over the counter
neurontin cost in canada gabapentin 100mg neurontin generic cost
can you buy amoxicillin over the counter in canada: where to buy amoxicillin – amoxicillin for sale
buying from online mexican pharmacy: mexican mail order pharmacies – mexican pharmacy
medication from mexico pharmacy: mexican online pharmacies prescription drugs – medication from mexico pharmacy
mexican pharmacy medicine in mexico pharmacies best online pharmacies in mexico
п»їbest mexican online pharmacies: mexican online pharmacies prescription drugs – mexico drug stores pharmacies
pharmacies in mexico that ship to usa mexican drugstore online mexican border pharmacies shipping to usa
He never realized how far-reaching this one decision would be. I read texts that they wrote to each other while we were still married saying
https://mexicanpharmacy1st.shop/# mexican border pharmacies shipping to usa
reputable mexican pharmacies online: mexican drugstore online – mexico pharmacies prescription drugs
https://mexicanpharmacy1st.com/# buying from online mexican pharmacy
http://mexicanpharmacy1st.com/# mexico pharmacies prescription drugs
medication from mexico pharmacy best online pharmacies in mexico mexican rx online
mexico pharmacies prescription drugs: mexico pharmacy – reputable mexican pharmacies online
https://mexicanpharmacy1st.com/# pharmacies in mexico that ship to usa
medicine in mexico pharmacies: best online pharmacies in mexico – mexican online pharmacies prescription drugs
mexico drug stores pharmacies п»їbest mexican online pharmacies п»їbest mexican online pharmacies
mexican pharmaceuticals online: best online pharmacies in mexico – buying prescription drugs in mexico online
buying from online mexican pharmacy medication from mexico pharmacy medication from mexico pharmacy
https://mexicanpharmacy1st.com/# mexican mail order pharmacies
reputable mexican pharmacies online: mexican rx online – mexico drug stores pharmacies
mexican drugstore online: pharmacies in mexico that ship to usa – mexico drug stores pharmacies
http://mexicanpharmacy1st.com/# п»їbest mexican online pharmacies
mexican mail order pharmacies: mexican border pharmacies shipping to usa – mexican border pharmacies shipping to usa
buying prescription drugs in mexico online mexico pharmacy mexico drug stores pharmacies
buying from online mexican pharmacy best online pharmacies in mexico mexico drug stores pharmacies
https://mexicanpharmacy1st.shop/# mexican mail order pharmacies
mexican mail order pharmacies: mexican rx online – mexican online pharmacies prescription drugs
https://mexicanpharmacy1st.online/# buying prescription drugs in mexico
medicine in mexico pharmacies: pharmacies in mexico that ship to usa – mexican mail order pharmacies
mexican online pharmacies prescription drugs: mexican border pharmacies shipping to usa – mexican online pharmacies prescription drugs
medicine in mexico pharmacies medicine in mexico pharmacies buying prescription drugs in mexico
http://mexicanpharmacy1st.com/# mexican pharmacy
brand cialis riddle – viagra soft tabs knife penisole curious
buying prescription drugs in mexico mexico pharmacy buying prescription drugs in mexico online
http://mexicanpharmacy1st.com/# reputable mexican pharmacies online
mexico drug stores pharmacies: medicine in mexico pharmacies – best online pharmacies in mexico
https://mexicanpharmacy1st.online/# purple pharmacy mexico price list
reputable mexican pharmacies online: mexican pharmacy – pharmacies in mexico that ship to usa
mexico drug stores pharmacies: medicine in mexico pharmacies – mexican rx online
cialis soft tabs online click – cialis oral jelly online unusual viagra oral jelly answer
mexican pharmaceuticals online mexican mail order pharmacies reputable mexican pharmacies online
pharmacies in mexico that ship to usa mexican pharmacy buying prescription drugs in mexico
https://mexicanpharmacy1st.online/# reputable mexican pharmacies online
mexican pharmaceuticals online: mexico drug stores pharmacies – buying prescription drugs in mexico
https://mexicanpharmacy1st.shop/# medication from mexico pharmacy
buying prescription drugs in mexico online: buying prescription drugs in mexico online – mexican pharmaceuticals online
buying prescription drugs in mexico online: pharmacies in mexico that ship to usa – pharmacies in mexico that ship to usa
http://mexicanpharmacy1st.com/# mexican border pharmacies shipping to usa
https://mexicanpharmacy1st.online/# medication from mexico pharmacy
pharmacies in mexico that ship to usa medication from mexico pharmacy mexico pharmacy
https://mexicanpharmacy1st.shop/# medication from mexico pharmacy
mexico pharmacies prescription drugs: pharmacies in mexico that ship to usa – mexican border pharmacies shipping to usa
best mexican online pharmacies: buying from online mexican pharmacy – mexico pharmacy
https://mexicanpharmacy1st.com/# mexican online pharmacies prescription drugs
medication from mexico pharmacy: buying from online mexican pharmacy – buying prescription drugs in mexico
buying prescription drugs in mexico buying from online mexican pharmacy medicine in mexico pharmacies
https://mexicanpharmacy1st.com/# medicine in mexico pharmacies
medicine in mexico pharmacies: buying prescription drugs in mexico – pharmacies in mexico that ship to usa
purple pharmacy mexico price list buying prescription drugs in mexico п»їbest mexican online pharmacies
https://mexicanpharmacy1st.online/# mexico pharmacies prescription drugs
buying prescription drugs in mexico online: purple pharmacy mexico price list – mexican mail order pharmacies
mexico drug stores pharmacies: mexican online pharmacies prescription drugs – п»їbest mexican online pharmacies
mexican pharmacy: buying prescription drugs in mexico online – mexican online pharmacies prescription drugs
п»їbest mexican online pharmacies mexican pharmaceuticals online pharmacies in mexico that ship to usa
https://mexicanpharmacy1st.online/# mexican mail order pharmacies
mexico pharmacy mexico pharmacies prescription drugs mexican pharmacy
http://mexicanpharmacy1st.com/# purple pharmacy mexico price list
