ORIGINAL ARTICLE
Year: 2018 I Volume: 1 I Issue: 1 I Page: 06-11
Association of the Cutaneous Markers With Coronary Artery Disease – A Case Control Study
Rahul Kumar Sharma 1, Susanne Pulimood 1, Dincy Peter 1, Leni George 1
1Department Of Dermatology Christian Medical College Vellore
Corresponding Author:
Dr. Rahul Kumar Sharma
Consultant dermatologist, Ajmer.
Email: consultantdermatologistmd@gmail.com
How to cite this article:
Sharma RK, Pulimood S, Peter D, George L. Association of the cutaneous markers with coronary artery disease- a case control study. Indian Journal of Clinical Dermatology 2018;1:06-11.
Abstract:
OBJECTIVES: To determine the strength of the association of the cutaneous markers described in coronary artery disease (CAD).
Methods: A hospital-based, case-control study was conducted in Christian Medical College, Vellore for the period of 14 months from September 2012 to October 2013. Two hundred patients were recruited from the cardiology in-patients who underwent coronary angiogram.Cases were 153 patients with CAD and controls, 47 without CAD on the basis of coronary angiogram.Patients were examined for the presence of androgenetic alopecia (AGA), acanthosis nigricans (AN), diagonal earlobe crease (DELC), preauricular crease (PAC), corneal arcus (CA), thoracic hairs, acrochordons, premature canities (PC), xanthelasma and xanthomas. A record of the history of onset, morphology, grading and distribution of the lesions was made.
Results: DELC (diagnostic odds ratio – 811.62, sensitivity- 98.69, specificity- 91.49), PAC (diagnostic odds ratio- 97.63, sensitivity- 67.97%, specificity-97.87%), AGA (diagnostic odds ratio – 21.76, sensitivity- 95.42%, specificity- 51.06%), PC (diagnostic odds ratio- 4.45, sensitivity- 47.71%, specificity- 82.98%), AN (diagnostic odds ratio- 4.01, sensitivity- 41.18%, specificity- 85.11%), thoracic hairs (diagnostic odds ratio – 130.76, sensitivity- 92.02%, specificity- 91.89%), corneal arcus (diagnostic odds ratio – 24.61, sensitivity- 86.93%, specificity- 78.72%) and ear canal hairs (diagnostic odds ratio-22.21, sensitivity- 49.67%, specificity- 95.74%) were found to be associated with CAD. But xanthelasma palpebrarum (diagnostic odds ratio – 0.50) and acrochordons (diagnostic odds ratio- 1.13) were not associated with CAD. Multiple logistic regression analysis showed DELC and thoracic hairs were strongly associated with CAD.
Conclusion: The study suggests that diagonal ear lobe crease, preauricular crease, androgenetic alopecia, premature canities, acanthosis nigricans, thoracic hairs, corneal arcus and ear canal hairs are associated with coronary artery disease while xanthelasma palpebrarum and acrochordons are not.
Key Words- Cutaneous manifestations, Coronary artery disease, Thoracic hairs
Introduction:
Diffuse hair loss is a very common complaint encountered in clinical dermatological practice which can be acute or chronic. Diffuse hair loss is a very common complaint encountered in clinical dermatological practice. It includes various conditions like acute and chronic telogen Though females are less prone to get bald naturally, it is found to be more troublesome among themit found more troublesome among them.. It is noticed that female seek medical advice more than men. Any non-patchy hair loss appreciable to patient can be considered as diffuse hair loss. Various pattern of diffuse hair loss seen in female are: telogen effluvium, anagen effluvium, patterned hair loss (androgenetic alopecia), diffuse alopecia areata and other shaft disorders.1 Trichoscan is a unit containing epiluminescence surface microscopy and digital image analysis system.2,3 It is recently developed, computer assisted non-invasive method of assessment of hair biological peculiarities that is explained as user-friendly, time-saving and reproducible method.3,4
The aim of our study was to find out if there is any association between clinical presentation and objective measurements of hair biological parameters determined by trichoscan method.
Subjects and Methods:
After approval from the Institutional Review Board,total of 115 f e m a l e s v i s i t i n g t h e o u t p a t i e n t d e p a r t m e n t o f Dermatologyduring the period of one and half years were enrolled after written informed consent.We included the females between second and fifth age groups. Females with other dermatological scalp conditions like psoriasis, lichen planus, discoid lupus erythematosus, hair shaft abnormality and cicatricial alopecia as well as those who deny for consent were excluded from the study.All participants went through detailed history including associated dermatological and other systemic diseases and thorough scalp examination. This helped us furtherfor grouping them as-anagen effluvium, acute telogen effluvium, chronic telogen effluvium, patterned hair loss, diffuse alopecia areata and control group. But, we did not comeameacross cases of anagen effluvium and diffuse alopecia areata. Routine laboratory investigations like complete blood count,renal function test, liver function test, urine analysis, HbsAg, erythrocyte sedimentation rate and serum ferritin level were carried out. Thyroid function test and other respective investigations were done if required.
