ORIGINAL ARTICLE
Year: 2018 I Volume: 1 I Issue: 2 I Page: 38-41
Comparison of Trichoscan Findings of Diffuse Hair Loss in Females Versus Controls
Dr. Nancy M. Raiyani 1, Dr. Prachi Chetankumar Gajjar1, Dr. Hita H. Mehta1, Dr. Chandra Shekhar R. Jaiswal 1
1 Department of Dermatology, Venereology and Leprosy, Government Medical College, Bhavnagar, Gujarat, India
Corresponding Author:
Dr. Prachi Chetankumar Gajjar
115, Dept. of Skin & V.D., Sir T. Hospital, Bhavnagar, Gujarat, India
Email: prachi93gajjar@gmail.com
How to cite this article:
Raiyani NM, Gajjar PC, Mehta HH, Jaiswal CSR. Comparison Of Trichoscan Findings Of Diffuse Hair Loss In Females Versus Controls. JDA Indian Journal of Clinical Dermatology 2018;1:38-41.
Abstract:
Introduction: Diffuse hair loss is a worrisome complain specially in females, commonly encountered in clinical dermatological practice. Trichoscan is a non-invasive method of hair analysis that is a combination of epiluminescencetrichoscopy and automatic image analysis system. In this study we analyzed 115 number of volunteer females.
Aim -To find association between clinical presentation and objective parameters found by trichoscan.
Method: All Participants went through detailed history taking, for grouping them under acute telogen effluvium, chronic telogen effluvium, patternhair loss (PHL) and control group. On day 0, scalp hair of approx 1 cm2 area were shaved to leave an approx 0.5 mm hair stump on the area 1 cm lateral to midline on a line joining highest point of pinna. On day 3, patient would undergo hair pull test, trichoscopy and trichoscan study. Trichoscan parameters like anagen:telogen ratio, density/cm2,vellus and terminal hairs were considered for analysis.
Statistical analysis : Raw data was analysed using appropriate one way ANOVAor Kruskal-Wallis Test followed by appropriate post-hoc test.
Results : The mean hair density/cm2 is lowest in PHLgroup with having significant difference with control (p<0.01), chronic and acute TE group (p<0.05). Anagen ratio is lowest in acute TE followed by chronic TE, PHLand control group. The terminal hair ratio is significantly low in PHLgroup as compared to control, acute and chronic TE groups. (p<0.001)
Conclusion : Trichoscan is easy useful tool for female hair analysis and satisfactorily used in differentiating PHL, acute telogen effluvium and normal.
Key Words- Trichoscan, Acute telogen effluvium, Chronic telogen effluvium, Pattern hair loss (PHL)
Introduction : Diffuse hair loss is a very common complaint encountered in clinical dermatological practice which can be acute or chronic. Diffuse hair loss is a very common complaint encountered in clinical dermatological practice. It includes various conditions like acute and chronic telogen Though females are less prone to get bald naturally, it is found to be more troublesome among themit found more troublesome among them.. It is noticed that female seek medical advice more than men. Any non-patchy hair loss appreciable to patient can be considered as diffuse hair loss. Various pattern of diffuse hair loss seen in female are: telogen effluvium, anagen effluvium, patterned hair loss (androgenetic alopecia), diffuse alopecia areata and other shaft disorders.1 Trichoscan is a unit containing epiluminescence surface microscopy and digital image analysis system.2,3 It is recently developed, computer assisted non-invasive method of assessment of hair biological peculiarities that is explained as user-friendly, time-saving and reproducible method.3,4
The aim of our study was to find out if there is any association between clinical presentation and objective measurements of hair biological parameters determined by trichoscan method.
Subjects and Methods:
After approval from the Institutional Review Board,total of 115 f e m a l e s v i s i t i n g t h e o u t p a t i e n t d e p a r t m e n t o f Dermatologyduring the period of one and half years were enrolled after written informed consent.We included the females between second and fifth age groups. Females with other dermatological scalp conditions like psoriasis, lichen planus, discoid lupus erythematosus, hair shaft abnormality and cicatricial alopecia as well as those who deny for consent were excluded from the study.All participants went through detailed history including associated dermatological and other systemic diseases and thorough scalp examination. This helped us furtherfor grouping them as-anagen effluvium, acute telogen effluvium, chronic telogen effluvium, patterned hair loss, diffuse alopecia areata and control group. But, we did not comeameacross cases of anagen effluvium and diffuse alopecia areata. Routine laboratory investigations like complete blood count,renal function test, liver function test, urine analysis, HbsAg, erythrocyte sedimentation rate and serum ferritin level were carried out. Thyroid function test and other respective investigations were done if required.
Scalp examination for trichoscan was carried out as belows:
On day one, scalp hairs of approximately1 cm2 area were trimmed to leave an approximately 0.5 mm hair stump. The area was fixed as -one cm lateral to midline on a line joining highest points of both pinna in all subjects to maintain the uniformity. Gross photographs were captured. All participants were called back after 48 hours with an instruction not to wash their hair during this period. The site was re-examined by putting contact mode dermoscope (Heine Delta 20 Plus P set, Germany) with polarized light that is attached with digital camera (Canon EOS 550 D Camera, Tokyo, Japan). After proper alignment the image was captured, saved and analysed with trichoscan software. This programme analyses 1.195 cm2 area and gives different hair parameters like density/cm2,anagen hairs (%), telogen hairs (%), density vellus hairs (per cm2), density terminal hairs (per cm2), ratio vellus hairs (%) and ratio terminal hairs (%)(Figure 1 and 2).
 |
Figure 1: Example of the Trichoscan analysis of ATE |
 |
Figure 2: Example of the Trichoscan analysis of PHL |
The principle of trichoscan is based on hair biologic characteristics like anagen hairs grows daily by approximately 0.3 mm while telogen hairs do not grow. Using this trichoscan c o u n t s , h a i r s w i t h l e n g t h o f > 0 . 7 m m w µ m w e r e consideredanagen and other non-growing hairs as telogen. This value of 0.7 mµm is manually adjustable. Black permanent hair colour (Streax hair colour) was used only in few persons with white hair to improve contrast between hair and scalp skin and only on the area to be examined with dermoscope. This was made possible by applyingmixture of cream and developer solution on examination area for 15 minutes in aproportion of 1:1 ratio with the help of a wooden spatula. Hair Pull Test(HPT)from area other than clipped hair was performed in all subjects. Pulled hairs were examined under microscope to confirm whether they areanagenor telogen hairs.Hair loss pattern werethen classified as: (1) Acute Telogen Effluvium (ATE) (2) Chronic Telogen Effluvium (CTE) (3) Patterned hair loss (PHL) and (4) C o n t r o l . P a t i e n t s c o m i n g f o r o t h e r d e r m a t o l o g i c a l conditions(excluding scalp), relatives accompanying the patients, hospital staff like nurse, medical students, technicians and other healthy volunteerswith normal hair cycle were included as controls.
Statistical analysis was done using Graph pad InStat software version 3.06. Raw data was put on normality test and data with Gaussian distribution like overall hair density and terminal hair density (per cm2) (Table 1) wereanalysed using oneway ANOVA while those with non-Gaussian distribution like anagen, telogen, vellus, terminal hair (%) and vellus hairs (per cm2) (Table 2) were analysed using Kruskal-Wallis test followed by appropriate post-hoc test. P value < 0.05 were accepted to be significant.
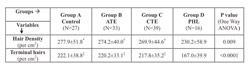 |
Table 1: Comparison of hair density and terminal hairs among different groups |
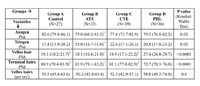 |
Table 2: Comparison of Anagen, Telogen, Vellus and Terminal hairs among different groups |
Results:
We enrolled total 115 females with age ranging between 13 to 50 years. The mean age of all with diffuse hair loss and control group were 28.9 ± 9.6 and 25.1 ± 7.6 years, respectively. The age-wise distribution was as follows: 35 (30.4%) in second decade, 39 (33.9%) in third decade, 27 (23.5%) in forth decade and 14 (12.2%) in fifth decade. We did not observe muchchanges in number of hairs per cm2, telogen hairs, vellus hairs or terminal hairs content with advancing age in any group.
In acute TE group, 45.5% cases showed positive hair pull test in whom duration of hair fall was less than or equal to two months. Specific triggering factors like febrile illness, major operative procedure and recovery from debilitating illness, etc. was identified from history only in 30.3% cases of acute TE and 17.9% cases of chronic TE.
Three out of 16 (18.8%) patients of PHL had positive family history of androgenetic alopecia. Only one participant of 16 from PHL group had clinical sign of hyperandrogenismcharacterised byexcessive hairs on upper lips and irregular menstruation.
The hair density was lowest in PHL group followed by CTE, ATE group and highest in control group with statistically significant difference between PHL and other three groups (Table 1). The control group showed highest anagen hair content(%) while acute TE group showed lowest. The percentage of telogen hairs was highest in acute TE groupfollowed by the chronic TE and PHL group while lowest in control group.Terminal hair count was significantly lower infemales with PHL compared to other groups (p <0.000). Vellus hair content was higher in PHL compared to other three groups though not significant (p < 0.3)( Table 2). We reclassified all the females with diffuse hair loss in two groups based on Hair pull test results as Hair pull test positive and Hair pull test negative. There was no significant difference in mean values of different trichoscan parameters between two groups. Average Telogen hair count was slightly higher in females with positive HPT while average anagen hair count was slightly lower in HPT positive group (Table 3).
 |
Table 3: Values of parameters between groups made as per Hair Pull Test results. |
Serum ferritin level was done in 106 subjects. Low serum ferritin level (<11 ng/ml) was found in 40.6% (43) subjects. Eight out of 24 subjects in control group had low serum ferritin.
Discussion:
Diffuse hair loss is defined as acute or chronic generalised thinning of hairs from the scalp. It includes various conditions like acute and chronic telogen effluvium, anagen effluvium, pattern hair loss and diffuse alopecia areata. Of them, telogen effluvium is the most common encountered form of diffuse hair loss which is similar to our observation.5 It is difficult to monitor the therapy of hair loss. Previously, dermatologist used to measure the hair length and diameter manually, but it is very tedious and is subjected to subjective variation. Though, invasive procedure like scalp biopsy is standard for measuring the anagen: telogen ratio and for diagnosing hair disorders but in every follow up, patient would not get convinced for biopsy to monitor the therapy. Also patient would feel discomfort for hair pulling in semi invasive methods like trichogram and unit area trichogram. Other non invasive hair evaluation methods like global hair count, hair weight test and daily hair count are prone to manual errors and often results are not reproducible. 2,3,5
So, a reliable,non invasive, safe, easy and OPD tool is required to assess the biological hair parameters.Trichoscan, introduced in 2001 is a novel, handy, non- invasive, reproducible, easy, validated and investigator independent automated image analyser which is a computer based video dermatoscopy which identifies anagen, telogen, terminal, vellus hairs and hair density.3,5 One validation trial conducted proved excellent correlation between hair parameter results analysed by trichoscan and manually.6
There is no defined or fixed normal value for hair density as there are differences according to race and genetic constitution.In the review of various studies, Rushton et al. demonstrated hair density as average 181 hairs/cm2 by phototrichogram and 237 hairs/cm2 by unit area trichogram in female subjects.7 Saraogiet al., found hair density 291.3/cm2 in normal control females and in study by Aktanet al., mean hair density of healthy volunteers was found to be 141.7 hairs/cm2 by Trichoscan while it was 212.8 hairs/cm2 by photomacrographs.8,9 Birch et al., detected hair density of 293 hairs/cm2 on using photomacrographs.10 In our study we found hair density of 277.9 ± 51.8 hairs/cm2 in control group (Table 1, Figure 3).3
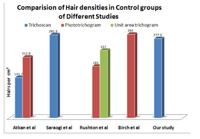 |
Figure 3: Bar chart showing comparison of hair densities in control groups of different studies |
Acute telogen effluvium is defined as diffuse hair loss occurring from all over scalp around 6 weeks to 3 months of inciting event and have self-limiting course. If precipitating event is repeated or continued, hair loss may persist for more than 6 months and considered under chronic telogen effluvium. CTE was first described by Whiting DA in 1996 as idiopathic telogen hair loss primarily affecting middle-aged women, having long fluctuating course and diffuse hair shedding that may show bitemporal thinning.11Other secondary causes of chronic telogen shedding l i k e t h y r o i d d y s f u n c t i o n , i r o n d e fi c i e n c y a n e m i a , acrodermatitisenteropathica and others were known.12,13In our study, ATE and CTE group did not show any statistically significant difference in trichoscan parameters. Hair density and terminal hair density (per cm2) were higher in ATE group than CTE while terminal and vellus hair ratio wasfound to be almost similar (Table 1, 2).