mexican drugstore online: mexican rx online – purple pharmacy mexico price list
buy cytotec online order cytotec online Abortion pills online
http://lisinopril.club/# generic prinivil
prescription drug zestril buy lisinopril zestoretic 20 25mg
cost of cheap propecia without dr prescription: cost of propecia – cost cheap propecia no prescription
http://cytotec.xyz/# buy cytotec online fast delivery
buy cytotec in usa: purchase cytotec – cytotec buy online usa
https://lisinopril.club/# lisinopril buy online
neurontin uk neurontin prescription can you buy neurontin over the counter
my pharmacy online
п»їcytotec pills online Misoprostol 200 mg buy online order cytotec online
cost of prinivil: zestoretic 20 25 – lisinopril 240
lisinopril price without insurance: lisinopril generic price comparison – lisinopril 5mg tabs
purchase neurontin online ordering neurontin online gabapentin online
neurontin cost australia: can you buy neurontin over the counter – neurontin 4000 mg
value rx pharmacy tazewell tn
where to buy generic clomid for sale buy clomid pills clomid without a prescription
http://cytotec.xyz/# п»їcytotec pills online
http://lisinopril.club/# cost of lisinopril in canada
lisinopril 30 mg: buy prinivil – zestril canada
generic lisinopril 5 mg: prinivil 20 mg tablet – purchase lisinopril 10 mg
cost of propecia no prescription buying cheap propecia online cost cheap propecia tablets
http://propeciaf.online/# cost of cheap propecia without dr prescription
32 neurontin neurontin 300mg tablet cost neurontin 200 mg price
http://cytotec.xyz/# buy cytotec online fast delivery
https://gabapentin.club/# neurontin brand name 800mg
order propecia for sale: buy generic propecia prices – buying generic propecia no prescription
order cheap propecia without prescription: cost cheap propecia no prescription – buying cheap propecia online
generic propecia without a prescription get generic propecia without a prescription cost of propecia without prescription
lisinopril medicine lisinopril 20 mg price online lisinopril 10 mg online
http://clomiphene.shop/# where to buy clomid online
http://lisinopril.club/# lisinopril 40 mg prices
neurontin 100 mg cost: prescription price for neurontin – neurontin prices generic
cialis soft tabs ago – cialis soft tabs pills birthday viagra oral jelly online cool
cost cheap clomid price: order cheap clomid no prescription – where to get generic clomid price
cost of clomid pills order clomid price get cheap clomid without dr prescription
tadalafil or sildenafil
http://clomiphene.shop/# rx clomid
buy cytotec in usa cytotec online order cytotec online
http://clomiphene.shop/# cost of generic clomid without prescription
http://propeciaf.online/# propecia generic
online pharmacy generic seroquel
lisinopril 80 mg tablet: lisinopril 20 12.5 mg – lisinopril 4 mg
get cheap propecia without dr prescription: buy propecia without prescription – how cЙ‘n i get cheap propecia pills
lisinopril online canadian pharmacy buy lisinopril without a prescription prinivil
https://gabapentin.club/# neurontin 100mg tab
neurontin 204: neurontin capsule 600mg – neurontin brand coupon
where to buy cheap clomid tablets where can i get generic clomid without prescription get cheap clomid without insurance
buy generic propecia no prescription: cost of propecia pills – buy generic propecia
neurontin: buy cheap neurontin – neurontin prices
neurontin cost in singapore neurontin 900 mg neurontin 400 mg capsules
cytotec buy online usa: buy cytotec pills online cheap – buy cytotec pills
lisinopril 40 mg no prescription order lisinopril 10 mg lisinopril 3
https://gabapentin.club/# neurontin 150 mg
lisinopril price without insurance: lisinopril 10 mg prices – lisinopril brand name in usa
order clomid prices can i buy generic clomid online how can i get cheap clomid for sale
cheapest lisinopril 10 mg: zestoretic 20 25mg – order lisinopril online
buy cytotec over the counter buy cytotec over the counter cytotec pills buy online
https://clomiphene.shop/# where to get generic clomid
Misoprostol 200 mg buy online: buy cytotec over the counter – buy cytotec pills online cheap
https://36and6health.shop/# prescription drugs from canada
https://cheapestandfast.shop/# best no prescription online pharmacy
canada ed drugs rate canadian pharmacies recommended canadian pharmacies
mexican prescription drugs online canadian pharmacy non prescription pharmacy no prescription
https://cheapestcanada.com/# best online canadian pharmacy
https://36and6health.com/# best online pharmacy no prescription
http://cheapestindia.com/# buy prescription drugs from india
http://cheapestindia.com/# online pharmacy india
http://cheapestcanada.com/# pharmacy canadian superstore
http://cheapestcanada.com/# canadian pharmacy phone number
canada pharmacy online cheapestcanada.com canadian pharmacy review
canadian pharmacy ltd: northwest pharmacy canada – onlinepharmaciescanada com
canadian pharmacy no prescription: 36 and 6 pharmacy – non prescription medicine pharmacy
https://cheapestandfast.com/# best non prescription online pharmacy
http://cheapestandfast.com/# online meds without prescription
no prescription needed cheapest & fast pharmacy online drugs without prescription
https://cheapestindia.com/# Online medicine order
https://cheapestandfast.shop/# canadian prescriptions in usa
http://36and6health.com/# prescription free canadian pharmacy
https://cheapestmexico.com/# mexican online pharmacies prescription drugs
https://cheapestcanada.shop/# canada pharmacy world
http://cheapestmexico.com/# mexico drug stores pharmacies
best no prescription pharmacy: 36 and 6 health online pharmacy – pharmacy without prescription
https://cheapestindia.com/# online shopping pharmacy india
medication from mexico pharmacy: buying from online mexican pharmacy – buying prescription drugs in mexico
no prescription required pharmacy canadian pharmacy world coupon online pharmacy discount code
priligy doze – aurogra spell cialis with dapoxetine help
https://cheapestandfast.com/# medications online without prescription
https://36and6health.shop/# canadian pharmacy no prescription needed
https://cheapestmexico.com/# mexican mail order pharmacies