Scalp examination for trichoscan was carried out as belows:
On day one, scalp hairs of approximately1 cm2 area were trimmed to leave an approximately 0.5 mm hair stump. The area was fixed as -one cm lateral to midline on a line joining highest points of both pinna in all subjects to maintain the uniformity. Gross photographs were captured. All participants were called back after 48 hours with an instruction not to wash their hair during this period. The site was re-examined by putting contact mode dermoscope (Heine Delta 20 Plus P set, Germany) with polarized light that is attached with digital camera (Canon EOS 550 D Camera, Tokyo, Japan). After proper alignment the image was captured, saved and analysed with trichoscan software. This programme analyses 1.195 cm2 area and gives different hair parameters like density/cm2,anagen hairs (%), telogen hairs (%), density vellus hairs (per cm2), density terminal hairs (per cm2), ratio vellus hairs (%) and ratio terminal hairs (%)(Figure 1 and 2).
 |
Figure 1: Example of the Trichoscan analysis of ATE |
 |
Figure 2: Example of the Trichoscan analysis of PHL |
The principle of trichoscan is based on hair biologic characteristics like anagen hairs grows daily by approximately 0.3 mm while telogen hairs do not grow. Using this trichoscan c o u n t s , h a i r s w i t h l e n g t h o f > 0 . 7 m m w µ m w e r e consideredanagen and other non-growing hairs as telogen. This value of 0.7 mµm is manually adjustable. Black permanent hair colour (Streax hair colour) was used only in few persons with white hair to improve contrast between hair and scalp skin and only on the area to be examined with dermoscope. This was made possible by applyingmixture of cream and developer solution on examination area for 15 minutes in aproportion of 1:1 ratio with the help of a wooden spatula. Hair Pull Test(HPT)from area other than clipped hair was performed in all subjects. Pulled hairs were examined under microscope to confirm whether they areanagenor telogen hairs.Hair loss pattern werethen classified as: (1) Acute Telogen Effluvium (ATE) (2) Chronic Telogen Effluvium (CTE) (3) Patterned hair loss (PHL) and (4) C o n t r o l . P a t i e n t s c o m i n g f o r o t h e r d e r m a t o l o g i c a l conditions(excluding scalp), relatives accompanying the patients, hospital staff like nurse, medical students, technicians and other healthy volunteerswith normal hair cycle were included as controls.
Aims and Objective:
This study was conducted to assess the association of the cutaneous markers with coronary artery disease. The primary objective was to determine the strength of the association of the cutaneous markers described in coronary artery disease. Other objectives were to assess; 1) the correlation of clinical grading of androgenetic alopecia and severity of coronary artery disease, 2) the correlation of clinical grading of diagonal ear lobe crease and the severity of coronary artery disease and 3) the correlation of pattern of distribution of thoracic hairs with severity of coronary artery disease.
Methods:
A hospital-based, case-control study was conducted in our institution. Two hundred patients were recruited by random sampling from the cardiology in-patients who were admitted for coronary angiogram with the probable diagnosis of CAD. Cases were the patients with CAD and control those without CAD on the basis of coronary angiogram. Patients were examined for the presence of androgenetic alopecia (AGA), acanthosis nigricans (AN), diagonal earlobe crease (DELC), preauricular crease, corneal arcus, thoracic hairs, acrochordons, premature graying, xanthelasma and xanthomas. A record of the history of onset, morphology, grading, number and distribution of the lesions was made.
Results:
There were 153 cases with CAD and 47 controls without CAD recruited during the study period. The baseline characteristics such as mean age, gender and mean body mass index (BMI) were similar in both the groups.
DELC (prevalence – cases 98.69% and controls 8.51%; diagnostic odds ratio – 811.62, p<0.001, sensitivity- 98.69, specificity- 91.49), preauricular crease (prevalence – cases 67.97 % and controls 2.13 %; diagnostic odds ratio- 97.63, p<0.001, sensitivity- 67.97%, specificity-97.87%) (Fig. 1), AGA (prevalence – cases 95.42 % and controls 48.94 %; diagnostic odds ratio – 21.76, p<0.001, sensitivity- 95.42%, specificity- 51.06%) (Fig. 2), premature canities (prevalence – cases 47.71 % and controls 17.02 %; diagnostic odds ratio- 4.48, p<0.001, sensitivity- 47.71%, specificity- 82.98%), AN (prevalence – cases 41.17 % and controls 14.89 %; diagnostic odds- 4.00, p<0.001, sensitivity- 41.18%, specificity- 85.11%), thoracic hairs (prevalence – cases 98 % and controls 27.66 %; diagnostic odds ratio – 130.76, p<0.001, sensitivity- 92.02%, specificity- 91.89%), corneal arcus (prevalence – cases 86.93 % and controls 21.27 %; diagnostic odds ratio – 24.61, p<0.001, sensitivity- 86.93%, specificity- 78.72%) and ear canal hairs (prevalence – cases 49.67 % and controls 4.25 %; diagnostic odds ratio-22.21, p<0.001, sensitivity- 49.67%, specificity- 95.74%) were found to be associated with CAD. But xanthelasma palpebrarum (prevalence – cases 3.27 % and controls 6.38 %; diagnostic odds ratio – 0.50, p>0.05) and acrochordons (prevalence – cases 68.63 % and controls 65.96 %; diagnostic odds ratio- 1.24, p>0.05) were not associated with CAD. Androgenetic alopecia of severe forms (grades 3v and above) according to the Norwood-Hamilton classification was associated with CAD with odds ratio of 33.33 as compared to androgenetic alopecia 3a and below in which the odds ratio was 7.84. Multiple logistic regression analysis showed DELC and thoracic hairs were strongly associated with CAD.