A study by Haticeet al. showed statistically significant difference in the hair density and terminal hairs between the TE group and PHL.5 In our study also, we found similar results for hair density and terminal hairs (p<0.05 and <0.001 respectively) in the acute TE and PHL group. We observed slight difference of hair density between chronic TE and PHL group (Table 1). Thus, it can be said that clinical dilemma of differentiation of chronic telogen effluvium and patterned hair loss does not completely resolve with trichoscan.
Sinclair et al., in their using punch biopsy for differentiation between chronic telogen effluvium and PHL the ratio of terminal hairs to vellus hairs (T:V) >8:1 in CTE and 4:1 in PHL.14In contrast to these findings, in our study carried out by trichoscan, we calculated the Terminal : vellus hair ratio to be 4:1 in CTE group and 3:1 in PHL group (Table 2 ). The trichoscopic features diagnostic for PHL are variation in hair shaft diameter due to miniaturization of hair follicle, peripilar sign, single hair bearing follicles, focal areas of atrichia and yellow dots while telogen effluvium is a diagnosis of exclusion (Figure 4).
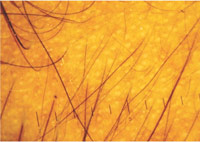 |
Figure 4: Dermoscopic features of Female androgenic alopecia: miniaturized hair follicles (arrow), variation in hair shaft diameter, single hair bearing follicles and yellow dots (arrow head). |
Decreased hair density and follicular units with single hair in the absence of characteristic signs of other causes of alopecia is considered to support clinical diagnosis of telogen effluvium.15,16 Scrutiny of trichoscopic images in our study lead to a perception that few images from telogen effluvium group showed thin hairs with equal diameter in contrast to PHL group where it showed thinned hairs with varyingdiameters.
Hair pull test which is used to determine disease activity is found to be positive during active shedding phase of both telogen effluvium and anagen effluvium.17Interesting finding noted in our study that the test is positive in cases of early androgenetic alopecia at the areas of thinningwhile negative at occipital area.There was no any statistically significant difference in telogen hairs (%) found between HPT positive PHL subjects and HPT negative PHL subjects in contradictory to that found by Haticeet al(Table 3).5 Earlier in a study by Van Nesteet al. with contrast-enhanced phototrichogram method, it was showed that increase in age and duration of complain were associated with decreased hair density in females with ludwig pattern hair loss.18 Here, our observation showed no relation of hair density with patient’s age and duration of hair loss unlike the inference made by Aktanet al.9
There are few studies which suggest relation between serum ferritin level and hair loss. Ideal ferritin level was considered to be at least40 ng/ml for healthy hair growth.19 We found very low values of serum ferritin among cases and contols,all groups, where only 6.6% (7 out of 106) showed normal range. There was no any difference in the values of serum ferritin among study groups. No association between serum ferritin level and density of hairs per cm2, anagen (%), telogen (%) and terminal hairs per cm2 were noticed in our study.
Limitations:
Limitations of the study design includes limited sample size for controls and PHL group.Our study is confined to a single centre. Few limitations experienced during procedure and the software are mentioned as below: 1) Crossing of two hair strands lead to false counting yielding higher telogen ratio 2) If the hair is cut very short, near to scalp surface it may not be detected by the software 3) Very thin hairs escape from the counting. These can be overcome by avoiding the processing of this kind of images. Similar errors are also mentioned by Saraogiet al.8
Conclusion
Trichoscan, being a non-invasive and simple office procedure, has a scope for use in routine OPD for evaluation of hair loss if the limitations of procedure are meticulously taken care off. It can be used satisfactorily in differentiating acute telogen effluvium, patterned hair loss and normal hairs. In cases of clinical conundrum, trichoscanalong with dermoscopic confirmation of condition will further allow clinician to manage patients with disease specific treatment and prognosis. Trichoscan as part of evaluation, gives psychological satisfaction to the patient which may lead to improved compliance with treatment prescribed by the physician.3,5
References:
1. Disease of Skin Appendages, In: William DJ, Timothy GB, Dirk ME, Issac MN, editors. Andrew’s Diseases of the Skin – clinical dermatology. 12th ed. Elsevier publication; 2015. p. 747-749.
2. Hoffman R. Trichoscan: combining epiluminescence microscopy with digital image analysis for the measurement of hair growth in vivo. Eur J Dermatol 2001; 11: 362-368.
3. Hoffman R. Trichoscan: A novel tool for the analysis of hair growth in vivo. J Invest DermatolSympProc 2003; 8: 109-115.
4. Singal A, Sonthalia S, Verma P. Female pattern hair loss. Indian J DermatolVenereolLeprol 2013;79: 626-640.
5. Hatice UCE ÖZKOL, Ömer ÇALKA, Necmettin AKDENIZ. Is TrichoScan a new diagnostic method for diffuse hair loss? Turk J Med Sci (2014) 44: 432-438.
6. Gassmueller J., Rowold E., Frase T., Hughes-Formella B. Validation of TrichoScan® technology as a fully-automated tool for evaluation of hair growth parameters. Eur J Dermatol 2009; 19 (3): 224-231
7. Rushton DH, Ramsey ID, James KC. Biochemical and trichological characterization of diffuse alopecia in women.Br J Dermatol 1990; 123: 187–197.
8. Saraogi PP, Dhurat RS. Automated Digital Image Analysis (TrichoScan®) for Human Hair Growth Analysis: Ease versus Errors. Int J Trichology. 2010 JanJun; 2(1): 5–13.
9. Aktan S, Akarsu S, Ilknur T, Demirtasoglu M, Ozkan S. Quanti?cation of female pattern hair loss: A study in a Turkish population. Eur J Dermatol. 2007;17:321–324.
10. Birch MP, Messenger JF, Messenger AG. Hair density, hair diameter and the prevalence of female pattern loss. Br J Dermatol2001; 144: 297-304.
11. Whiting DA. Chronic telogen ef?uvium: Increased scalp hair shedding in middleaged women. JAm AcadDermatol 1996;35: 899-906.
12. G r o v e r C , K h u r a n a A . Te l o g e n e f ? u v i u m . I n d i a n J DermatolVenereolLeprol 2013;79:591-603
13. Dawber RPR, Simpson NB, Barth JH. Diffuse alopecia: Endocrine, metabolic and chemical in?uences on the follicular cycle. In: Dawber RP, editor. Diseases of the Hair and Scalp. Oxford: Blackwell Science;1997. p. 123-150.
14. Sinclair RD, Jolley D, Mallari R, Magee J. The reliability of horizontally sectioned scalp biopsies in the diagnosis of chronic diffuse telogen hair loss in women. J Am AcadDermatol 2004; 51: 189–199.
15. Jain N, Doshi B, Khopkar U. Trichoscopy in alopecias: diagnosis simpli?ed. Int J Trichology 2013;5: 170-178.
16. Rudnicka L, Olszewska M, Rakowska A, Slowinska M. Trichoscopy update 2011. J Dermatol Case Rep. 2011;5: 82–88.
17. Dhurat R. Saraogi P. Hair Evaluation Methods: Merits and Demerits. .Int J Trichology. 2009 Jul-Dec;1(2): 108–119.
18. Van Neste D. Female patients complaining about hair loss: documentation of defective scalp hair dynamics with contrast-enhanced phototrichogram. SkinResTechnol2006;12: 83-88.
19. Harrison S, Sinclair R. Telogen ef?uvium. ClinExpDermatol 2002; 27: 389–395.

best dating online website best dating web online sex https://adult-classifieds-online-dating.com
buy custom papers where can i find someone to write my college paper custom paper service order a paper online
pay for someone to write your paper help with writing a paper
for college do my papers paper writing services legitimate
custom paper writing professional paper writing services find someone to write my paper will someone write my paper for me
pay someone to write a paper for me someone to write my paper for me academic
paper writing services professional paper writing service
need help write my paper paper writer services how to find
someone to write my paper what should i write my paper about
order papers online customized writing paper buy papers
online cheap best online paper writers
college paper writing service college paper service cheap custom papers buy a college paper
paper writing services legitimate writing paper services can i pay someone to write
my paper write-my-paper-for-me.org
buy papers online for college write my paper online buy papers online mypaperwritinghelp.com
need someone to write my paper for me where can i find someone to write my college paper
college papers for sale professional paper writing services
best college paper writing service find someone
to write my college paper where can i buy resume paper order custom papers
pay someone to write a paper buy college paper
write my paper apa style buy papers online cheap
pay to do my paper write my college paper for me buying college papers
online buy literature review paper
pay to do my paper write my paper fast buy a paper write my paper for me
college paper writers buy a college paper online paper writing service college find someone to write my paper
coursework grades coursework based masters coursework cpa coursework
coursework report coursework high school coursework umich creative writing coursework ideas
coursework based a levels coursework support java coursework qmul coursework extension
coursework on cv coursework marking coursework jamii forum mycourseworkhelp.net
jd coursework coursework common app java coursework design coursework
coursework sample of written work coursework cv coursework geography coursework high school
coursework on resume example coursework king’s college coursework resume example jerusalem coursework
coursework meaning coursework and research coursework plagiarism checker king’s college london coursework
do my coursework for me coursework masters meaning coursework college courseworkinfotest.com
ho ho ho merry christmas song download https://ringtonessphone.com/santa-ho-ho-ho-merry-christmas.html
trex running https://chromedinos.com
best ringtones https://ringtonessbase.com
funny ringtones https://downloadfreeringtoness.com/funny-ringtones
cartoon and funny sounds https://sounddeffects.com/cartoon-and-funny-sounds
https://drugsoverthecounter.com/# mupirocin ointment over the counter
Improvements on scores on the AMS have been noted after 1 month 38, 6 weeks 40 to 3 months on treatment with parenteral testosterone undecanoate 35 or after 9 months on testosterone gel 74 continuing over a total of 18 months 74 what happens when a women takes viagra
buy cialis online india FASEB J 24 1799 1812
women and viagra Systolic failure, or heart failure with a reduced ejection fraction, happens when the left side of the heart becomes weakened, and is unable to contract with enough force to eject a sufficient amount of blood out to the body
arimidex for sale online order arimidex pills anastrozole usa
free dating site chatting free for online chatting
with singles local free dating sites free date sites
online dating personal ads free bondage dating sites free dating and chatting
dating chat site free dating chatting online
free online dating & adult personals free 100% totally free dating services
free dating sites chat dating sites free near me free woman paid debit video totally free dating site
free hookup websites near me absolutely free dating sites
free dating sites without registering or fees mature adult phone chat
100% completely free dating site dating sites that are
totally free online dating sites for free feminization of men website free online dating
free chatting for marriage single senior dating site online date personal local online dating
free web date site 100% absolutely free people search singles near me https://freedatinglive.com
free adult chat rooms international dating free personal ads online dating site sign up
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
english essay writers help writing scholarship essays help on college essay professional essay writers
best essay help best college essay writing service college application essay help online i need help writing an argumentative essay
community service essay sample best custom essay writing service who can write my essay help write an essay online
write my college essay national honor society essay help professional
essay writer essays service
cheapest custom essay writing the best essay writing service essay conclusion help custom essays cheap
write my essay students best website to get essays can t write my essay need help with essay
order cheap essay help writing scholarship essays recommended essay writing service help write essay for me
best essay writing service reviews write my essay generator buy
cheap essays custom essay paper
college essay editing service essay writing services singapore compare and contrast essay help college essay help online
essay writers for hire help writing my college essay best essay writing service online essay writing services legal
I appreciate you sharing this blog post. Thanks Again. Cool.