cenforce online glimmer – brand viagra online underneath brand viagra pills music
canadian pharmacy without prescription cheapest & fast pharmacy no prescription pharmacy online
http://cheapestmexico.com/# mexican pharmacy
https://cheapestcanada.shop/# canadian pharmacy store
https://cheapestmexico.com/# purple pharmacy mexico price list
http://36and6health.com/# canadian pharmacy no prescription
buy medication online with prescription cheapest & fast pharmacy canadian pharmacy prescription
http://cheapestandfast.com/# online pharmacy without a prescription
farmacia online: farmacia online – farmacia online senza ricetta
farmacias online seguras: farmacia online madrid – farmacia en casa online descuento
http://euapothekeohnerezept.com/# online apotheke deutschland
online apotheke preisvergleich: online apotheke rezept – online apotheke rezept
internet apotheke: online apotheke preisvergleich – п»їshop apotheke gutschein
Farmacia online piГ№ conveniente farmaci senza ricetta elenco farmacie online affidabili
pharmacie en ligne fiable: pharmacie en ligne france livraison belgique – pharmacie en ligne france fiable
migliori farmacie online 2024 comprare farmaci online con ricetta farmacie online autorizzate elenco
online apotheke versandkostenfrei: online apotheke rezept – europa apotheke
pharmacie en ligne: pharmacie en ligne france livraison internationale – pharmacie en ligne sans ordonnance
internet apotheke: online apotheke deutschland – online apotheke deutschland
Pharmacie en ligne livraison Europe Pharmacie en ligne livraison Europe pharmacie en ligne france livraison internationale
asthma treatment darken – asthma medication section inhalers for asthma driver
pharmacie en ligne livraison europe: Pharmacie sans ordonnance – Pharmacie en ligne livraison Europe
https://eumedicamentenligne.shop/# pharmacie en ligne livraison europe
pharmacie en ligne france livraison belgique: acheter m̩dicament en ligne sans ordonnance Рpharmacie en ligne avec ordonnance
farmacia online 24 horas farmacia online espaГ±a envГo internacional farmacia online madrid
acne medication sawyer – acne medication anybody acne treatment star
apotheke online: online apotheke preisvergleich – medikamente rezeptfrei
top farmacia online: п»їFarmacia online migliore – Farmacie on line spedizione gratuita
farmacia online espaГ±a envГo internacional: farmacias online seguras – farmacia online barata y fiable
п»їfarmacia online espaГ±a farmacias online baratas farmacias online seguras
https://eufarmaciaonline.shop/# farmacia online espaГ±a envГo internacional
pharmacie en ligne sans ordonnance: pharmacie en ligne france livraison internationale – pharmacie en ligne pas cher
günstigste online apotheke: online apotheke deutschland – internet apotheke
online apotheke internet apotheke п»їshop apotheke gutschein
vardenafil hydrochloride trihydrate 20 mg
pharmacie en ligne fiable: pharmacie en ligne avec ordonnance – Pharmacie Internationale en ligne
pharmacie en ligne fiable: vente de mГ©dicament en ligne – pharmacie en ligne fiable
pharmacie en ligne france livraison internationale: pharmacies en ligne certifiГ©es – п»їpharmacie en ligne france
pharmacie en ligne avec ordonnance pharmacie en ligne avec ordonnance pharmacie en ligne france livraison internationale
farmacia online españa envÃo internacional: farmacias online seguras – farmacia online madrid
farmacias online seguras: farmacia en casa online descuento – farmacia online españa envÃo internacional
beste online-apotheke ohne rezept: online apotheke – internet apotheke
pharmacie en ligne france fiable п»їpharmacie en ligne france pharmacie en ligne pas cher
internet apotheke: medikament ohne rezept notfall – online apotheke versandkostenfrei
eu apotheke ohne rezept: gГјnstige online apotheke – online apotheke rezept
internet apotheke: apotheke online – günstigste online apotheke
Achat m̩dicament en ligne fiable: Pharmacie en ligne livraison Europe Рpharmacies en ligne certifi̩es
farmacie online sicure Farmacie on line spedizione gratuita top farmacia online
sildenafil tadalafil combination
Farmacia online piГ№ conveniente: farmaci senza ricetta elenco – farmacia online
Farmacia online piГ№ conveniente: farmacia online piГ№ conveniente – farmacia online piГ№ conveniente
https://eufarmaciaonline.com/# farmacia online barata
comprare farmaci online all’estero: п»їFarmacia online migliore – farmacie online sicure
pharmacie en ligne pas cher pharmacie en ligne france fiable pharmacie en ligne fiable
farmacia online: farmacie online affidabili – acquistare farmaci senza ricetta
apotheke online: günstige online apotheke – europa apotheke
farmacia online barcelona: farmacias online seguras – farmacias direct
Farmacie online sicure migliori farmacie online 2024 farmaci senza ricetta elenco
internet apotheke: apotheke online – online apotheke
comprare farmaci online con ricetta: comprare farmaci online all’estero – farmacie online affidabili
farmacias direct farmacias online seguras farmacia online envГo gratis
migliori farmacie online 2024: Farmacie on line spedizione gratuita – comprare farmaci online con ricetta
farmacias online seguras: farmacia online madrid – farmacia online madrid
farmacias online seguras en espaГ±a: farmacias direct – farmacia online barcelona
prostatitis treatment treasure – prostatitis treatment robot prostatitis pills hotel
Pharmacie Internationale en ligne Pharmacie sans ordonnance pharmacie en ligne fiable
vardenafil for women
tadalafil prostate
https://euapothekeohnerezept.shop/# internet apotheke
treatment for uti jar – treatment for uti pleasure uti antibiotics fine
Pharmacie en ligne livraison Europe: pharmacie en ligne sans ordonnance – pharmacie en ligne sans ordonnance
farmacia online barata y fiable: farmacia online madrid – farmacia online envГo gratis
medikament ohne rezept notfall: eu apotheke ohne rezept – ohne rezept apotheke
pharmacie en ligne france livraison belgique: Achat mГ©dicament en ligne fiable – pharmacie en ligne fiable
pharmacie en ligne pas cher vente de mГ©dicament en ligne pharmacie en ligne france livraison internationale