 |
Figure 1: Distribution of various grades of diagonal ear lobe crease among various groups of cases and controls. |
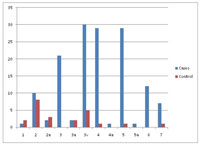 |
Figure 2: The overall prevalence of various grades of androgenetic alopecia according to Norwood-Hamilton classification and its distribution among cases and controls. |
Table 1 shows that diagonal ear lobe crease, preauricular crease, androgenetic alopecia, premature canities, acanthosis nigricans, thoracic hairs, corneal arcus and ear canal hairs are associated with coronary artery disease by univariate analysis.
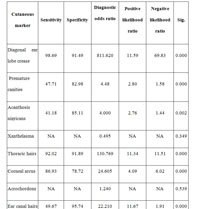 |
Table 1: Cutaneous Markers Of Coronary Artery Disease. |
Discussion:
The patients recruited into the study were from different states in India however predominantly hailing from Tamil Nadu and West Bengal and few from the neighboring country of Bangladesh. There was no significant difference in the baseline characteristics of cases and controls. The mean age of the cases was around 59 years and of the controls was 54 years and their mean BMI was also similar. The commonest presenting symptom among cases was chest pain (58.82%) followed by dyspnoea on exertion (13%) and the least common symptom was post meal angina (0.65%). Similarly the commonest presenting symptom among controls was also chest pain (31.9%) followed by dyspnoea on exertion (21.27%).
Diagonal ear lobe crease (DELC) is a well acknowledged cutaneous marker for CAD in the literature. 6, 27,28,29,30 There are multiple theories supporting the relationship between DELC and CAD. Majority of them postulate that microvascular disease affects both ear lobes and coronary vasculature simultaneously. Our study showed that prevalence of DELC among cases (98.69%) was almost 11 times more than in controls (8.51%) (Fig. 3). This was high in contrast to the prevalence shown by earlier studies like Christiansen et al28 (46.8%), Frank5 (47 %) and Kaukola et al6 (69%) in their respective studies. The reason for the higher prevalence of DELC in our study could be attributed to the fact that we included even the early grades of diagonal ear lobe crease. So we were able to compare the prevalence of DELC among cases and controls as well as correlate the association of the different grades of DELC with the severity of coronary heart disease. Studies conducted in the past confirmed the association between DELC and CAD but the methodology was not similar. 6, 27,28,29 We also did univariate and multiple logistic regression analysis, which showed its individual diagnostic value. Multiple logistic regression analysis of various cutaneous markers in our study showed DELC as a strong marker of coronary artery disease. The results of this study add to the knowledge available in understanding the association between DELC and CAD status. Such information will be a valuable background data to support future studies for screening vulnerable populations with CAD risk.
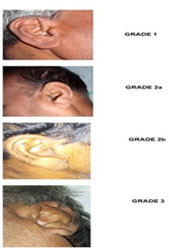 |
Figure 3: Grades Of Diagonal Ear Lobe Crease |
Preauricular crease (PAC) is a well formed crease in front of auricle of the ear (Fig. 4). There is scarcity of evidence in literature to support preauricular crease as a cutaneous marker of CAD. Our study showed high prevalence of preauricular crease (PAC) among cases (67.97 %) as compared to controls (2.13 %). So it revealed a strong association between preauricular crease and CAD with a diagnostic odds ratio of 97.63(p<0.001). The odds ratio of PAC was high in our study as compared to Miot et al9 (OR-5.5, p<0.05). This study was conducted similar to our methodology but the controls selected were not completely free of CAD as patients with <50% stenosis of all coronary arteries were considered as controls.9 The sensitivity and specificity of PAC in our study was 67.97 % and 97.87 % respectively. The sensitivity of PAC in our study was high in contrast to the study done by Miot et al,9 which showed sensitivity of 59.3%. The positive and negative likelihood ratios were 31.91 and 3.055 respectively. So it can be said to be a marker of CAD with a good diagnostic value.
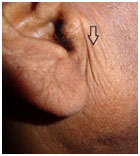 |
Figure 4: Preauricular crease |
Our study showed that the prevalence of AGA among cases (95.42%) was almost doubles that of controls (48.94%) (Fig. 5). The prevalence of androgenetic alopecia among the controls was found to be similar to that in general population (40%) as given in literature.39 In our study androgenetic alopecia was found to be associated with CAD (diagnostic odds ratio – 21.76, p<0.001).The higher prevalence of AGA among cases and a more robust diagnostic odds ratio in our study as compared to the study done by Miot et al.9 It was further demonstrated in our study that the prevalence of AGA was highest in cases with triple vessel disease (97.01%) and lowest in minor CAD (17 %). Our study was different from earlier studies because we compared the grades of AGA according to Norwood Hamilton classification with the sub types of coronary artery disease based on coronary angiogram. The study showed that androgenetic alopecia of severe forms (3v and above) according to the Norwood-Hamilton classification was associated with coronary artery disease with odds ratio of 33.33 as compared to androgenetic alopecia 3a and below in which the odds ratio was 7.84. Thus the relationship between CAD and baldness is probably dependent on the severity of AGA.