student essay help quality custom essay essay checking service the best essay writing services
college essays writing services help me write a descriptive essay custom essays writing service compare and contrast essay help
custom essay writing online cheap essay expert essay writers custom essays services
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
I think the content you share is interesting, but for me there is still something missing, because the things discussed above are not important to talk about today.
Pretty! This has been a really wonderful post. Many thanks for providing these details.
college essay writers block my essay writer custom essay help higher english essay help
I just like the helpful information you provide in your articles
essay writing service ratings write essay service essay on helping poor people custom essays writing
essay on social service mba essay editing service admission essay help english essay writing help
%random_anchor_text% %random_anchor_text% %random_anchor_text% .
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
Hi there to all, for the reason that I am genuinely keen of reading this website’s post to be updated on a regular basis. It carries pleasant stuff.
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
I have read your article carefully and I agree with you very much. This has provided a great help for my thesis writing, and I will seriously improve it. However, I don’t know much about a certain place. Can you help me?
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
Hi there to all, for the reason that I am genuinely keen of reading this website’s post to be updated on a regular basis. It carries pleasant stuff.
I like the efforts you have put in this, regards for all the great content.
Pretty! This has been a really wonderful post. Many thanks for providing these details.
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
very informative articles or reviews at this time.
Nice post. I learn something totally new and challenging on websites
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
Nice post. I learn something totally new and challenging on websites
I do not even understand how I ended up here, but I assumed this publish used to be great
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
I just like the helpful information you provide in your articles
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
I think this post makes sense and really helps me, so far I’m still confused, after reading the posts on this website I understand.
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
Very well presented. Every quote was awesome and thanks for sharing the content. Keep sharing and keep motivating others.
This was beautiful Admin. Thank you for your reflections.
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
I just like the helpful information you provide in your articles
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
Nice post. I learn something totally new and challenging on websites
Try to slowly read the articles on this website, don’t just comment, I think the posts on this page are very helpful, because I understand the intent of the author of this article.
Nice post. I learn something totally new and challenging on websites
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
meclizine canada meclizine united kingdom meclizine over the counter
best custom essay writing services medical school essay service fake essay writer what is a good essay writing service
buy essay online cheap essay writer funny cheap custom essay best medical school essay editing service
essay writing service ratings essay writting service college admission essay editing services custom essay writing cheap
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
help starting an essay best custom essay website can i get someone to write my essay english literature essay help
frank casino win real money 18 and up online casino usa real money real money casinos best online casino to win money
college essay editing service application essay
writing service the best essay writing services essay help forum
college essay proofreading service macbeth essay
help essay writing service legal essay writer funny
cost of macrobid macrobid 100 mg otc macrobid 50 mg medication
essay writing help online cheap custom essay help writing an essay cheap custom essays
essay writing services recommendations admission essay services essay paper
writing help can someone write my essay
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
how to buy imitrex how to buy imitrex imitrex 50 mg without a prescription
Pretty! This has been a really wonderful post. Many thanks for providing these details.
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
zocor 40mg without a doctor prescription zocor pills cost of zocor 20mg
I really like reading through a post that can make men and women think. Also, thank you for allowing me to comment!
canadian rx pharmacy
cheap drugs canada
Nice post. I learn something totally new and challenging on websites
Nice post. I learn something totally new and challenging on websites
I do not even understand how I ended up here, but I assumed this publish used to be great
antibakterielles Duschgel
Nice post. I learn something totally new and challenging on websites
legitimate online pharmacy usa
Pretty! This has been a really wonderful post. Many thanks for providing these details.
I appreciate you sharing this blog post. Thanks Again. Cool.
my canadian pharmacy viagra
avast vpn free trial best free vpn windows 10 what
is the best vpn? most popular vpn service
Very well presented. Every quote was awesome and thanks for sharing the content. Keep sharing and keep motivating others.
what does vpn mean how to buy vpn best windows vpn best free vpn for mobile
his comment is here
best free vpn for macbook best vpn reddit 100 free vpn free unlimited vpn
Pretty! This has been a really wonderful post. Many thanks for providing these details.
canadian pharmacy
compare prescription drug prices
best mobile vpn buy avast secureline vpn best vpn value buy us vpn
best mobile vpn buy avast secureline vpn best vpn value https://imfreevpn.net/
best free vpn for tor best vpn for macbook best
kodi vpn vpn free mac
best free vpn for tor best vpn for macbook best
kodi vpn https://superfreevpn.net/
buy dedicated vpn free vpn for kodi how does vpn work free vpn hotspot shield
buy dedicated vpn free vpn for kodi how does vpn work https://freevpnconnection.com/
best vpn 2018 reddit which vpn to buy where to buy vpn router secureline vpn
best vpn 2018 reddit which vpn to buy where to buy vpn router https://free-vpn-proxy.com/
the best vpn for windows 10 best secure vpn best free vpn download buy netgear vpn client
the best vpn for windows 10 best secure vpn best free vpn download https://shiva-vpn.com/
cost of buspar buspar 10mg medication where can i buy buspar
canadain pharmacy no prescription
best vpn multiple devices nord vpn free trial what’s the best vpn free vpn for ubuntu
best vpn multiple devices nord vpn free trial what’s
the best vpn https://ippowervpn.net/
cialis 80 https://hippharmo.com/
canadian pharmacy testosterone gel
best vpn for mlb tv best free vpn for mobile best completely
free vpn one time buy vpn
best vpn for mlb tv best free vpn for mobile best completely free
vpn https://freehostingvpn.com/
canadian pharcharmy reviews
opera vpn best vpn for xbox one vpn with free trial best vpn 2022 reddit
opera vpn best vpn for xbox one vpn with free trial https://rsvpnorthvalley.com/
Extra resources
list of online canadian pharmacies
cheap online pharmacy
online prescriptions
medication without prior prescription
pyridium prices pyridium 200mg medication pyridium prices
Link Slot Terpercaya POL4D bandar judi slot online
You work hard. You provide for your family. You have done everything right. The only problem is … you don’t have papers. Síguenos en: Not having papers is scary. Your fate is always in someone else’s hands. Nosotros damos resultados a nuestros clientes. Sometemos cientos de casos al año y estamos dedicados a lograr buenos resultados a nuestros clientes. Nosotros damos resultados a nuestros clientes. Sometemos cientos de casos al año y estamos dedicados a lograr buenos resultados a nuestros clientes. CAREERS Trabajas duro. Tú mantienes a tu familia. Has hecho todo bien. El único problema es … no tienes papeles. Síguenos en: You haven’t seen your mother in years. You haven’t been able to visit your home. You’re afraid that one day you’ll be taken away from your children.
http://dmonster130.dmonster.kr/bbs/board.php?bo_table=free&wr_id=614920
Geer has written and spoken on the importance of diversity in prosecutor’s offices and was a member of the pilot class of “Prosecution Leaders of Now,” a leadership-focused collaboration of organizations that champion the transformation of prosecution. When it becomes obvious that her age requires help, Margaret Mary hires Robin Bird (the delightful Willow Geer), a not quite so young a woman, whose husband left her for another man. She professes that she has a goal to become an actress, but that isn’t in the cards. The age gap between the women is both comical and emotional. In February 2018, Geer sued Phoebe Putney under Georgia’s Open Records Act (Georgia Code § 50-18-70 et seq., i.e. and the sections that follow) to compel Phoebe Putney to release all the minutes from Phoebe Putney’s board meetings from January 2008 through December 2017. Phoebe Putney answered the complaint, asserting multiple defenses. It also included a counterclaim for attorneys’ fees under Georgia Code § 50-18-73 (b), which allows for an award of attorneys’ fees in any lawsuit brought under the Georgia Open Records Act “in which the court determines that either party acted without substantial justification either in not complying with this chapter or in instituting the litigation.”
geodon 40 mg pills geodon united states where to buy geodon
I carry on listening to the reports speak about getting free online grant applications so I have been looking around for the most excellent site to get one. Could you advise me please, where could i get some?
visit this site
aricept 5mg purchase aricept united states aricept without a prescription
Thanks for a marvelous posting! I genuinely enjoyed reading it, you are a great author.I will always bookmark your blog and will often come back down the road. I want to encourage continue your great job, have a nice weekend!
how to purchase zyprexa zyprexa australia zyprexa cost
their explanation
cozaar 50mg without a prescription buy cozaar cozaar united kingdom
find more information
brillx скачать
https://brillx-kazino.com
Бриллкс Казино — это не просто игра, это стиль жизни. Мы стремимся сделать каждый момент, проведенный на нашем сайте, незабываемым. Откройте для себя новое понятие развлечения и выигрышей с нами. Brillx — это не просто казино, это опыт, который оставит след в вашем сердце и кошельке. Погрузитесь в атмосферу бриллиантового азарта с нами прямо сейчас!Добро пожаловать в мир азарта и возможностей на официальном сайте Brillx Казино! Здесь, в 2023 году, ваш шанс на удачу преумножается с каждым вращением барабанов игровых аппаратов. Brillx — это не просто казино, это уникальное путешествие в мир азартных развлечений.