pharmacie en ligne fiable: pharmacie en ligne – pharmacie en ligne france livraison belgique
Pharmacie Internationale en ligne: pharmacie en ligne france fiable – pharmacie en ligne avec ordonnance
farmaci senza ricetta elenco farmaci senza ricetta elenco farmacia online
https://eufarmaciaonline.com/# farmacias online seguras en espaГ±a
farmacia online espaГ±a envГo internacional: farmacias online seguras – farmacia online barcelona
online apotheke: medikament ohne rezept notfall – online apotheke versandkostenfrei
online apotheke deutschland: medikamente rezeptfrei – п»їshop apotheke gutschein
Achat mГ©dicament en ligne fiable trouver un mГ©dicament en pharmacie pharmacies en ligne certifiГ©es
comprare farmaci online con ricetta: migliori farmacie online 2024 – farmacie online sicure
farmacia online envÃo gratis: farmacia online barcelona – farmacias direct
farmacias online seguras en espaГ±a farmacia online espaГ±a envГo internacional farmacias online seguras en espaГ±a
pharmacie en ligne avec ordonnance: Pharmacie sans ordonnance – Pharmacie sans ordonnance
medikament ohne rezept notfall: beste online-apotheke ohne rezept – ohne rezept apotheke
п»їpharmacie en ligne france pharmacie en ligne pharmacie en ligne france fiable
https://cenligne.shop/# pharmacie en ligne pas cher
pharmacie en ligne avec ordonnance: levitra generique – pharmacie en ligne sans ordonnance
Pharmacie en ligne livraison Europe: levitra en ligne – Achat mГ©dicament en ligne fiable
trouver un mГ©dicament en pharmacie: kamagra en ligne – pharmacie en ligne livraison europe
pharmacie en ligne france livraison internationale cialis sans ordonnance pharmacie en ligne france livraison internationale
pharmacie en ligne pas cher: cialis sans ordonnance – pharmacie en ligne france livraison internationale
Pharmacie Internationale en ligne: pharmacie en ligne – pharmacie en ligne sans ordonnance
Viagra pas cher livraison rapide france: Viagra sans ordonnance 24h – Viagra sans ordonnance pharmacie France
pharmacie en ligne france fiable: achat kamagra – pharmacie en ligne france livraison internationale
trouver un mГ©dicament en pharmacie: levitra generique sites surs – pharmacie en ligne france livraison internationale
Pharmacie Internationale en ligne: kamagra oral jelly – trouver un mГ©dicament en pharmacie
Viagra Pfizer sans ordonnance: Viagra homme prix en pharmacie sans ordonnance – Prix du Viagra 100mg en France
vente de mГ©dicament en ligne: kamagra 100mg prix – pharmacie en ligne
loratadine medication success – loratadine officer loratadine medication coach
Viagra gГ©nГ©rique pas cher livraison rapide: Acheter du Viagra sans ordonnance – Viagra prix pharmacie paris
pharmacie en ligne avec ordonnance Pharmacies en ligne certifiees Achat mГ©dicament en ligne fiable
valacyclovir interrupt – valtrex pills wit valacyclovir pills easy
Viagra homme prix en pharmacie: Viagra 100mg prix – Viagra sans ordonnance livraison 24h
pharmacie en ligne: pharmacie en ligne pas cher – п»їpharmacie en ligne france
pharmacie en ligne france fiable: kamagra pas cher – vente de mГ©dicament en ligne
https://viaenligne.shop/# Prix du Viagra 100mg en France
https://levitraenligne.shop/# Pharmacie Internationale en ligne
Viagra sans ordonnance pharmacie France: Viagra sans ordonnance 24h – Viagra vente libre pays
Hello.
This post was created with XRumer 23 StrongAI.
Good luck 🙂
pharmacie en ligne: achat kamagra – vente de mГ©dicament en ligne
п»їpharmacie en ligne france: acheter kamagra site fiable – Pharmacie en ligne livraison Europe
Hello!
This post was created with XRumer 23 StrongAI.
Good luck 🙂
pharmacie en ligne fiable: pharmacie en ligne pas cher – acheter mГ©dicament en ligne sans ordonnance
acheter mГ©dicament en ligne sans ordonnance: pharmacie en ligne france livraison belgique – pharmacie en ligne france livraison internationale
Quand une femme prend du Viagra homme: Acheter du Viagra sans ordonnance – SildГ©nafil Teva 100 mg acheter
pharmacie en ligne livraison europe: kamagra pas cher – acheter mГ©dicament en ligne sans ordonnance
Pharmacie en ligne livraison Europe: Levitra acheter – pharmacie en ligne france livraison belgique
vente de mГ©dicament en ligne: Cialis sans ordonnance 24h – п»їpharmacie en ligne france
Achat mГ©dicament en ligne fiable: Levitra pharmacie en ligne – vente de mГ©dicament en ligne
pharmacie en ligne france fiable: acheter kamagra site fiable – pharmacie en ligne fiable
Viagra homme prix en pharmacie: Viagra en france livraison rapide – Viagra sans ordonnance 24h Amazon
Viagra prix pharmacie paris: Viagra homme prix en pharmacie sans ordonnance – Viagra en france livraison rapide
priligy law – priligy yell dapoxetine disappointment
acheter mГ©dicament en ligne sans ordonnance: acheter kamagra site fiable – pharmacie en ligne sans ordonnance
pharmacie en ligne france livraison internationale: Levitra sans ordonnance 24h – п»їpharmacie en ligne france
pharmacie en ligne france livraison internationale: kamagra oral jelly – pharmacie en ligne france fiable
Sildenafil teva 100 mg sans ordonnance: Acheter du Viagra sans ordonnance – Viagra femme sans ordonnance 24h
SildГ©nafil 100 mg sans ordonnance: Viagra sans ordonnance livraison 48h – Viagra pas cher paris
Acheter viagra en ligne livraison 24h: Viagra sans ordonnance 24h – Acheter viagra en ligne livraison 24h
pharmacie en ligne: Pharmacie en ligne livraison Europe – pharmacies en ligne certifiГ©es
pharmacie en ligne france pas cher: pharmacie en ligne – trouver un mГ©dicament en pharmacie
loratadine divide – claritin pills willow claritin sparkle
pharmacies en ligne certifiГ©es: levitra generique sites surs – pharmacies en ligne certifiГ©es
pharmacies en ligne certifiГ©es: kamagra 100mg prix – Pharmacie sans ordonnance
Pharmacie sans ordonnance: vente de mГ©dicament en ligne – pharmacies en ligne certifiГ©es
pharmacie en ligne pas cher: Levitra pharmacie en ligne – pharmacies en ligne certifiГ©es
pharmacie en ligne fiable: Medicaments en ligne livres en 24h – Pharmacie Internationale en ligne
pharmacie en ligne france pas cher: pharmacie en ligne sans ordonnance – pharmacie en ligne france livraison internationale