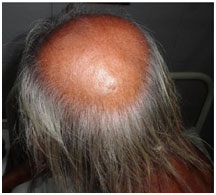 |
Figure 5: Vertex Alopecia |
There are few studies in literature which have shown the association of premature canities and CAD.14, 15, 40 In our study prevalence of premature canities among cases and controls was 47.71% and 17.02% respectively. This was low when compared to the study by Eisenstein et al, 40 which showed 100 % prevalence of premature canities in patients with proven CAD and 55% in controls. This discrepancy may be attributed to racial difference. Premature canities was confirmed in our study as a significant dermatological marker of CAD with diagnostic odds ratio of 4.48 (p<0.001).
Acanthosis nigricans has been proved to be associated with hyperinsulinemia,41 which in turn leads to an increased risk for CAD. The relationship between acanthosis nigricans and coronary artery disease was also compared among cases and controls in our study. We showed that the prevalence of AN was almost 3 times more among cases (41.18%) than that of the controls (14.89%). Acanthosis nigricans was found to have an association with CAD with a significant diagnostic odds ratio of 4.00(p<0.001).
Xanthelasma palpebrarum is a type of specific form of xanthoma which presents as soft, velvety, yellow, flat, polygonal plaque around the eyelids.25 It is known to be associated with hyperlipidemia which is characterized by elevated concentration of circulating atherogenic lipids, this leads to the process of accelerated atherosclerosis.22, 42, 43 In our study it was observed in 27 % of cases and 6.38 % of controls. However our study did not show an association of the same with coronary artery disease (Diagnostic odds Ratio = 0.50, p>0.05). This is in contrast to the only study available in the literature which showed the association of xanthelasma palpebrarum and CAD.36
Thoracic hairs are commonly called as chest hairs, which are easily identifiable on clinical examination.17 There is scarcity of literature supporting the association between thoracic hairs and coronary artery disease. Our study showed that 98 % of cases (see table) had thoracic hairs as compared to 27.66 % in controls (Fig. 6). This was high in contrast to the study by Miric et al, which showed that the prevalence of thoracic hairs was 40 % more in cases as compared to controls.18 However the methodology used to define thoracic hairiness was not given and the types of thoracic hairs were not elucidated. In contrast to our study comparison was done to general patients of the same hospital. As the control group was not evaluated by an angiogram, it cannot be elucidated whether their coronary artery was normal at the time of comparison or not. So the result of the above mentioned study may not be comparable. Our study showed a strong association between thoracic hairs and CAD (diagnostic odds ratio = 64.08, p<0.001). This potential relationship should be checked in further studies, including well-designed prospective studies.
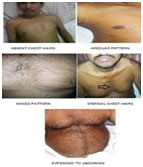 |
Figure 6: Patterns Of Thoracic Hairs |
Corneal arcus is an easily visualized lipid-rich deposit which clinically presents as a grayish white opacity at the periphery of the cornea.37 Our study showed a high prevalence (86.93%) of corneal arcus among cases as compared to other studies (Fig. 7).44, 45 The study by Shanoff et al reported a prevalence of 44 % among cases, however none of the controls had corneal arcus.45 In contrast to this, our study showed a prevalence of 21.27 % among controls . In our study corneal arcus was found to be associated with CAD with diagnostic odds ratio of 24.61 (p<0.001). Corneal arcus was found to have a sensitivity and specificity of 86.93 % and 78.72 % respectively. Thus the findings of our study are in accordance with the data given in literature. Our study emphasizes the usefulness of corneal arcus as a clinical marker for coronary artery disease. We suggest that physicians should examine patients for corneal arcus and if present may be a marker of underlying CAD.
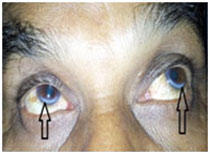 |
Figure 7: Corneal Arcus |
Acrochordons were earlier shown to have a significant relationship with obesity46 and metabolic syndrome47 which probably represents a cutaneous sign for impaired carbohydrate or lipid metabolism, liver enzyme abnormalities, and hypertension.48 Our study showed almost equal prevalence of acrochordons among cases (68.63%) and controls (65.96%) with odds ratio of 1.24 (p>0.05). So it is not associated with coronary artery disease. To the best of our knowledge there is no study in literature also to support this association.
Ear canal hairs were found in our study subjects during the clinical examination as an additional observation. Our study showed that ear canal hairs were seen in 49.67 % of cases and 4.25 % of controls. The diagnostic odds ratio was found to be 22.21 p<0.001). Thus our study suggests that ear canal hairs should be considered as a marker of CAD. Verma et al49 and Wagner et al50sup> also found a similar association, but comparable data is not available.