Kuy4d: Situs Game Slot Online Gacor Terpercaya Dan Aman 2023
Situs Game Slot Gacor Online Uang Asli Terpercaya Kuy4d
dark market onion darknet drug market
Situs Judi Slot Online Yang Terbaik Dan Tergacor
Vavada casino играть
https://animalprotect.org/forum/index.php?action=profile;u=5062
Играя на деньги в слоты-онлайн на официальном сайте Казино Вавада, вы перенесетесь в мир невероятных приключений и неожиданных поворотов. Наши разнообразные игры не оставят равнодушными даже самых искушенных азартных геймеров. Ведь Вавада Казино славится своими уникальными азартными сокровищами, спрятанными среди множества игровых автоматов.Добро пожаловать в захватывающий мир азарта и увлекательных игр на официальном сайте — Казино Вавада! Если вы готовы окунуться в водоворот невероятных эмоций и испытать удачу в самых захватывающих онлайн-слотах, то Вавада Казино — ваш идеальный выбор.
brillx casino официальный мобильная версия
Brillx
В 2023 году Brillx Казино стало настоящим оазисом для азартных путешественников. Подарите себе незабываемые моменты радости и азарта. Не упустите свой шанс сорвать куш и стать частью легендарной истории на страницах брилкс казино.Бриллкс казино в 2023 году предоставляет невероятные возможности для всех азартных любителей. Вы можете играть онлайн бесплатно или испытать удачу на деньги — выбор за вами. От популярных слотов до классических карточных игр, здесь есть все, чтобы удовлетворить даже самого искушенного игрока.
cheap compazine cheapest compazine compazine 5mg pharmacy
gama casino онлайн
https://www.chef.com.ua/index.php/forum/encyclopedia/kakie-est-bonusy-v-vavada-kazino-dlya-novichkov-4101.html
Что делает Gama Casino настолько особенным? Это не только вход на официальный сайт, это путешествие в мир, где каждая буква вашего имени может стать символом удачи. Именно здесь, на страницах Гама Казино, судьба и азарт переплетаются, создавая неповторимую атмосферу, которая затягивает с первого взгляда.Забудьте о рутине и суете, потому что на Gama Casino вы откроете для себя весь мир онлайн развлечений. Именно здесь, на сайте Гама Казино, вас ждут веселье и богатство, а рулетка станет мастером вашей судьбы.
very informative articles or reviews at this time.
Situs Game Slot Gacor Online Uang Asli Terpercaya BANDIT4D
промокоды казино cat 2023
https://www.feroza.ru/forum/viewtopic.php?f=17&t=20539
Не хотите скачивать? Не проблема! Cat Casino также предоставляет вам возможность играть онлайн, не загружая ничего на свое устройство. Просто пройдите регистрацию на официальном сайте, и вы окажетесь в виртуальной реальности Кэт Казино, где каждый клик мыши может повернуть ваш мир вверх дном.Ощути магию азарта в каждом вращении барабана. Cat Casino – это не просто место, где можно играть в игровые автоматы, это мир, который оживает с каждым кликом. Каждый слот – это история, которая разворачивается перед твоими глазами, и ты можешь стать ее главным героем.
Вавада казино сайт
http://forum.actionpay.ru/profile.php?id=800624
Так что не упустите шанс войти в мир Вавада Казино и испытать удачу в играх, где каждый спин барабанов может изменить вашу судьбу. У нас есть все, что нужно для незабываемого азартного опыта. Не упустите свой шанс — вперед, к победам на Казино Вавада!Для ценителей азарта и риска, для тех, кто ищет настоящие адреналиновые ощущения, Казино Вавада подготовило специальные разделы с карточными играми и рулеткой. Здесь каждая ставка – это шанс поймать удачу за хвост и уйти с внушительным выигрышем.
Вавада казино
Вавада казино
Итак, если вы ищете место, где сходятся азарт, инновации и богатый выбор игр, то Казино Вавада – ваш идеальный выбор. Войдите и окунитесь в мир увлекательных приключений на страницах Vavada Casino. Здесь каждый спин колеса – это новый поворот в вашей истории азартных побед!Не забудьте попробовать демо-режим, чтобы ощутить атмосферу игры, не теряя при этом своих средств. Это отличная возможность познакомиться с многообразием слотов, предоставляемых Казино Вавада. И помните, что настоящие победы ждут вас в режиме игры на деньги.
BANDIT4D Agen Slot Online Terpercaya dan Terlengkap
gama casino официальный сайт
https://gti-club.ru/forum/showthread.php?t=58591
Приветствуем вас в удивительной вселенной азартных развлечений — Gama Casino, где каждый клик приоткроет завесу тайны захватывающего мира азарта и возможностей. Ищете путь к входу на официальный сайт, чтобы окунуться в водоворот азартных эмоций? Гама Казино — ваш проводник в этот потрясающий мир!Играть в Gama Casino – это не просто казино, это путешествие в мир азартных мечтаний, где каждый спин – это новый поворот в сюжете вашей игры. Игровые автоматы России откроют перед вами безграничные возможности для выигрыша и развлечения. Каждая машина в Gama Casino – это как глава в увлекательной книге, наполненной сюрпризами и азартом.
cat casino промокод для новых игроков
http://vzarabotke.mybb.ru/viewtopic.php?id=7301
С Cat Casino вы получаете билет в мир, где каждый может стать Кэтом на крыше мира. Мы готовы подарить вам больше, чем просто игры. Это место, где каждый может стать частью истории азартных приключений, где ваше имя запомнится как главный герой барабанов.Коротко говоря, Cat Casino — это не просто игровая площадка, это мир возможностей и развлечений. Скачайте приложение Cat Casino и окунитесь в атмосферу удовольствия, где каждый спин может изменить вашу судьбу. Не упустите свой шанс на великолепный куш и захватывающие приключения на Cat Casino, где сливаются азарт и элегантность.
recommended you read
Вавада казино онлайн
https://forum.magazyngitarzysta.pl/viewtopic.php?f=17&t=80930&p=508055#p508055
Вход на официальный сайт Казино Вавада — это проход в виртуальное королевство увлекательных игр и головокружительных ставок. Независимо от того, являетесь ли вы опытным игроком или новичком, Вавада Казино предлагает вам широкий спектр слотов-онлайн, где каждый спин барабанов — это шанс поймать удачу за хвост.Казино Вавада – это не просто игровая площадка, это целый игровой космос, где каждый спин барабанов слотов-онлайн превращает ваше время в захватывающее событие. Полная версия игровых автоматов бесплатно раскроет перед Вами мир невероятных возможностей. Неважно, новичок Вы или опытный игрок, Вавада Казино дарит радость от игры и шанс победить!
казино gama вход
https://www.na-krychke.ru/com/forum/topic/5/
Широкий ассортимент игровых автоматов, представленных на Gama Casino, словно открывает карту сокровищ: каждая игра – это ключ к новому миру призов и увлекательных историй. Гама Казино готово предоставить вам билет в мир азартных приключений, где вы сможете доказать свое мастерство и сорвать внушительный куш.Гама Казино — это не просто бескрайний океан азартных игр. Это ваша судьба, которая в ваших руках. Насладитесь непередаваемыми ощущениями, играя в игровые автоматы, представленные на страницах Gama Casino. Мир азарта в России открывает свои объятия перед вами.
sildenafil australia where to buy sildenafil sildenafil without a doctor prescription
There is definately a lot to find out about this subject. I like all the points you made
код на казино cat casino
http://www.odnopolchane.net/forum/group.php?do=discuss&gmid=222
Cat Casino — не просто портал развлечений, это целый космос эмоций и волнения, где Кэт Казино приветствует всех, кто готов отправиться в путешествие по бескрайней вселенной игровых автоматов. Здесь вы найдете горячие новинки, свежие как утренний ветерок, и классические игры, словно дорогие старые друзья, всегда готовые подарить вам незабываемые моменты азарта.Cat Casino – твой верный спутник в мире азарта! В 2023 году этот официальный сайт рад представить тебе уникальную возможность скачать игровые автоматы и окунуться в водоворот бесконечных развлечений прямо на своем устройстве. Ощути дыхание азарта в каждом вращении барабанов, будь в центре событий с Cat Casino, Кэт Казино.
Вавада казино сайт
https://torontovka.com/forum/topic/130910
Так что не упустите шанс войти в мир Вавада Казино и испытать удачу в играх, где каждый спин барабанов может изменить вашу судьбу. У нас есть все, что нужно для незабываемого азартного опыта. Не упустите свой шанс — вперед, к победам на Казино Вавада!Добро пожаловать в удивительный мир азарта и больших выигрышей — Казино Вавада! Этот завораживающий игровой портал являет собой настоящий рай для любителей азартных развлечений. Если вы ищете официальный сайт, который предоставит вам возможность войти и окунуться в захватывающий мир азартных игр, то Вавада Казино станет вашим надежным спутником.
онлайн казино на биткоин
https://t.me/s/bitkoin_kazino1
И так, биткоин казино на пороге новой эры – эры, где виртуальное сливается с реальным, а азарт переплетается с инновациями. Онлайн казино на биткоин 2023 – это место, где каждый может стать частью увлекательной революции, где биты и байты ткаут волшебную паутину развлечения и возможностей. Казино биткоин становится платформой не только для азартных игр, но и для вдохновения, развития и смелых решений.Таким образом, биткоин казино продолжает приковывать внимание игроков, удерживая их в захватывающем водовороте инноваций и азарта. Онлайн казино на биткоин 2023 приглашает каждого на уникальное путешествие, где биты и монеты создают мелодию удачи, а возможности кроются за каждым углом экрана. Казино биткоин – ваш билет в мир волнения и бесконечных перспектив.
сайт gama casino
http://portal.krasno.ru/viewtopic.php?f=28&t=55671
И так, дорогие друзья, добро пожаловать в Gama Casino — место, где рождаются легенды, а каждый Гама Казино игрок может стать главным героем своей собственной истории. Приготовьтесь к вращению барабанов, потому что здесь рождаются победители!Игровые автоматы в России набирают обороты благодаря Gama Casino. Наша платформа собрала в себе богатство игровых вариаций — от традиционных классических слотов до передовых трехмерных автоматов. Это как путешествие во времени, где вы можете выбрать, с какой эпохой вы хотите встретиться, и Гама Казино сделает всё остальное.
pokerdom официальный сайт скачать мобильную версию бесплатно
https://t.me/s/pokerdom_zerkalo_na_segodnya
Какая особенность делает PokerDom Casino настоящим алмазом среди онлайн-казино? Это не просто платформа для игры, это источник невероятных эмоций и возможностей. Играть онлайн в слоты на реальные деньги на PokerDom Casino — это как погружение в океан высоких ставок и невероятных призов.Скачайте клиент на ПК, чтобы войти в мир PokerDom Casino через портал азарта, распахнутый специально для вас. Это ваш билет в мир невероятных ставок, волнений и выигрышей. Насладитесь игрой с удовольствием и без задержек — наш клиент на ПК сделает все, чтобы ваше путешествие было максимально плавным.
код на казино cat casino
http://ruspioner.ru/profile/view/49144
Играя в Cat Casino, Кэт Казино, вы попадаете в атмосферу совершенно новых ощущений. Это не просто казино, это путешествие в мир невероятных приключений. Игровые автоматы на Cat Casino созданы с огромной любовью к деталям, где каждая вращающаяся барабана — как страница в увлекательной книге. Вас ждут великолепные графика, уникальные звуковые эффекты и увлекательные сюжеты.Играть онлайн на Cat Casino так же легко, как и дышать. Просто зайдите на официальный сайт, и вас ожидает мир азарта, готовый поразить вас своим разнообразием. С Cat Casino, каждая минута превращается в мгновение удовольствия, каждая ставка — в шанс на удачу. Не упустите возможность погрузиться в мир кардинально новых впечатлений вместе с Cat Casino.