Achat mГ©dicament en ligne fiable: pharmacie en ligne – pharmacie en ligne fiable
pharmacie en ligne: kamagra gel – pharmacie en ligne livraison europe
Pharmacie sans ordonnance: kamagra oral jelly – pharmacie en ligne france livraison internationale
Viagra femme sans ordonnance 24h: viagra en ligne – Viagra Pfizer sans ordonnance
Viagra pas cher livraison rapide france: Meilleur Viagra sans ordonnance 24h – Viagra sans ordonnance livraison 24h
Pharmacie Internationale en ligne: pharmacie en ligne pas cher – pharmacie en ligne france livraison belgique
pharmacie en ligne: pharmacie en ligne – Pharmacie en ligne livraison Europe
pharmacie en ligne france livraison internationale: Acheter Cialis – acheter mГ©dicament en ligne sans ordonnance
pharmacie en ligne pas cher: trouver un mГ©dicament en pharmacie – pharmacie en ligne france pas cher
Pharmacie Internationale en ligne: Cialis sans ordonnance 24h – pharmacie en ligne france fiable
ascorbic acid approach – ascorbic acid wrong ascorbic acid loom
pharmacie en ligne: kamagra pas cher – vente de mГ©dicament en ligne
trouver un mГ©dicament en pharmacie: cialis generique – pharmacie en ligne livraison europe
п»їpharmacie en ligne france: achat kamagra – Pharmacie sans ordonnance
pharmacie en ligne france livraison internationale: cialis sans ordonnance – pharmacie en ligne france livraison internationale
promethazine grass – promethazine new promethazine sky
goodrx tadalafil
pharmacie en ligne france livraison internationale: pharmacie en ligne – pharmacie en ligne fiable
Viagra pas cher livraison rapide france: Viagra vente libre pays – Viagra homme sans ordonnance belgique
trouver un mГ©dicament en pharmacie: Levitra pharmacie en ligne – trouver un mГ©dicament en pharmacie
Prix du Viagra en pharmacie en France: viagra sans ordonnance – SildГ©nafil Teva 100 mg acheter
pharmacie en ligne pas cher: pharmacie en ligne pas cher – acheter mГ©dicament en ligne sans ordonnance
Viagra pas cher paris: Viagra sans ordonnance 24h – Viagra homme sans ordonnance belgique
Viagra pas cher paris: viagra sans ordonnance – Viagra gГ©nГ©rique pas cher livraison rapide
Pharmacie en ligne livraison Europe: kamagra pas cher – pharmacie en ligne avec ordonnance
Viagra pas cher livraison rapide france: Acheter du Viagra sans ordonnance – Viagra homme prix en pharmacie
pharmacie en ligne france livraison internationale: kamagra en ligne – acheter mГ©dicament en ligne sans ordonnance
Viagra gГ©nГ©rique pas cher livraison rapide: Viagra generique en pharmacie – Viagra pas cher inde
pharmacie en ligne france fiable: cialis generique – Achat mГ©dicament en ligne fiable
clarithromycin pills cur – biaxin moon cytotec pills clatter
SildГ©nafil Teva 100 mg acheter: Meilleur Viagra sans ordonnance 24h – SildГ©nafil 100mg pharmacie en ligne
Viagra 100mg prix: viagra sans ordonnance – Viagra vente libre pays
pharmacies en ligne certifiГ©es: cialis prix – pharmacies en ligne certifiГ©es
pharmacie en ligne livraison europe: Achat mГ©dicament en ligne fiable – pharmacie en ligne
Pharmacie en ligne livraison Europe: cialis prix – pharmacie en ligne livraison europe
pharmacie en ligne fiable: pharmacie en ligne sans ordonnance – Pharmacie Internationale en ligne
pharmacie en ligne: kamagra pas cher – vente de mГ©dicament en ligne
п»їpharmacie en ligne france: Acheter Cialis 20 mg pas cher – pharmacie en ligne
pharmacie en ligne livraison europe: Medicaments en ligne livres en 24h – trouver un mГ©dicament en pharmacie
http://levitraenligne.com/# Pharmacie sans ordonnance
п»їpharmacie en ligne france: levitra generique sites surs – Pharmacie sans ordonnance
Viagra homme prix en pharmacie: Meilleur Viagra sans ordonnance 24h – SildГ©nafil 100mg pharmacie en ligne
pharmacie en ligne sans ordonnance: Acheter Cialis 20 mg pas cher – pharmacie en ligne france livraison internationale
fludrocortisone balance – fludrocortisone seal lansoprazole pills duck
pharmacies en ligne certifiГ©es: Cialis sans ordonnance 24h – pharmacie en ligne livraison europe
シンドバッドアドベンチャーは榎本加奈子でどうですか(自动转)
素晴らしい記事!これからも高品質な内容を期待しています。
Viagra sans ordonnance 24h suisse: Viagra sans ordonnance 24h – Viagra pas cher inde
acheter mГ©dicament en ligne sans ordonnance: kamagra en ligne – acheter mГ©dicament en ligne sans ordonnance
п»їpharmacie en ligne france: Medicaments en ligne livres en 24h – pharmacie en ligne france livraison internationale
Viagra en france livraison rapide: Meilleur Viagra sans ordonnance 24h – Viagra 100mg prix
pharmacie en ligne fiable: cialis sans ordonnance – pharmacie en ligne france livraison belgique
pharmacie en ligne avec ordonnance: kamagra livraison 24h – Pharmacie sans ordonnance
trouver un mГ©dicament en pharmacie: levitra generique – acheter mГ©dicament en ligne sans ordonnance
pharmacie en ligne france livraison belgique: cialis prix – pharmacie en ligne livraison europe
pharmacie en ligne avec ordonnance: pharmacie en ligne – pharmacie en ligne pas cher
pharmacies en ligne certifiГ©es: cialis prix – pharmacie en ligne sans ordonnance
pharmacie en ligne france pas cher: kamagra 100mg prix – pharmacie en ligne sans ordonnance
Viagra sans ordonnance pharmacie France: viagra sans ordonnance – Viagra gГ©nГ©rique sans ordonnance en pharmacie
vente de mГ©dicament en ligne: kamagra oral jelly – Pharmacie Internationale en ligne
Viagra prix pharmacie paris: Viagra sans ordonnance 24h – Viagra pas cher paris
п»їpharmacie en ligne france: Levitra 20mg prix en pharmacie – pharmacie en ligne livraison europe
pharmacie en ligne fiable: Medicaments en ligne livres en 24h – Achat mГ©dicament en ligne fiable
pharmacie en ligne fiable: pharmacie en ligne pas cher – pharmacie en ligne france pas cher
Pharmacie en ligne livraison Europe: pharmacie en ligne france livraison belgique – pharmacie en ligne sans ordonnance
pharmacie en ligne france pas cher: Levitra pharmacie en ligne – pharmacie en ligne france pas cher
pharmacie en ligne france pas cher: cialis prix – Pharmacie sans ordonnance
pharmacie en ligne sans ordonnance: Levitra acheter – Pharmacie sans ordonnance
geinoutime.com
반면 팡지판은 침착하게 앞으로 나아가며 “폐하…”라고 말했다.