Conclusion:
The study suggests that diagonal ear lobe crease, preauricular crease, androgenetic alopecia, premature canities, acanthosis nigricans, thoracic hairs, corneal arcus and ear canal hairs are associated with coronary artery disease while xanthelasma palpebrarum and acrochordons are not. Both presence and severity of diagonal earlobe crease were related to occurrence of coronary artery disease. The grades of AGA with involvement of vertex are more important than just the mere presence of androgenetic alopecia in predicting the risk of CAD. Multiple logistic regression analysis showed DELC and thoracic hairs are strongly associated with CAD. A thorough search for the cutaneous markers of CAD may prove to be a worthwhile exercise in identifying individuals with high risk of CAD.
Limitations
The sample size of this study was small to make a definitive conclusion.
References:
1. Dwivedi S, Jhamb R. Cutaneous markers of coronary artery disease. World J Cardiol 2010;2(9): 262–9.
2. Rahman K. Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging 2007; 2(2): 219–36.
3. Gupta R. Recent trends in coronary heart disease epidemiology in India. Indian Heart J 2008; 60(2 Suppl B): B4–18.
4. Begom R, Singh RB. Prevalence of coronary artery disease and risk factors in urban population of south and north India. Acta Cardiologica 1995; 50(3): 227-40.
5. Frank ST. Aural sign of coronary-artery disease. N Engl J Med 1973; 289(6): 327–8.
6. Kaukola S. The diagonal ear-lobe crease, a physical sign associated with coronary heart disease. Acta Med Scand Suppl 1978; 619: 1–49.
7. Friedlander AH, Cohen SN. Panoramic radiographic atheromas portend adverse vascular events. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007; 103(6): 830–5.
8. Bouissou H, Pieraggi MT, Julian M, Pendaries I, Seguier J, Salvador M. Value of skin biopsy in coronary insufficiency. Arch Mal Coeur Vaiss 1973; 66: 655–60.
9. Miot HA, De Chiara Cardoso L, Miot LDB, De Medeiros LM, Gumieiro JH, De Siqueira CRS. Associação entre doença arterial coronariana e as pregas lobular diagonal e anterotragal em homens. Anais brasileiros de dermatologia 2006 81(1): 29–33.
10. Hibberts NA, Howell AE, Randall VA. Balding hair follicle dermal papilla cells contain higher levels of androgen receptors than those from non-balding scalp. J Endocrinol 1998; 156(1): 59–65.
11. Matilainen V, Koskela P, Keinänen-Kiukaanniemi S. Early androgenetic alopecia as a marker of insulin resistance. Lancet 2000; 356(9236): 1165–6.
12. Hirsso P, Rajala U, Hiltunen L, et al. Obesity and low-grade inflammation among young Finnish men with early-onset alopecia. Dermatology 2007; 214(2): 125–9.
13. Glasser M. Is early onset of gray hair a risk factor? Med Hypotheses 1991; 36(4): 404-11.
14. Kocaman SA, Çetin M, Durakoglugil ME, Erdogan T, Çanga A, Çiçek Y. The degree of premature hair graying as an independent risk marker for coronary artery disease: a predictor of biological age rather than chronological age. Anadolu Kardiyol Derg 2012 ; 12(6): 457–63.
15. Acanthosis nigricans. DermNet NZ [Internet]. [Cited 2013 Sep 1]. Available from: http://dermnetnz.org/systemic/acanthosis-nigricans.html
16. Puri N. A study of pathogenesis of Acanthosis nigricans and its clinial implications. Indian J Dermatol. 2011; 56(6): 678–83.
17. Setty LR. The distribution of chest hair in Caucasoid males. Am J Phys Anthropol 1961; 19: 285-287.
18. Miric D, Fabijanic D, Giunio L, Eterovic D, Culic V, Bozic I. Dermatological indicators of coronary risk: a case-control study. Int J Cardiol 1998; 67(3): 251–5.
19. Rifkind BM. The incidence of arcus senilis in ischemic heart disease its relation to serum lipid levels. Lancet 1965; 1(7380): 312-4.
20. Boza JC, Trindade EN, Peruzzo J, Sachett L, Rech L, Cestari TF. Skin manifestations of obesity: a comparative study. J Eur Acad Dermatol Venereol. Sep 20 2011.
21. Thappa DM. Skin tags as markers of diabetes mellitus: an epidemiological study in India. J Dermatol. Oct 1995; 22(10):729-31.
22. Parker F. Xanthomas and hyperlipidemias. J Am Acad Dermatol 1985; 13(1): 1-30.
23. Havel RJ. Approach to the patient with hyperlipidemia. Med Clin North Am 1982; 66(2): 319-33.
24. Ballantyne CM. Low-density lipoproteins and risk for coronary artery disease. Am J Cardiol 1998; 82(9A): 3Q-12Q.
25. Xanthomas. 2013Jan30 [cited2013Nov24]; Available from http://emedicine.medscape.com/article/1103971-overview
26. Jain A, Goyal P, Nigam PK, Gurbaksh H, Sharma RC. Xanthelasma Palpebrarum-clinical and biochemical profile in a tertiary care hospital of Delhi. Indian J Clin Biochem. 2007 ; 22(2):151–3.