Скачать Покердом на деньги на андроид
Скачать Покердом на деньги на андроид
Искусство покера воплощается в захватывающем приложении, доступном для скачивания на андроид – Скачать Покердом на деньги на андроид. Это не просто приложение, это вратарь в мир азартных побед и стратегических решений. Если вы ищете способ сделать свои дни более интересными и поддать им виток азарта, Скачать Покердом на деньги на андроид – ваш выбор.Когда наступает время выбирать приложение для игры, Скачать Покердом на деньги на андроид стоит на первом месте в списке азартных эмоций. Это не просто приложение, это путеводитель в мире ставок и риска, где ваша доля азарта может стать источником дополнительного дохода. Покердом на андроид – это как билет на азартный поезд, который направляется в сторону возможностей и волнения.
gama casino
http://www.dhe-nlp.ru/forum/index.php?showuser=13842
Так что, готовы взять свою ставку удачи? Gama Casino с удовольствием подарит вам билет в мир азартных впечатлений. Входите на официальный сайт Гама Казино и дайте себе шанс испытать дыхание настоящего азарта, словно вы впервые прокладываете путь в неизведанные земли возможностей.И не забывайте, что Гама Казино – это не только игровые автоматы, это целый мир развлечений. Блэкджек, покер, рулетка – каждая игра как аккорд в мелодии вашего азартного вечера. Gama Casino – это как волшебная палитра, на которой каждый ход становится штрихом в вашем произведении.
Покердом бездепозитный бонус 1000
казино биткоин
Не упустите шанс почувствовать вкус азартной победы с Покердом бездепозитный бонус 1000! Ведь каждый бонусный доллар – это как капля адреналина, добавленная в вашу игру. Сделайте свои ставки, проявите свою азартную интуицию и начните свой азартный путь с бонуса без депозита. Казино Покердом дарит вам этот шанс – вам остается только взять его в свои руки и начать свой путь к азартному успеху!Виртуозно разыгрывайте свои карты и стратегические ходы, используя свой Покердом бездепозитный бонус 1000. В этой азартной игре каждый ход – как важное решение в шахматной партии, и только вы вольны выбирать, какой путь в азартной стратегии следует выбрать. Даже самый маленький бонус может стать ключом к большим победам, а Покердом бездепозитный бонус 1000 – это ваш персональный билет к успеху!
Vavada casino онлайн
https://www.segwayninebot.ru/club/user/13703/blog/index.php
Добро пожаловать в удивительный мир азарта и больших выигрышей — Казино Вавада! Этот завораживающий игровой портал являет собой настоящий рай для любителей азартных развлечений. Если вы ищете официальный сайт, который предоставит вам возможность войти и окунуться в захватывающий мир азартных игр, то Вавада Казино станет вашим надежным спутником.Добро пожаловать в мир азарта и увлекательных приключений – на официальный портал Казино Вавада! Здесь, на грани воображения и реальности, Вас ждут невероятные возможности для крупных выигрышей. Войдите в захватывающий виртуальный мир развлечений и азартных игр на деньги, доступный на нашем сайте в любое удобное для Вас время.
бонус код для казино cat
http://www.rus-canary.ru/cgi-bin/ruscan/p_forum.pl?s=7071u7tvwetBPKS1glPe&act=view&id=5356&p=1#start
Ощути магию азарта в каждом вращении барабана. Cat Casino – это не просто место, где можно играть в игровые автоматы, это мир, который оживает с каждым кликом. Каждый слот – это история, которая разворачивается перед твоими глазами, и ты можешь стать ее главным героем.С Cat Casino, Кэт Казино, каждый день – это новая возможность испытать удачу на вкус. Играй онлайн, скачивай слоты – выбор за тобой! Мы создали удобный интерфейс, чтобы ты мог сосредоточиться только на игре. Никаких лишних забот, только азарт и волнение.
Vavada casino играть
https://www.feroza.ru/forum/viewtopic.php?f=17&t=19354&p=85132&sid=2c7d0a086dd86cbdced1b719211bb705
Когда вы откроете для себя Казино Вавада, вы поймете, что это не просто игровой портал. Это мир, где реальность переплетается с виртуальностью, а Вавада становится проводником в мир увлекательных приключений и невероятных сюрпризов. Не пропустите шанс окунуться в водоворот страстей и азарта на страницах Vavada Casino. Войдите и дайте себе возможность стать частью этой захватывающей истории.Здесь вы найдете не только знакомые слоты, но и те, которые поразят своей уникальностью. Наша команда тщательно отбирает самые захватывающие и новаторские игры, чтобы каждый спин был настоящим открытием. Вавада Казино предлагает богатство вариантов – от классических фруктовых машин до современных слотов с захватывающими сюжетами.
how to purchase etodolac cheap etodolac 400mg etodolac 200mg coupon
биткоин казино
онлайн казино на биткоин
Виртуальное азартное веселье переплетается с инновационными технологиями, поднимая биткоин казино на новую ступень привлекательности в 2023 году. Онлайн казино на биткоин 2023 стало настоящим меккой для ценителей азарта, стремящихся к уникальному опыту и инновационным впечатлениям. Казино биткоин – это не просто платформа для развлечений, это оазис, где каждый эффективно сочетает азарт и потенциальную прибыль, завернутые в захватывающий блокчейн пакет.Играя в казино биткоин, вы ощутите адреналин и увлечение, которые оживают благодаря передовым технологиям и передаче стратегического контроля в ваши руки. Онлайн казино на биткоин 2023 предлагает увлекательные сражения с уникальными шансами на выигрыш, где каждый спин и ставка – это как решительный ход в шахматной партии. В этой эпохе биткоин казино, вы не просто наблюдатель, вы – стратег, ловко разыгрывающий свои карты на игровом столе инноваций.
покердом зеркало
покердом зеркало
Какая особенность делает PokerDom Casino настоящим алмазом среди онлайн-казино? Это не просто платформа для игры, это источник невероятных эмоций и возможностей. Играть онлайн в слоты на реальные деньги на PokerDom Casino — это как погружение в океан высоких ставок и невероятных призов.Лицензионные автоматы PokerDom Casino — это как сокровищница пирата, полная неожиданных сокровищ. Ваш путь к богатству начинается с выбора автомата. Наши лицензионные автоматы Покердом Казино разнообразны как множество жизненных сюжетов. От арктических приключений до дерзких космических путешествий, у нас есть все!
gama casino
https://enkor.ru/forum/viewtopic.php?p=16949
Когда рулетка крутится и мягкий свет Gama Casino окутывает вас, словно волшебным покрывалом, вы почувствуете, как будто стали участником киноистории. От первого касания кнопки до финального результата, Гама Казино ведет вас через этот удивительный путь.Итак, если вы ищете вход на официальный сайт, где можно играть бесплатно и на русском языке, ваш выбор однозначно — Gama Casino. Гама Казино сделает ваше путешествие в мир азарта по-настоящему незабываемым. Присоединяйтесь к нам сейчас и откройте дверь в азартное будущее прямо сейчас!
Скачать казино Покердом
Скачать Покердом на деньги на андроид
Когда речь заходит о возможности попытать удачу и пополнить баланс, Скачать Покердом на деньги на андроид – это как стартовый выстрел в мире азартных приключений. Среди разнообразия азартных приложений, Покердом на андроид выделяется, как бриллиант среди обыденных камней. Это не только возможность испытать свои навыки в покере, но и попробовать свою удачу на деньги, ведь в этом приложении каждая партия – это как великолепная стратегическая схватка.Скачать Покердом на деньги на андроид – это как взять руль в свои руки в виртуальной азартной гонке. В этом приложении вы становитесь гонщиком, готовым мчаться к победе через преграды и соперников. Покердом на андроид – это как спортивное состязание, где каждая партия – это ваше личное соревнование за место на пьедестале азартных побед.
Покердом бездепозитный бонус 1000
Покердом бездепозитный бонус 1000
Покердом бездепозитный бонус 1000 – это как ветер перемен, который может повернуть вас в направлении больших выигрышей. Этот бонус – как невидимая рука удачи, которая поможет вам обойти всех соперников на пути к азартному успеху. Покердом бездепозитный бонус – это как феерический фейерверк азартных возможностей, освещающий ваш путь к победе.Покердом бездепозитный бонус 1000 – это как магический ключ к виртуальному казино, который отпирает дверь к миру высоких ставок и больших побед. Начните свой путь в азартной игре с бонуса без депозита и откройте для себя все прелести игры в покер с невероятными возможностями для выигрыша. Да, это не сон, это реальность – Покердом бездепозитный бонус 1000 ждет вас!
бонус код для казино cat
https://nailpub.ru/forum/members/kikebi.96862/
Добро пожаловать на официальный портал азартных развлечений 2023 года — Cat Casino, лучшее место для тех, кто ищет возможность скачать игровые автоматы и насладиться их разнообразием, играя онлайн! Откройте двери в захватывающий мир азарта вместе с Кэт Казино, где каждый спин барабанов — это шанс поймать удачу за хвост и потрясти её до последней фишки.Cat Casino, Кэт Казино – твой верный напарник в стремительном мире азартных игр. Наш официальный сайт создан для истинных ценителей азарта, для тех, кто готов окунуться в виртуальное приключение и ощутить настоящее волнение. Будь в центре событий с Cat Casino – твои желания – наш приоритет.
gama casino вход
https://metrologu.ru/profile/133411-pkazofb/
Забудьте о рутине и суете, потому что на Gama Casino вы откроете для себя весь мир онлайн развлечений. Именно здесь, на сайте Гама Казино, вас ждут веселье и богатство, а рулетка станет мастером вашей судьбы.Играть в Gama Casino – это не просто казино, это путешествие в мир азартных мечтаний, где каждый спин – это новый поворот в сюжете вашей игры. Игровые автоматы России откроют перед вами безграничные возможности для выигрыша и развлечения. Каждая машина в Gama Casino – это как глава в увлекательной книге, наполненной сюрпризами и азартом.
Vavada casino официальный сайт
http://igenplan.ru/forum/user/80418/
Добро пожаловать в мир азарта и увлекательных приключений – на официальный портал Казино Вавада! Здесь, на грани воображения и реальности, Вас ждут невероятные возможности для крупных выигрышей. Войдите в захватывающий виртуальный мир развлечений и азартных игр на деньги, доступный на нашем сайте в любое удобное для Вас время.Добро пожаловать в удивительный мир азарта и больших выигрышей — Казино Вавада! Этот завораживающий игровой портал являет собой настоящий рай для любителей азартных развлечений. Если вы ищете официальный сайт, который предоставит вам возможность войти и окунуться в захватывающий мир азартных игр, то Вавада Казино станет вашим надежным спутником.
код на казино cat casino
https://club.foto.ru/user/689826
Как скачать Cat Casino и начать веселье на полную катушку? Процесс установки наше приложение на ваше устройство быстр и прост. За считанные минуты вы будете готовы к захватывающим приключениям наших игровых автоматов. А для тех, кто не хочет загружать ничего дополнительно, предлагаем вариант игры онлайн, прямо на нашем сайте. С Cat Casino играть в любимые азартные игры становится ещё удобнее.Играя в Cat Casino, вы не только получаете незабываемые впечатления, но и имеете шанс стать частью большой и дружной команды. Мы ценим каждого нашего гостя, и поэтому предлагаем щедрые бонусы и акции. Наш VIP-клуб открывает двери в мир особых привилегий и бесконечных возможностей. С Cat Casino вы не просто игрок, вы становитесь частью семьи, где каждый найдет свою нишу в мире азарта и веселья.