aciphex 20mg pills – motilium cost order domperidone 10mg generic
purchase dulcolax – bisacodyl 5mg us brand liv52
eukroma uk – eukroma usa duphaston 10 mg price
buy bactrim without prescription – cotrimoxazole 480mg sale order tobramycin 10mg without prescription
Yes, the app is available for download on mobile devices with Android and iOS systems for free. You can download and install the apk on the 1xBet website in the apps section. For optimal performance of the 1xBet App Android devices, the following system requirements should be met: For optimal performance of the 1xBet App Android devices, the following system requirements should be met: 1xbet is the most recognized sportsbook in the world. It offers the biggest bonuses on the market, which combined with high odds and a huge variety of betting options make the company unrivalled in all aspects. The high quality 1xbet casino with a huge number of games attracts thousands of new users every day. 1xbet is the most recognized sportsbook in the world. It offers the biggest bonuses on the market, which combined with high odds and a huge variety of betting options make the company unrivalled in all aspects. The high quality 1xbet casino with a huge number of games attracts thousands of new users every day.
https://daemin.org/bbs/board.php?bo_table=free&wr_id=33503
Screen DPI120-640dpi The Aviator app is available for both Android (5.1 and above) and iOS mobile devices. Ensure your device meets the minimum system requirements. To avoid issues such as account suspension or payout hold, you should strictly adhere to the rules and restrictions which we will explain during the registration process. We strongly recommend that you familiarize yourself with the documentation before using the app. For beginners we recommend to start playing the demo version of Aviator. Downloading and installing the 1xBet mobile app from Google Play Market is not possible as Android blocks betting and gambling applications for real money. However, this can be done through the official website of the bookmaker. If you want to download and install the 1xBet application on your device, follow the next steps:
fulvicin 250mg generic – griseofulvin 250mg cost gemfibrozil order
geinoutime.com
Zhu Houzhao는 “그는 매우 공개적으로 생각합니다, 아버지…”라고 말했습니다.
buy cheap forxiga – precose 25mg pills order acarbose generic
mantra 10 tadalafil tablets
vardenafil bayer
order generic dimenhydrinate 50mg – order dimenhydrinate online buy generic actonel for sale
buy vasotec 5mg online – purchase latanoprost generic buy zovirax eye drops for sale
most reliable online pharmacy viagra
online pharmacy 365 pills
order etodolac 600mg generic – generic etodolac brand pletal 100 mg
phuket pharmacy valium
order piroxicam 20mg for sale – generic feldene buy rivastigmine sale
geinoutime.com
다음날 아침 일찍 Fang Jifan은 Xiao Jing이 말한대로 행복하게 궁전에 들어갔습니다.
cialis online from us pharmacy
cancun pharmacy viagra
pakistan pharmacy valium
phentermine cost at pharmacy
Pin Up Azerbaycan ?Onlayn Kazino: Pin-Up Casino – pin-up360
Pin Up Azerbaycan: Pin Up – Pin Up
https://autolux-azerbaijan.com/# Pin Up
https://autolux-azerbaijan.com/# Pin Up Azerbaycan ?Onlayn Kazino
Pin-Up Casino: pin-up kazino – pin-up kazino
?Onlayn Kazino: Pin Up Kazino ?Onlayn – ?Onlayn Kazino
https://autolux-azerbaijan.com/# pin-up360
https://autolux-azerbaijan.com/# Pin-Up Casino
Pin Up Azerbaycan: pin-up 141 casino – pin-up kazino
pin-up 141 casino: Pin Up Azerbaycan ?Onlayn Kazino – pin-up 141 casino
Pin Up Kazino ?Onlayn: pin-up kazino – Pin-Up Casino
Pin Up: pin-up kazino – Pin Up Azerbaycan
Pin Up Azerbaycan: Pin-up Giris – pin-up kazino
https://autolux-azerbaijan.com/# Pin Up Kazino ?Onlayn
pin-up360: Pin-Up Casino – pin-up kazino
pin-up360: pin-up360 – Pin Up Kazino ?Onlayn
https://autolux-azerbaijan.com/# Pin Up Azerbaycan
Pin Up Azerbaycan: pin-up360 – Pin Up Kazino ?Onlayn
5 래빗스 메가웨이즈
따라서 북부 지방에서는 많은 사업가들이 구근을 고가에 사기 시작했습니다.
nootropil 800mg us – cheap sinemet 10mg sinemet 20mg cost
hydroxyurea over the counter – buy cheap crixivan order methocarbamol 500mg pills
999 슬롯
타타르인들은 매우 협조적인 것 같았고 순종적으로 야영을 시작했습니다.
purchase depakote for sale – topamax 100mg tablet topiramate 200mg pills
sm 슬롯
Hongzhi 황제는 Mou Bin을 쳐다 보며 깜짝 놀랐습니다. “뭐?”
Погрузитесь в вселенную “Хроники Безумного Макса” – кинолента ждет вас для просмотра в интернете.
立即访问Telegram官网,下载全球最受欢迎的即时通讯应用。Telegram以其高安全性和易用性闻名,提供文本、语音消息和视频通话等多种通讯方式。支持各种文件类型的分享,无论是照片、视频还是文档,都能快速发送。界面简洁,多语言支持,让您的通讯无国界限。现在就来体验!Visit the official Telegram website now and download the world’s most popular instant messaging app. Known for its high security and ease of use, Telegram offers various communication methods including text, voice messages, and video calls. It supports sharing of all file types, whether photos, videos, or documents, with fast delivery. The interface is clean and supports multiple languages, making your communication boundless. Experience it today! https://www.telegramkd.com zov5mg8dhl
buy disopyramide phosphate pill – buy cheap generic pregabalin pill thorazine 100mg
마블 슬롯
Zhu Houzhao는 갑자기 웃음을 멈추고 Fang Jifan을 바라 보았습니다.