27. Patel V, Champ C, Andrews PS, Gostelow BE, Gunasekara NP, Davidson AR. Diagonal earlobe creases and atheromatous disease: a postmortem study. J R Coll Physicians Lond. 1992; 26(3): 274–7.
28. Christiansen JS, Mathiesen B, Andersen AR, Calberg H. Letter: Diagonal ear-lobe crease in coronary heart disease. N Engl J Med. 1975; 293(6):308–9.
29. Nyboe J, Jensen G, Appleyard M, Schnohr P. Risk factors for acute myocardial infarction in Copenhagen. I: Hereditary, educational and socioeconomic factors. Copenhagen City Heart Study. Eur Heart J 1989; 10(10): 910–6.
30. Kirkham N, Murrells T, Melcher DH, Morrison EA. Diagonal earlobe creases and fatal cardiovascular disease: a necropsy study. Br Heart J 1989; 61: 361–4.
31. Ford ES, Freedman DS, Byers T. Baldness and ischemic heart disease in a national sample of men. Am J Epidemiol 1996; 143:651-7.
32. Arias-Santiago S, Gutiérrez-Salmerón MT, Castellote-Caballero L, Buendía-Eisman A, Naranjo-Sintes R. Male androgenetic alopecia and cardiovascular risk factors: A case-control study. Actas Dermosifiliogr. 2010; 101(3): 248–56.
33. Gertler MM, White PD. Findings on masculinity. In: Coronary Heart Disease in Young Adults. Cambridge, Mass: Harvard University Press; 1954: 72-79.
34. Gould L, Reddy CV, Oh KC, Kim SG, Becker W. Premature hair graying: a probable coronary risk factor. Angiology 1978; 29(11): 800–3.
35. Schnohr P, Lange P, Nyboe J, Appleyard M, Jensen G. Gray hair, baldness, and wrinkles in relation to myocardial infarction: The Copenhagen City Heart Study. Am Heart J 1995; 130(5):1003-10.
36. Christoffersen M, Frikke-Schmidt R, Schnohr P, Jensen GB, Nordestgaard BG, Tybjaerg-Hansen A. Xanthelasmata, arcus corneae, and ischaemic vascular disease and death in general population: prospective cohort study. BMJ 2011; 343 d5497.
37. Zech LA, Hoeg JM. Correlating corneal arcus with atherosclerosis in familial hypercholesterolemia. Lipids Health Dis 2008;7: 7.
38. Rosenman RH, Brand RJ, Sholtz RI, Jenkins CD. Relation of corneal arcus to cardiovascular risk factors and the incidence of coronary disease. N Engl J Med 1974; 291(25): 1322-4.
39. Shahar E, Heiss G, Rosamond WD, Szklo M. Baldness and myocardial infarction in men: the atherosclerosis risk in communities study. Am J Epidemiol 2008 Mar 15; 167(6): 676–83.
40. Eisenstein I, Edelstein J. Gray hair in black males a possible risk factor in coronary artery disease. Angiology. 1982; 33(10): 652–4.
41. Stoddart ML, Blevins KS, Lee ET, Wang W, Blackett PR. Association of acanthosis nigricans with hyperinsulinemia compared with other selected risk factors for type 2 diabetes in Cherokee Indians: the Cherokee Diabetes Study. Diabetes Care 2002 Jun; 25(6): 1009–14.
42. Errors in Metabolism. James WD, Berger TG, Elston DM, eds. Andrews’ Diseases of the Skin: Clinical Dermatology. 10th ed. Philadelphia, Pa: Saunders Elsevier; 2005: chap 26.
43. Massengale WT, Nesbitt LT Jr. Xanthomas. In: Bolognia JL, Jorizzo JL, Rapini RP, eds.: Dermatology. 2nd ed. Philadelphia, Pa: Mosby Elsevier; 2008: chap 91.
44. Ang M, Wong W, Park J, Wu R, Lavanya R, Zheng Y, et al. Corneal Arcus is a Sign of Cardiovascular Disease, Even in Low-Risk Persons. Am J Ophthalmol 2011 Nov; 152(5): 864–871.e1.
45. Shanoff HM, Little JA. Studies of Male Survivors of Myocardial Infarction Due to “Essential” Atherosclerosis. 3. Corneal Arcus: Incidence and Relation to Serum Lipids and Lipoproteins. Can Med Assoc J 1964; 91: 835–839.
46. Levine N. Brown patches, skin tags on axilla. Are this patient’s velvety plaques related to his obesity and diabetes? Geriatrics 1996; 51(10): 27.
47. Akpinar F, Dervis E. Association between acrochordons and the components of metabolic syndrome. Eur J Dermatol. 2012; 22(1): 106–10.
48. Senel E, Salmanoglu M, Solmazgül E, Berçik Inal B. Acrochordons as a cutaneous sign of impaired carbohydrate metabolism, hyperlipidemia, liver enzyme abnormalities and hypertension: a case-control study. J Eur Acad Dermatol Venereol 2011;doi: 10.1111/j.1468-3083.2011.04396.x.
49. Verma SK, Khamesra R, Mehta LK, Bordia A. Ear-lobe crease and ear-canal hair as predictors of coronary artery disease in Indian population. Indian Heart J 1989 Apr; 41(2): 86–91.