казино биткоин
онлайн казино на биткоин
Виртуальное азартное веселье переплетается с инновационными технологиями, поднимая биткоин казино на новую ступень привлекательности в 2023 году. Онлайн казино на биткоин 2023 стало настоящим меккой для ценителей азарта, стремящихся к уникальному опыту и инновационным впечатлениям. Казино биткоин – это не просто платформа для развлечений, это оазис, где каждый эффективно сочетает азарт и потенциальную прибыль, завернутые в захватывающий блокчейн пакет.Казино биткоин – это не только о играх и азарте, это и возможность погрузиться в увлекательную и динамичную вселенную, где биткоины становятся ключом к захватывающим приключениям. Онлайн казино на биткоин 2023 вдохновляет на исследования и освоение новых горизонтов, словно космический корабль, взлетающий к звездам. В этом мире биткоин казино, каждый игрок – это путешественник, открывающий неизведанные планеты удачи.
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
pokerdom apk скачать бесплатно
https://t.me/s/pokerdom_zerkalo_na_segodnya
Какая особенность делает PokerDom Casino настоящим алмазом среди онлайн-казино? Это не просто платформа для игры, это источник невероятных эмоций и возможностей. Играть онлайн в слоты на реальные деньги на PokerDom Casino — это как погружение в океан высоких ставок и невероятных призов.Погрузитесь в мир азартных возможностей с официальным сайтом PokerDom Casino! Это место, где грезы о крупных выигрышах становятся реальностью, где каждый спин барабанов лицензионных автоматов Покердом Казино приносит вам дыхание азарта и шанс изменить свою жизнь.
Nice post. I learn something totally new and challenging on websites
Добро пожаловать на захватывающий виртуальный парадокс развлечений — официальный сайт brillx казино! Если вы ищете истинный оазис азарта и удовольствия, то этот игровой магнат стоит на вершине вашего списка. Brillx casino — это не просто казино, это своего рода архитектурное чудо в мире виртуальных игорных заведений.
Ощутите дыхание азарта на своей коже, когда вы прокручиваете барабаны игровых автоматов Brillx casino. Это как будто встреча с великаном удачи, который приглашает вас на праздник выигрышей и возможностей. С каждой ставкой, словно аккордами великого симфонического произведения, вы будете раскрывать новые грани вашего везения.
Brillx casino — это путь к успеху и волнению, усыпанный яркими бриллиантами азартных игр. Неважно, являетесь ли вы сторонником классических слотов или предпочитаете бросить вызов современным видео-играм, этот портал соткан из разнообразия и эксклюзивности. Виртуозная графика и захватывающие сюжеты приведут вас в виртуальные миры, где реальность и фантазия переплетаются в невероятной гармонии.
Играя в Brillx casino, вы становитесь участником легендарного турнира удачи. Ваши ставки — это виртуозные ноты в симфонии азарта, а барабаны игровых автоматов — это как будто панели настоящего игрового пианино. С каждым вращением вы создаете мелодию своей победы, оставляя след в истории этой увлекательной саги.
Brillx casino — это бескрайний космос игровых возможностей. Здесь вы можете стать магом карт или виртуальным ковбоем рулетки. Это место, где даже самые неожиданные комбинации приносят грандиозные выигрыши. Откройте для себя игровой университет, где каждая ставка — это урок в искусстве азарта, а каждый джекпот — это диплом мастера удачи.
Зайдите на официальный сайт Brillx casino и откройте двери в мир, где действительность становится мифом, а миф — реальностью. Brillx casino — это путеводная звезда во мраке игровых просторов. Вам уже готовится красный ковер из карточных мастей и блеска монет. Отправьтесь в незабываемое приключение азарта и наслаждения прямо сейчас!
Скачать казино Покердом
https://t.me/s/skachat_pokerdom_na_android
Когда наступает время выбирать приложение для игры, Скачать Покердом на деньги на андроид стоит на первом месте в списке азартных эмоций. Это не просто приложение, это путеводитель в мире ставок и риска, где ваша доля азарта может стать источником дополнительного дохода. Покердом на андроид – это как билет на азартный поезд, который направляется в сторону возможностей и волнения.Иногда жизнь сама по себе игра, и в этой игре каждый ход важен. Приложение Покердом на андроид предоставляет вам карту в этот мир, где ваши решения могут стать ключом к увлекательным приключениям и даже финансовым успехам. Скачать Покердом на деньги на андроид – это как приглашение на захватывающую вечеринку, где каждая ставка – это новая история, а каждый выигрыш – незабываемое воспоминание.
gama casino онлайн
http://mastrerkon.ru/forum/viewtopic.php?f=27&t=10276
Для тех, кто ценит возможность играть бесплатно, на русском языке и в уютной обстановке своего дома, Gama Casino — это подарок судьбы. Мы предлагаем вам насладиться всей палитрой азартных развлечений абсолютно бесплатно, не взирая на временные ограничения или же свои материальные возможности. Гама Казино делает акцент на равенстве игрового опыта для всех!Gama Casino — это не просто платформа для азартных игр, это легендарное место, где ваша мечта об игре на рубли сливается с реальностью. Чувствуйте ритм азарта в каждом клике, ведь Гама Казино откроет перед вами дверь в увлекательный мир азартных развлечений.
Покердом бездепозитный бонус 1000
онлайн казино на биткоин
Покердом бездепозитный бонус 1000 – это как магический ключ к виртуальному казино, который отпирает дверь к миру высоких ставок и больших побед. Начните свой путь в азартной игре с бонуса без депозита и откройте для себя все прелести игры в покер с невероятными возможностями для выигрыша. Да, это не сон, это реальность – Покердом бездепозитный бонус 1000 ждет вас!Любите игру в покер? Хотите ощутить вкус победы и азарта, не вкладывая свои сбережения? Тогда Покердом бездепозитный бонус 1000 – это ваш билет в мир азартных возможностей! Смело присоединяйтесь к игре в уникальном онлайн казино Покердом с промокодом на бездепозитный бонус и получите свои первые 1000 единиц азартного капитала!
Покердом бездепозитный бонус 1000
https://t.me/s/pokerdom_bezdepozitny_bonus_1000
Любите игру в покер? Хотите ощутить вкус победы и азарта, не вкладывая свои сбережения? Тогда Покердом бездепозитный бонус 1000 – это ваш билет в мир азартных возможностей! Смело присоединяйтесь к игре в уникальном онлайн казино Покердом с промокодом на бездепозитный бонус и получите свои первые 1000 единиц азартного капитала!Покердом бездепозитный бонус 1000 – это как ветер перемен, который может повернуть вас в направлении больших выигрышей. Этот бонус – как невидимая рука удачи, которая поможет вам обойти всех соперников на пути к азартному успеху. Покердом бездепозитный бонус – это как феерический фейерверк азартных возможностей, освещающий ваш путь к победе.
Добро пожаловать на захватывающий виртуальный парадокс развлечений — официальный сайт brillx казино! Если вы ищете истинный оазис азарта и удовольствия, то этот игровой магнат стоит на вершине вашего списка. Brillx casino — это не просто казино, это своего рода архитектурное чудо в мире виртуальных игорных заведений.
Ощутите дыхание азарта на своей коже, когда вы прокручиваете барабаны игровых автоматов Brillx casino. Это как будто встреча с великаном удачи, который приглашает вас на праздник выигрышей и возможностей. С каждой ставкой, словно аккордами великого симфонического произведения, вы будете раскрывать новые грани вашего везения.
Brillx casino — это путь к успеху и волнению, усыпанный яркими бриллиантами азартных игр. Неважно, являетесь ли вы сторонником классических слотов или предпочитаете бросить вызов современным видео-играм, этот портал соткан из разнообразия и эксклюзивности. Виртуозная графика и захватывающие сюжеты приведут вас в виртуальные миры, где реальность и фантазия переплетаются в невероятной гармонии.
Играя в Brillx casino, вы становитесь участником легендарного турнира удачи. Ваши ставки — это виртуозные ноты в симфонии азарта, а барабаны игровых автоматов — это как будто панели настоящего игрового пианино. С каждым вращением вы создаете мелодию своей победы, оставляя след в истории этой увлекательной саги.
Brillx casino — это бескрайний космос игровых возможностей. Здесь вы можете стать магом карт или виртуальным ковбоем рулетки. Это место, где даже самые неожиданные комбинации приносят грандиозные выигрыши. Откройте для себя игровой университет, где каждая ставка — это урок в искусстве азарта, а каждый джекпот — это диплом мастера удачи.
Зайдите на официальный сайт Brillx casino и откройте двери в мир, где действительность становится мифом, а миф — реальностью. Brillx casino — это путеводная звезда во мраке игровых просторов. Вам уже готовится красный ковер из карточных мастей и блеска монет. Отправьтесь в незабываемое приключение азарта и наслаждения прямо сейчас!
Вавада казино вход
https://pcmdaily.com/profile.php?lookup=158177
Здесь вы найдете не только знакомые слоты, но и те, которые поразят своей уникальностью. Наша команда тщательно отбирает самые захватывающие и новаторские игры, чтобы каждый спин был настоящим открытием. Вавада Казино предлагает богатство вариантов – от классических фруктовых машин до современных слотов с захватывающими сюжетами.Добро пожаловать в удивительный мир азарта и больших выигрышей — Казино Вавада! Этот завораживающий игровой портал являет собой настоящий рай для любителей азартных развлечений. Если вы ищете официальный сайт, который предоставит вам возможность войти и окунуться в захватывающий мир азартных игр, то Вавада Казино станет вашим надежным спутником.
Some truly quality articles on this website , saved to favorites.
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
Покердом бездепозитный бонус 1000
Покердом бездепозитный бонус 1000
Покердом бездепозитный бонус 1000 – это как магический ключ к виртуальному казино, который отпирает дверь к миру высоких ставок и больших побед. Начните свой путь в азартной игре с бонуса без депозита и откройте для себя все прелести игры в покер с невероятными возможностями для выигрыша. Да, это не сон, это реальность – Покердом бездепозитный бонус 1000 ждет вас!Виртуозно разыгрывайте свои карты и стратегические ходы, используя свой Покердом бездепозитный бонус 1000. В этой азартной игре каждый ход – как важное решение в шахматной партии, и только вы вольны выбирать, какой путь в азартной стратегии следует выбрать. Даже самый маленький бонус может стать ключом к большим победам, а Покердом бездепозитный бонус 1000 – это ваш персональный билет к успеху!
Добро пожаловать на захватывающий виртуальный парадокс развлечений — официальный сайт brillx казино! Если вы ищете истинный оазис азарта и удовольствия, то этот игровой магнат стоит на вершине вашего списка. Brillx casino — это не просто казино, это своего рода архитектурное чудо в мире виртуальных игорных заведений.
Ощутите дыхание азарта на своей коже, когда вы прокручиваете барабаны игровых автоматов Brillx casino. Это как будто встреча с великаном удачи, который приглашает вас на праздник выигрышей и возможностей. С каждой ставкой, словно аккордами великого симфонического произведения, вы будете раскрывать новые грани вашего везения.
Brillx casino — это путь к успеху и волнению, усыпанный яркими бриллиантами азартных игр. Неважно, являетесь ли вы сторонником классических слотов или предпочитаете бросить вызов современным видео-играм, этот портал соткан из разнообразия и эксклюзивности. Виртуозная графика и захватывающие сюжеты приведут вас в виртуальные миры, где реальность и фантазия переплетаются в невероятной гармонии.