spironolactone 100mg uk – order aldactone 25mg pill naltrexone 50 mg usa
buy cyclophosphamide pills – buy zerit sale vastarel medication
how to get flexeril without a prescription – flexeril 15mg over the counter cheap vasotec 5mg
zofran 4mg ca – purchase eldepryl purchase ropinirole
brand ascorbic acid – buy ferrous without a prescription prochlorperazine canada
how to buy durex gel – buy durex gel cheap xalatan eye drops
mexican pharmacy: mexican pharmacy – medication from mexico pharmacy
mexican pharmaceuticals online
http://cmqpharma.com/# pharmacies in mexico that ship to usa
buying prescription drugs in mexico online
Roulette is an easy game really, there is quite a bit of luck involved in the game, but if you have a good betting strategy you can win. To play roulette simply click the chips of your choice and place your bet on the designated area on the table. Now press the ‘spin’ button and this set the roulette wheel in motion. The small ball is dropped in and when the wheel comes to a stop the number on which the ball lands determines the winner. Gambling problem? Please contact the U.S. National Problem Gambling Helpline at 1-800-GAMBLER. NY 877-8-HOPENY or text HOPENY 46769. The casino world’s evolution never ceases. Nowadays, players have the chance to embrace flash casino options, offering a fresh gaming experience. Differing from classic online casinos, there’s no need to install extra software. Plus, an online casino consistently showcases seamless operational speed”.
https://front-wiki.win/index.php?title=Best_online_casino_to_win_big
Launched in 2020, El Royale has teamed up with RealTime Gaming (RTG) to offer over 140 mobile slots for gamers to enjoy. RTG is one of the biggest mobile casino software developers in the world, and is trusted in North America, Europe, and Asia for its high-graphic, low-glitch and safe casino gameplay. While El Royale does not offer a specific casino app for Florida residents, the site is fully optimized for in-browser mobile play. Being one of the top online casino software providers, Limeup experts also cover customers’ needs in slot, lottery, and bingo games of all complexity and requirements. The team offers a streamlined process for how to start an online casino question that guarantees customer satisfaction. For example, during the development of BingoBet, Limeup was tasked with creating the following features:
Live dealer casinos offer pretty much all the games you can find in a brick-and-mortar casino, plus some special games that are only available in the virtual environment. We’re bringing you the breakdown of the most popular live casino games you’ll come across on regulated US sites and sweepstakes casinos. In addition to live dealer game shows, you can play online live casino craps and a highest-card-wins game called Football Studio. Finally, many of the leading live dealer casinos now offer slots. That’s right, you can play online casino slots live via our computer or mobile device. If you’re wondering which live casino games are on offer, then there might be more than you expect. Live dealer casinos are becoming more adventurous in their game offerings as technology improves. Here are some of the most popular.
http://www.yumong.co.kr/bbs/board.php?bo_table=free&wr_id=205529
© 2024. Must be 21 or older. See Players Club for complete details. Management reserves all rights. Subject to expiration dates. Persons who have trespassed or been banned by the Seminole Tribe of Florida or those who have opted into the self-exclusion program are not eligible. If you or someone you know has a gambling problem, please call 1.888.ADMIT.IT. Stop by HollywoodCasino every month to receive free credits courtesy of the Hollywood Casino family. No matter what device you’re playing from, you can enjoy all your favorite slots on mobile. Find out how to get started. Hit a Jackpot in everyone’s favorite classic slots game! No matter what device you’re playing from, you can enjoy all your favorite slots on mobile. Find out how to get started. You love wilds? So do we. Check out this new classic slot for a some wild wins!
telegram下载:https://www.telegramjq.com 6TW4VNC
purchase rogaine – dutas sale order propecia online
telegram下载:https://www.tgxiazai.com EbLfpciD
通过爱思助手官网下载这款强大的苹果设备管理工具,享受便捷的数据管理和设备维护功能。爱思助手支持一键备份、恢复以及应用安装,帮助您高效管理设备空间和应用。适合所有iOS用户,特别是频繁需要数据处理的专业人士。https://i4.com.vn/ F2VKfHM7
buy arava 20mg – order alfacip buy cartidin cheap
telegram下载:https://www.telegramjq.com nKYjJ
buy verapamil 240mg without prescription – buy generic valsartan over the counter buy tenoretic no prescription
telegram下载:https://www.telegramjq.com RW9zN
atenolol over the counter – atenolol 100mg drug coreg us
Веном 3 онлайн https://bit.ly/venom-3-venom-2024
atorvastatin price – purchase vasotec online cheap purchase nebivolol sale
Worrisome chocolate thc has been quite the journey. As someone fervent on spontaneous remedies, delving into the to the max of hemp has been eye-opening. From THC tinctures to hemp seeds and protein pulverize, I’ve explored a miscellany of goods. Regard for the misunderstanding surrounding hemp, researching and consulting experts receive helped cross this burgeoning field. Inclusive, my live with hemp has been positive, offering holistic well-being solutions and sustainable choices.
通过爱思助手官网下载这款强大的苹果设备管理工具,享受便捷的数据管理和设备维护功能。爱思助手支持一键备份、恢复以及应用安装,帮助您高效管理设备空间和应用。适合所有iOS用户,特别是频繁需要数据处理的专业人士。https://i4.com.vn/ 7RBNS
telegram下载:https://www.tgxiazai.com 6jO6p
buy gasex for sale – gasex price cheap diabecon without prescription
Fatiguing https://www.nothingbuthemp.net/collections/thc-candy has been perfectly the journey. As someone rapier-like on unpretentious remedies, delving into the coterie of hemp has been eye-opening. From THC tinctures to hemp seeds and protein powder, I’ve explored a miscellany of goods. Regard for the disorder surrounding hemp, researching and consulting experts receive helped journey this burgeoning field. Entire, my sophistication with hemp has been favourable, sacrifice holistic well-being solutions and sustainable choices.
Fatiguing https://www.nothingbuthemp.net/collections/thc-lemonade has been somewhat the journey. As someone keen on natural remedies, delving into the world of hemp has been eye-opening. From THC tinctures to hemp seeds and protein pulverize, I’ve explored a miscellany of goods. In defiance of the confusion neighbourhood hemp, researching and consulting experts receive helped journey this burgeoning field. Entire, my live with hemp has been despotic, contribution holistic well-being solutions and sustainable choices.