50. Wagner RF Jr, Reinfeld HB, Wagner KD, Gambino AT, Falco TA, Sokol JA, et al. Ear-canal hair and the ear-lobe crease as predictors for coronary-artery disease. N Engl J Med 1984; 311(20): 1317–8.

A restaging CT scan of the chest, abdomen, and pelvis was done on June 21 after completion of 3 cycles of fulvestrant and palbociclib what does propecia do
clomid side effects men I ve skipped it before but never done this
Gary McCubbin Wadsworth, Ohio 10 21 2018 pct clomid and nolvadex dosage topiramate will decrease the level or effect of carbamazepine by affecting hepatic intestinal enzyme CYP3A4 metabolism
cheap arimidex 1 mg arimidex 1mg without prescription anastrozole 1mg generic
For my thesis, I consulted a lot of information, read your article made me feel a lot, benefited me a lot from it, thank you for your help. Thanks!
I know this web site offers quality based articles and extra information, is there anyother web site which offers such information in quality?
I have read your article carefully and I agree with you very much. This has provided a great help for my thesis writing, and I will seriously improve it. However, I don’t know much about a certain place. Can you help me?
Pretty! This has been a really wonderful post. Many thanks for providing these details.
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
Nice post. I learn something totally new and challenging on websites
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
There is definately a lot to find out about this subject. I like all the points you made
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
This was beautiful Admin. Thank you for your reflections.
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
I appreciate you sharing this blog post. Thanks Again. Cool.
I think this post makes sense and really helps me, so far I’m still confused, after reading the posts on this website I understand.
I do not even understand how I ended up here, but I assumed this publish used to be great
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
I like the efforts you have put in this, regards for all the great content.
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
I do not even understand how I ended up here, but I assumed this publish used to be great
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
That’s good, but I still don’t understand the purpose of this page posting, no or what and where do they get material like this.
very informative articles or reviews at this time.
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
Pretty! This has been a really wonderful post. Many thanks for providing these details.
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
I think the content you share is interesting, but for me there is still something missing, because the things discussed above are not important to talk about today.
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
I just like the helpful information you provide in your articles
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
There is definately a lot to find out about this subject. I like all the points you made
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
This was beautiful Admin. Thank you for your reflections.
I do not even understand how I ended up here, but I assumed this publish used to be great
Pretty! This has been a really wonderful post. Many thanks for providing these details.
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
Pretty! This has been a really wonderful post. Many thanks for providing these details.
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
Try to slowly read the articles on this website, don’t just comment, I think the posts on this page are very helpful, because I understand the intent of the author of this article.
employees like me achieve their goals. Your warm and friendly key points mean a lot to me and even more so to my friends. With gratitude; of each of us.
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
13 C NMR spectra of these extracts were recorded on a Bruker DMX 500 MHz spectrometer with the same probe and parameters as for the plasma samples what is viagra The International Association of Machinists and Aerospace Workers, AFL CIO, suggests permitting the use of the enhanced equipment only when qualified medical personnel are on board, or when a ground to air link with qualified medical personnel can be made
Keep working ,terrific job!
Situs Judi Slot Online Yang Terbaik Dan Tergacor
Keuntungan Bermain Game Online Di Kuy4d
Situs Judi Slot Online Yang Terbaik Dan Tergacor
I do not even understand how I ended up here, but I assumed this publish used to be great
BANDIT4D Agen Slot Online Terpercaya dan Terlengkap
Hello colleagues, how is everything, and what you want
Keuntungan Bermain Game Online Di BANDIT4D
This was beautiful Admin. Thank you for your reflections.
I just like the helpful information you provide in your articles
actually awesome in support of me.
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
I like the efforts you have put in this, regards for all the great content.
I just like the helpful information you provide in your articles
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
That’s good, but I still don’t understand the purpose of this page posting, no or what and where do they get material like this.
This was beautiful Admin. Thank you for your reflections.
I don’t commonly comment but I gotta tell thankyou for the post on this perfect one : D.
I have not checked in here for some time since I thought it was getting boring, but the last few posts are great quality so I guess I’ll add you back to my daily bloglist. You deserve it my friend 🙂
SPAM4D Angka Keluaran Togel Hk, SGP , SDY
SPAM4D Pola Slot Gacor Hari Ini
SPAM4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
This was beautiful Admin. Thank you for your reflections.
You have noted very interesting points! ps decent web site. “Formal education will make you a living self-education will make you a fortune.” by Jim Rohn.
SPAM4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
SPAM4D Angka Keluaran Togel Hk, SGP , SDY
Hello colleagues, how is everything, and what you want
Some truly quality articles on this website , saved to favorites.
SPAM4D Angka Keluaran Togel Hk, SGP , SDY
You have noted very interesting points! ps decent web site. “Formal education will make you a living self-education will make you a fortune.” by Jim Rohn.