Играя в Brillx casino, вы становитесь участником легендарного турнира удачи. Ваши ставки — это виртуозные ноты в симфонии азарта, а барабаны игровых автоматов — это как будто панели настоящего игрового пианино. С каждым вращением вы создаете мелодию своей победы, оставляя след в истории этой увлекательной саги.
Brillx casino — это бескрайний космос игровых возможностей. Здесь вы можете стать магом карт или виртуальным ковбоем рулетки. Это место, где даже самые неожиданные комбинации приносят грандиозные выигрыши. Откройте для себя игровой университет, где каждая ставка — это урок в искусстве азарта, а каждый джекпот — это диплом мастера удачи.
Зайдите на официальный сайт Brillx casino и откройте двери в мир, где действительность становится мифом, а миф — реальностью. Brillx casino — это путеводная звезда во мраке игровых просторов. Вам уже готовится красный ковер из карточных мастей и блеска монет. Отправьтесь в незабываемое приключение азарта и наслаждения прямо сейчас!
pokerok
https://forum.chemodan.com.ua/profile/275204-vovanaruto/
PokerOK предлагает множество вариаций покера, начиная от традиционных и до совершенно экзотических. Здесь вы найдете Texas Hold’em, Omaha, 7 Card Stud, и многие другие варианты, которые даже вряд ли вам когда-либо встречались. Но это лишь вершина айсберга — PokerOK радует своих гостей также турнирами на любой вкус и уровень мастерства.Покер – это не только игра карт и вероятностей, это настоящее искусство управления рисками и анализа оппонентов. Если вы ищете идеальное место для своего покерного мастерства, вашим лучшим выбором будет официальный сайт PokerOK. В мире онлайн-гемблинга этот ресурс — настоящая находка для истинных ценителей азартных развлечений.
click this over here now
cat casino скачать
https://abitu.net/events/topic/view/event_id/4172/topic_id/7516/post_id/34545
Cat Casino предоставляет широкий выбор игровых автоматов – от классических до современных, от простых до сложных. Каждый слот – это уникальный мир, где сокровища ждут своего обладателя. Скачай игровые автоматы на свое устройство и окунись в океан разнообразия и волнения.Cat Casino — это не просто сайт для игры в азартные игры, это истинное шоу, где каждый спин на игровых автоматах может стать настоящим произведением искусства. В 2023 году мы снова удивляем своих гостей неординарной графикой и инновационными механиками игры. Это не просто казино, это виртуальная страна удовольствий, где каждый вращение барабана — это билет в мир фантазий и желаний.
ggpokerok
http://kladovka.forumkz.ru/viewtopic.php?id=16427
Шагните в мир страсти и мастерства с нами. Чем отличается PokerOK? Это не только официальный сайт, это своего рода атмосферная лавка, где вы сможете сыграть в покер с острыми противниками, разделяя волнение и эмоции, словно сидя за столом в роскошном казино. Ощутите неповторимую магию игры, словно легендарный алхимик, воплощающий мастерство в каждом своем движении.Ищете захватывающий мир азартных испытаний и уникальных стратегий? Присоединяйтесь к нам на PokerOK — истинном раем для покерных энтузиастов, где страсть к игре переплетается с передовыми технологиями. Откройте дверь в увлекательное покерное путешествие прямо сейчас.
I do not even know how I ended up here, but I thought this post was great. I don’t know who you are but definitely you’re going to a famous blogger if you aren’t already 😉 Cheers!
gama casino вход
http://forum.animal.by/profile/39589-pkazofb/
Играть бесплатно в онлайн казино на русском языке – это не просто возможность попробовать свою удачу без риска, это способ окунуться в пучину азартных эмоций даже без вложений. Gama Casino предоставляет вам эту невероятную возможность. Гама Казино делает ставку на ваше удовольствие и комфорт, предлагая уникальный опыт игры.Играть в Gama Casino – это не просто казино, это путешествие в мир азартных мечтаний, где каждый спин – это новый поворот в сюжете вашей игры. Игровые автоматы России откроют перед вами безграничные возможности для выигрыша и развлечения. Каждая машина в Gama Casino – это как глава в увлекательной книге, наполненной сюрпризами и азартом.
how to purchase pyridium where can i buy pyridium 200 mg pyridium 200 mg prices
Вавада казино онлайн
https://myelimu.com/User-pololoyy
Казино Вавада – это не просто игровая площадка, это целый игровой космос, где каждый спин барабанов слотов-онлайн превращает ваше время в захватывающее событие. Полная версия игровых автоматов бесплатно раскроет перед Вами мир невероятных возможностей. Неважно, новичок Вы или опытный игрок, Вавада Казино дарит радость от игры и шанс победить!Казино Вавада – это не просто игровая площадка, это атмосфера азарта, уникальных возможностей и незабываемых впечатлений. Здесь каждый спин, каждая ставка – это шанс изменить свою судьбу. Войдите в увлекательный мир Вавада Казино и станьте частью этой захватывающей истории выигрышей и успехов!
Добро пожаловать на захватывающий виртуальный парадокс развлечений — cat casino! Если вы ищете истинный оазис азарта и удовольствия, то этот игровой магнат стоит на вершине вашего списка. Cat casino — это не просто казино, это своего рода архитектурное чудо в мире виртуальных игорных заведений.
Ощутите дыхание азарта на своей коже, когда вы прокручиваете барабаны игровых автоматов Кэт казино. Это как будто встреча с великаном удачи, который приглашает вас на праздник выигрышей и возможностей. С каждой ставкой, словно аккордами великого симфонического произведения, вы будете раскрывать новые грани вашего везения.
Cat casino — это путь к успеху и волнению, усыпанный яркими бриллиантами азартных игр. Неважно, являетесь ли вы сторонником классических слотов или предпочитаете бросить вызов современным видео-играм, этот портал соткан из разнообразия и эксклюзивности. Виртуозная графика и захватывающие сюжеты приведут вас в виртуальные миры, где реальность и фантазия переплетаются в невероятной гармонии.
Играя в Cat casino, вы становитесь участником легендарного турнира удачи. Ваши ставки — это виртуозные ноты в симфонии азарта, а барабаны игровых автоматов — это как будто панели настоящего игрового пианино. С каждым вращением вы создаете мелодию своей победы, оставляя след в истории этой увлекательной саги.
Cat casino — это бескрайний космос игровых возможностей. Здесь вы можете стать магом карт или виртуальным ковбоем рулетки. Это место, где даже самые неожиданные комбинации приносят грандиозные выигрыши. Откройте для себя игровой университет, где каждая ставка — это урок в искусстве азарта, а каждый джекпот — это диплом мастера удачи.
Зайдите на официальный сайт Cat casino и откройте двери в мир, где действительность становится мифом, а миф — реальностью. Кэт казино — это путеводная звезда во мраке игровых просторов. Вам уже готовится красный ковер из карточных мастей и блеска монет. Отправьтесь в незабываемое приключение азарта и наслаждения прямо сейчас!
naturally like your web site however you need to take a look at the spelling on several of your posts. A number of them are rife with spelling problems and I find it very bothersome to tell the truth on the other hand I will surely come again again.
cat casino регистрация
https://forum.resmihat.kz/viewtopic.php?f=2&t=1024707&p=2335176
Добро пожаловать в удивительный мир азартных развлечений — Cat Casino, парадоксальное слияние элегантности и азарта. В этом 2023 году, Cat Casino стал бесспорным героем азартной индустрии, обещая незабываемые впечатления и бесконечное веселье. На Cat Casino, Кэт Казино вас ждет множество возможностей, будь вы опытный азартный игрок или новичок, только начинающий свой путь в мире карточных столов и игровых автоматов.Cat Casino — это не просто сайт для игры в азартные игры, это истинное шоу, где каждый спин на игровых автоматах может стать настоящим произведением искусства. В 2023 году мы снова удивляем своих гостей неординарной графикой и инновационными механиками игры. Это не просто казино, это виртуальная страна удовольствий, где каждый вращение барабана — это билет в мир фантазий и желаний.
Добро пожаловать на захватывающий виртуальный парадокс развлечений — cat casino официальный сайт! Если вы ищете истинный оазис азарта и удовольствия, то этот игровой магнат стоит на вершине вашего списка. Cat casino — это не просто казино, это своего рода архитектурное чудо в мире виртуальных игорных заведений.
Ощутите дыхание азарта на своей коже, когда вы прокручиваете барабаны игровых автоматов Кэт казино. Это как будто встреча с великаном удачи, который приглашает вас на праздник выигрышей и возможностей. С каждой ставкой, словно аккордами великого симфонического произведения, вы будете раскрывать новые грани вашего везения.
Cat casino — это путь к успеху и волнению, усыпанный яркими бриллиантами азартных игр. Неважно, являетесь ли вы сторонником классических слотов или предпочитаете бросить вызов современным видео-играм, этот портал соткан из разнообразия и эксклюзивности. Виртуозная графика и захватывающие сюжеты приведут вас в виртуальные миры, где реальность и фантазия переплетаются в невероятной гармонии.
Играя в Cat casino, вы становитесь участником легендарного турнира удачи. Ваши ставки — это виртуозные ноты в симфонии азарта, а барабаны игровых автоматов — это как будто панели настоящего игрового пианино. С каждым вращением вы создаете мелодию своей победы, оставляя след в истории этой увлекательной саги.
Cat casino — это бескрайний космос игровых возможностей. Здесь вы можете стать магом карт или виртуальным ковбоем рулетки. Это место, где даже самые неожиданные комбинации приносят грандиозные выигрыши. Откройте для себя игровой университет, где каждая ставка — это урок в искусстве азарта, а каждый джекпот — это диплом мастера удачи.
Зайдите на официальный сайт Cat casino и откройте двери в мир, где действительность становится мифом, а миф — реальностью. Кэт казино — это путеводная звезда во мраке игровых просторов. Вам уже готовится красный ковер из карточных мастей и блеска монет. Отправьтесь в незабываемое приключение азарта и наслаждения прямо сейчас!
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
покердом официальный сайт вход
покердом официальный сайт вход
покердом официальный сайт вход
gama casino играть
https://yandexforum.ru/member.php?u=240295
Гама Казино — это не просто бескрайний океан азартных игр. Это ваша судьба, которая в ваших руках. Насладитесь непередаваемыми ощущениями, играя в игровые автоматы, представленные на страницах Gama Casino. Мир азарта в России открывает свои объятия перед вами.Забудьте о рутине и суете, потому что на Gama Casino вы откроете для себя весь мир онлайн развлечений. Именно здесь, на сайте Гама Казино, вас ждут веселье и богатство, а рулетка станет мастером вашей судьбы.
ПокерДом на АНДРОИД
ПокерДом на АНДРОИД
ПокерДом на АНДРОИД
Реальные деньги ПОКЕРДОМ
Реальные деньги ПОКЕРДОМ
Реальные деньги ПОКЕРДОМ
Вавада казино сайт
https://investsocial.com/member/1414890-dookwa
Здесь вы найдете не только знакомые слоты, но и те, которые поразят своей уникальностью. Наша команда тщательно отбирает самые захватывающие и новаторские игры, чтобы каждый спин был настоящим открытием. Вавада Казино предлагает богатство вариантов – от классических фруктовых машин до современных слотов с захватывающими сюжетами.Ощутите адреналин, когда каждый спин барабанов будет наполнен предвкушением выигрыша на Vavada Casino. На нашем портале, где азарт витает в воздухе, вы найдете полную версию игровых автоматов, доступную абсолютно бесплатно. Пробуйте разные стратегии, оттачивайте свои навыки, и только потом принимайтесь за настоящий взлет на волне удачи.