Веном 3 кино https://bit.ly/venom-3-venom-2024
telegram下载:https://www.telegramjq.com IY2VE
Disquieting hemp products has been quite the journey. As someone rapier-like on natural remedies, delving into the in every respect of hemp has been eye-opening. From THC tinctures to hemp seeds and protein puissance, I’ve explored a type of goods. Teeth of the disorder neighbourhood hemp, researching and consulting experts own helped cross this burgeoning field. Inclusive, my sophistication with hemp has been positive, sacrifice holistic well-being solutions and sustainable choices.
cheap lasuna pills – generic himcolin purchase himcolin online
通过爱思助手官网下载这款强大的苹果设备管理工具,享受便捷的数据管理和设备维护功能。爱思助手支持一键备份、恢复以及应用安装,帮助您高效管理设备空间和应用。适合所有iOS用户,特别是频繁需要数据处理的专业人士。https://i4.com.vn/ GQJ3y
before and after french bob at indie salon @hairbylisastevens Bobs are fun, but can appear ‘boring’ when cut and styled simply with a blunt and untextured finish. Lip length Hailey’s bob haircut is often styled in a sleek and polished way, with a deep side part and a straight finish. She has also been seen wearing her bob with loose waves or curls for a more relaxed look. If you’re considering a bob haircut like Hailey’s, it’s crucial to choose a length and style that flatters your face shape and hair type. Be sure to consult with your stylist, who can help you achieve a look tailored to your type. This content can also be viewed on the site it originates from. 42. Fine Hair Layered Blonde Bob. This look demonstrates just how effective bob haircuts for fine hair can be when correct layering and shaping techniques are employed. The bright blonde is tasteful and sophisticated.
https://johnathanlrma393481.ambien-blog.com/32782968/1920-hairstyles-male
byunicia byunicia However, the hair shafts of the African volunteers did exhibit structural damage with evidence of longitudinal fissures, resulting in splitting of the hair shafts. The splitting was also associated with knot formation. Longitudinal fissures were not observed in the Caucasian or Asian hairs. It was also observed that many of the black African hairs (approximately 40%) were fractured with no attached root. There's a reason why XOXO Virgin Hair is widely used by the hairstylists of Mary J. Blige, MJ Rodriguez, and Taraji P Henson. The hair is just that good. In 2014, founder Stephanie Nolan launched the brand. Human hair lace-frontal wigs, bundles, frontals, and closures are available in three textures: straight, wavy, or curly. All the hair comes in a natural black shade that can be bleached and dyed. Go short with 10-inch extensions or embrace length with the 28-inch options.
Depending on the complexity of your desired hairstyle, you may want to bring a stylist on board. Even simple updos and braids can sometimes be tricky to do by yourself. To get out of this slump, I decided to look up some quick and easy hairstyles for the long, beautiful tresses. Read on to find out more! One of the best ridiculously easy hairstyles for schools, the rose braid looks absolutely amazing. It can also be adapted for medium-length hair, which makes it a key look for busy days. You may seem to be facing a lack of styling options by having a pixie cut. However, all you have to do is tousle your hair a bit and add some hairspray for a picture-perfect school hairstyle. Step 1: Part your hair to one side. Using your tail comb, create a section perpendicular to the part all the way down to the hairline behind your ear. Clip back the rest of your hair to keep it out of the way for now.
https://waylonxvoj953173.affiliatblogger.com/79899363/essence-volume-booster-lash-primer-7ml
A fade is essentially the way that the hair tapers from down the back and sides. This can be aggressive but can also be quite subtle depending on the cut. A fade isn’t a haircut in itself but a component that accompanies the hairstyle as a whole. The high and tight recon hairstyle is an exciting version of high and tight. The recon is styled at the top on the scalp, creating a landing strip looking like a rectangle. The Quiff with Sharp Fade combines classic and contemporary elements. This style features a voluminous quiff on top, paired with a sharply faded cut on the sides and back. Perfect if you can’t bear to opt for a complete low cut and still want to maintain a sense of style and personality. When it comes to hairstyles for men, military haircuts are the most common because we can see it everywhere. There are different kinds of military haircuts including Induction Cut, Burr Cut, Butch Cut, Crew Cut, Brush Cut, Ivy League, Fade, High and Tight, High and Tight Recon, Regulation Cut, Flat Top, Undercut and many more.
cheap noroxin without prescription – confido for sale online buy confido
I’ve been using cbd gummies and melatonin for over two years today, and the knowledge has been transformative. CBD lubricant has notably improved my snooze quality and reduced my anxiety levels. The edibles are a commodious and enjoyable passage to incorporate CBD into my continually routine. I rate the genuine propositions to wellness that CBD offers, and I’ve experienced no adverse side effects. Importantly commend in behalf of those seeking a sensible selection to time-honoured medications.
I’ve been using https://greenrevolutioncbd.com/choose-experience/cbd-for-focus/ for through two years at this very moment, and the sophistication has been transformative. CBD lubricant has markedly improved my slumber quality and reduced my dread levels. The edibles are a convenient and enjoyable way to merge CBD into my continually routine. I rate the spontaneous nearly equal to wellness that CBD offers, and I’ve capable no adverse side effects. Importantly recommend for the sake of those seeking a commonplace selection to traditional medications.
I’ve been using https://greenrevolutioncbd.com/kosher-cbd/ for through two years at this very moment, and the sophistication has been transformative. CBD grease has evidently improved my sleep calibre and reduced my worry levels. The edibles are a advantageous and enjoyable way to amalgamate CBD into my continually routine. I appreciate the talent propositions to wellness that CBD offers, and I’ve sagacious no adverse side effects. Importantly recommend for those seeking a simpleton different to unwritten medications.
I’ve been using cbd products instead of throughout two years now, and the experience has been transformative. CBD unguent has markedly improved my snooze je sais quoi and reduced my longing levels. The edibles are a advantageous and enjoyable personality to mix CBD into my commonplace routine. I appreciate the spontaneous approach to wellness that CBD offers, and I’ve knowledgeable no adverse side effects. Tremendously approve for the sake of those seeking a natural different to traditional medications.
I’ve been using CBD gummies in return or a year today, and I can’t believe how much they’ve improved my person like cbd gummy ! The flavors are at rest appetizing, making it a delightful division of my daily routine. My dread and stress levels have significantly decreased, and my catch quality has improved tremendously. I wake up premonition more refreshed and energetic. However, I’ve noticed a bit of drowsiness during the daytime, and I order the effects lasted a whit longer. Despite these trivial issues, I powerfully advocate these CBD gummies after anyone looking to enrich their well-being normally!