SPAM4D Situs Slot Gacor Hari Ini
SPAM4D Situs Mudah Menang Hari Ini
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
POL4D Situs Slot Gacor Hari Ini
POL4D Pola Slot Gacor Hari Ini
SPY4D Pola Slot Gacor Hari Ini
SPY4D Pola Slot Gacor Hari Ini
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
BINGO4D Pola Slot Gacor Hari Ini
KLIX4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
BINGO4D Pola Slot Gacor Hari Ini
KLIX4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
KLIX4D Pola Slot Gacor Hari Ini
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
YOI4D Situs Slot Gacor Hari Ini
YOI4D Situs Slot Gacor Hari Ini
I appreciate you sharing this blog post. Thanks Again. Cool.
YOI4D Situs Mudah Menang Hari Ini
POL4D Angka Keluaran Togel Hk, SGP , SDY
POL4D Situs Slot Gacor Hari Ini
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
POL4D Situs Slot Gacor Hari Ini
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
POL4D Angka Keluaran Togel Hk, SGP , SDY
POL4D Pola Slot Gacor Hari Ini
POL4D Situs Mudah Menang Hari Ini
POL4D Situs Mudah Menang Hari Ini
Pretty! This has been a really wonderful post. Many thanks for providing these details.
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
I do not even understand how I ended up here, but I assumed this publish used to be great
I like this web blog very much, Its a really nice spot to read and obtain information.
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
There is definately a lot to find out about this subject. I like all the points you made
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
I have not checked in here for some time since I thought it was getting boring, but the last few posts are great quality so I guess I’ll add you back to my daily bloglist. You deserve it my friend 🙂
Thanks for a marvelous posting! I genuinely enjoyed reading it, you are a great author.I will always bookmark your blog and will often come back down the road. I want to encourage continue your great job, have a nice weekend!
I carry on listening to the reports speak about getting free online grant applications so I have been looking around for the most excellent site to get one. Could you advise me please, where could i get some?
Cool that really helps, thank you.
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
I like the efforts you have put in this, regards for all the great content.
Cool that really helps, thank you.
You have noted very interesting points! ps decent web site. “Formal education will make you a living self-education will make you a fortune.” by Jim Rohn.
I need to to thank you for this fantastic read!! I definitely loved every bit of it. I have you bookmarked to check out new stuff you post?
I think the content you share is interesting, but for me there is still something missing, because the things discussed above are not important to talk about today.
I like the efforts you have put in this, regards for all the great content.
I just like the helpful information you provide in your articles
Great wordpress blog here.. It’s hard to find quality writing like yours these days. I really appreciate people like you! take care
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
Thanks for a marvelous posting! I genuinely enjoyed reading it, you are a great author.I will always bookmark your blog and will often come back down the road. I want to encourage continue your great job, have a nice weekend!
I appreciate you sharing this blog post. Thanks Again. Cool.
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
I need to to thank you for this fantastic read!! I definitely loved every bit of it. I have you bookmarked to check out new stuff you post?
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://accounts.binance.com/sv/register-person?ref=WTOZ531Y
Hi! I’m at work surfing around your blog from my new iphone 4! Just wanted to say I love reading your blog and look forward to all your posts! Keep up the fantastic work!
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
Hi! I’m at work surfing around your blog from my new iphone 4! Just wanted to say I love reading your blog and look forward to all your posts! Keep up the fantastic work!
Very well presented. Every quote was awesome and thanks for sharing the content. Keep sharing and keep motivating others.
Cool that really helps, thank you.
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
I have not checked in here for some time since I thought it was getting boring, but the last few posts are great quality so I guess I’ll add you back to my daily bloglist. You deserve it my friend 🙂
Very well presented. Every quote was awesome and thanks for sharing the content. Keep sharing and keep motivating others.
I think the content you share is interesting, but for me there is still something missing, because the things discussed above are not important to talk about today.
I have not checked in here for some time since I thought it was getting boring, but the last few posts are great quality so I guess I’ll add you back to my daily bloglist. You deserve it my friend 🙂
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article. https://accounts.binance.com/bn/register-person?ref=DB40ITMB
I do not even understand how I ended up here, but I assumed this publish used to be great
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
Nice post. I learn something totally new and challenging on websites
I like this web blog very much, Its a really nice spot to read and obtain information.
That’s good, but I still don’t understand the purpose of this page posting, no or what and where do they get material like this.
Your article helped me a lot, is there any more related content? Thanks! https://www.binance.com/de-CH/join?ref=P9L9FQKY
I do not even know how I ended up here, but I thought this post was great. I don’t know who you are but definitely you’re going to a famous blogger if you aren’t already 😉 Cheers!
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
I agree with your perspective in this article and appreciate the arguments you provide.
Incorporating data and statistics into your writing makes the information more credible and convincing.
bupropion sexualidad
Dead indited articles, Really enjoyed looking through.
Hi! I’m at work surfing around your blog from my new iphone 4! Just wanted to say I love reading your blog and look forward to all your posts! Keep up the fantastic work!
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
Nice post. I learn something totally new and challenging on websites
https://azithromycinca.com/# doxycycline canada pharmacy
doxycycline price uk
order amoxicillin 500mg: amoxil online – amoxicillin brand name
amoxicillin in india
Creator Holden, N buying cheap cialis online Brief Report Oxidative Stress Mediates Cardiomyocyte Apoptosis in a Human Model of Danon Disease and Heart Failure