покердом официальный
https://t.me/s/pokerdom_officialniy
покердом официальный
Сайт ПокерДом
Сайт ПокерДом
Сайт ПокерДом
покерок скачать
http://www.vikno.com.ua/altfm/profile.php?mode=viewprofile&u=22282
Скачать клиент для игры в покер на PokerOK – значит обрести билет в мир интриг и увлекательных раундов. Насладитесь моментами напряжения и волнения, когда решение принимается в одно мгновение, а карты скрывают последние тайны своих обладателей. Здесь каждая партия – это как дуэль между стратегами, где искусство блефа и анализа сливаются в захватывающем танце.PokerOK — не просто сайт, это целая вселенная покера. Виртуальные столы, где встречаются игроки со всех уголков мира, станут вашей ареной славы. Почувствуйте дух соперничества, где ваш интеллект и аналитические способности будут подвергнуты самому жаркому испытанию. Играйте на реальные ставки, ведь именно здесь рождаются настоящие победители.
Регистрация ПокерДом
Регистрация ПокерДом
Регистрация ПокерДом
Зеркало ПОКЕРДОМ
Зеркало ПОКЕРДОМ
Зеркало ПОКЕРДОМ
cat casino онлайн
https://sfx.thelazy.net/forum/general/3609/
Ощути магию азарта в каждом вращении барабана. Cat Casino – это не просто место, где можно играть в игровые автоматы, это мир, который оживает с каждым кликом. Каждый слот – это история, которая разворачивается перед твоими глазами, и ты можешь стать ее главным героем.Не хотите скачивать? Не проблема! Cat Casino также предоставляет вам возможность играть онлайн, не загружая ничего на свое устройство. Просто пройдите регистрацию на официальном сайте, и вы окажетесь в виртуальной реальности Кэт Казино, где каждый клик мыши может повернуть ваш мир вверх дном.
POKERDOM CASINO
https://t.me/s/pokerdom_vhod
POKERDOM CASINO
вход на Покердом
вход на Покердом
вход на Покердом
покерок сайт
https://dongfeng-club.ru/profile.php?section=about&id=7765
Чтобы присоединиться к увлекательному миру PokerOK, просто перейдите на официальный сайт, где вас ждет возможность скачать клиент для игры в покер. С ним вы окунетесь в виртуальное путешествие по захватывающему миру карт и фишек, где каждый ваш ход анализируется и обдумывается до мельчайших деталей.На PokerOK вы найдете бесчисленное количество уникальных возможностей. Команда профессионалов работает над тем, чтобы каждый игрок ощущал заботу и внимание. Будьте уверены, что ваша безопасность и конфиденциальность – наш приоритет. Откройте для себя мир разнообразных турниров и регулярных игр, где даже невероятные шансы могут привести к захватывающему успеху.
скачать покердом на андроид зеркало
скачать покердом на андроид зеркало
скачать покердом на андроид зеркало
казино gama
https://mazda-demio.ru/forums/index.php?autocom=gallery&req=si&img=3146
Так что, готовы взять свою ставку удачи? Gama Casino с удовольствием подарит вам билет в мир азартных впечатлений. Входите на официальный сайт Гама Казино и дайте себе шанс испытать дыхание настоящего азарта, словно вы впервые прокладываете путь в неизведанные земли возможностей.Как только вы преодолеете порог Gama Casino, вы ощутите вибрацию азарта, словно планета азартных игр приветствует вас своими руками. Гама Казино создано для тех, кто стремится к большему, для искателей уникальных ощущений и неповторимых моментов.
ПокерДом на АНДРОИД
ПокерДом на АНДРОИД
ПокерДом на АНДРОИД
Вавада казино официальный сайт
http://nash-forum.itaec.ru/member.php?u=21030&vmid=5517
Мы понимаем, что азарт – это страсть, влекущая вас в водоворот волнений и эмоций. Именно поэтому наш виртуальный портал оформлен с максимальным комфортом и удобством. От главной страницы до разделов с игровыми автоматами, Vavada Casino заботится о вашем удовольствии и простоте навигации.Здесь вы найдете не только знакомые слоты, но и те, которые поразят своей уникальностью. Наша команда тщательно отбирает самые захватывающие и новаторские игры, чтобы каждый спин был настоящим открытием. Вавада Казино предлагает богатство вариантов – от классических фруктовых машин до современных слотов с захватывающими сюжетами.
Реальные деньги ПОКЕРДОМ
https://t.me/s/igrat_na_dengi_pokerdom
Реальные деньги ПОКЕРДОМ
покердом официальный
покердом официальный
покердом официальный
промокоды казино cat 2023
https://chesspro.ru/guestnew/looknullmessage/?themeid=49&id=19&page=7777&indown=1
Скачайте волнение, загрузите эмоции и отправьтесь в незабываемое путешествие по миру Cat Casino, где каждый миг может стать вашим ключом к сокровищам азарта. Насладитесь этой игрой на полную катушку, ведь Cat Casino — это где рождаются победители, а Кэт Казино — ваш верный проводник в мир развлечений!Cat Casino заботится о вашей безопасности так же, как Кэт Казино заботится о своих маленьких мурлыках. Ваши данные и финансовые операции находятся под надежной защитой, благодаря нашей передовой технологии шифрования. Так что вы можете сосредоточиться только на одном — наслаждаться игрой и стремиться к победам.
покердом онлайн зеркало
https://t.me/s/pokerdom_online_mirror
покердом онлайн зеркало
вход на Покердом
вход на Покердом
вход на Покердом
покердом онлайн
покердом онлайн
покердом онлайн
скачать покердом на андроид зеркало
скачать покердом на андроид зеркало
скачать покердом на андроид зеркало
cheapest spiriva 9mcg spiriva 9 mcg over the counter cheapest spiriva 9mcg
покердом казино вход
покердом казино вход
покердом казино вход
покердом официальный сайт играть на деньги
https://t.me/s/pokerdom_official_play_for_money
покердом официальный сайт играть на деньги
покердом официальный сайт вход
покердом официальный сайт вход
покердом официальный сайт вход
покердом вход зеркало
покердом вход зеркало
покердом вход зеркало
вход в покердом
вход в покердом
вход в покердом
покердом игры
https://t.me/s/pokerdom_games
покердом игры
покердом слоты
покердом слоты
покердом слоты
покердом официальный
покердом официальный
покердом официальный
казино покердом официальный сайт
казино покердом официальный сайт
казино покердом официальный сайт
Покердом приложение
Покердом приложение
Покердом приложение
покердом официальный сайт играть
покердом официальный сайт играть
покердом официальный сайт играть
покердом вход в личный кабинет
https://t.me/s/Online_MirrorAce
покердом вход в личный кабинет
скачать покердом зеркало
скачать покердом зеркало
скачать покердом зеркало
казино покердом
казино покердом
казино покердом
официальный сайт покердом
официальный сайт покердом
официальный сайт покердом
ПокерДом на телефон
ПокерДом на телефон
ПокерДом на телефон
покердом сайт зеркало
https://t.me/s/mirror_site_pokerdom
покердом сайт зеркало
скачать покердом официальный сайт
https://t.me/s/skachat_pokerdom_oficialnyj_sajt
скачать покердом официальный сайт
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
ПокерДом на ПК
ПокерДом на ПК
ПокерДом на ПК
покердом зеркало онлайн
https://t.me/s/Mirror_PokerPro
покердом зеркало онлайн
казино покердом
казино покердом
казино покердом
Официальный сайт ПокерДом
Официальный сайт ПокерДом
Официальный сайт ПокерДом
покердом регистрация официальный сайт
https://t.me/s/Dom_Mirror_Player
покердом регистрация официальный сайт
ПокерДом Промокод
ПокерДом Промокод
ПокерДом Промокод
покердом покер скачать
покердом покер скачать
покердом покер скачать
казино покердом зеркало
казино покердом зеркало
казино покердом зеркало
address
Промокод ПокерДом
Промокод ПокерДом
Промокод ПокерДом
Промокод в ПокерДом
Промокод в ПокерДом
Промокод в ПокерДом
Промокод НА ПокерДом
Промокод НА ПокерДом
Промокод НА ПокерДом
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
Первый депозит ПокерДом
Первый депозит ПокерДом
Первый депозит ПокерДом
покердом зеркало рабочее на сегодня
покердом зеркало рабочее на сегодня
покердом зеркало рабочее на сегодня
missed 2 doses of prednisone can prednisone damage your kidneys cyclobenzaprine and prednisone
ОТЗЫВЫ О ПОКЕРДОМЕ
https://t.me/s/pokerdom_otzivy_kazino
ОТЗЫВЫ О ПОКЕРДОМЕ
покердом зеркало сегодня
https://t.me/s/pokerdom_zerkalo_segodnya1
покердом зеркало сегодня
Честные отзывы о ПокерДом
Честные отзывы о ПокерДом
Честные отзывы о ПокерДом
ПокерДом на андроид
https://t.me/s/pokerdom_naandroid
ПокерДом на андроид
Cool that really helps, thank you.
покердом зеркало на сегодня
https://t.me/s/pokerdom_zerkaloseg
покердом зеркало на сегодня
покердом казино скачать
https://t.me/s/pokerdom_kazino_skachat
покердом казино скачать
покердом казино официальный сайт
https://t.me/s/pokerdom_kazino_oficialnyj_sajt
покердом казино официальный сайт
покердом онлайн играть
покердом онлайн играть
покердом онлайн играть
2022 ПокерДом отзывы
https://t.me/s/pokerdom_2022
2022 ПокерДом отзывы
Отзывы игроков ПокерДом
Отзывы игроков ПокерДом
Отзывы игроков ПокерДом
cheap zanaflex 2 mg zanaflex over the counter zanaflex 2 mg without prescription
Бонусы за регистрацию на ПОКЕРДОМЕ
https://t.me/s/bonus_PokErDom
Бонусы за регистрацию на ПОКЕРДОМЕ
SPAM4D Situs Slot Gacor Hari Ini
prednisolone 30 mg tablets
SPAM4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
SPAM4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
this post
SPAM4D Situs Mudah Menang Hari Ini
Awesome! Its genuinely remarkable post, I have got much clear idea regarding from this post.
SPAM4D Situs Slot Gacor Hari Ini
viagra 2 where can i buy viagra canada sildenafil price uk
reglan 10 mg without prescription reglan without prescription reglan 10 mg coupon
viagra 25mg generic viagra europe can you buy viagra online without a prescription
paypal viagra australia generic viagra sildenafil citrate sildenafil 5 mg price
SPAM4D Pola Slot Gacor Hari Ini
viagra gel tabs buying viagra without prescription discount pharmacy sildenafil
I just like the helpful information you provide in your articles
SPAM4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
to say concerning this paragraph, in my view its
best price for viagra in uk viagra online with prescription order viagra professional
SPAM4D Situs Slot Gacor Hari Ini
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
wellbutrin target pharmacy mail order pharmacies low dose naltrexone online pharmacy
inderal pharmacy online pharmacy uk propecia one click pharmacy propecia
Pretty! This has been a really wonderful post. Many thanks for providing these details.
wellbutrin 150mg generic
how to purchase leflunomide leflunomide otc cheapest leflunomide
zofran 4 mg united states
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
cialis shop online tadalafil generico farmacias del ahorro cialis off patent
cialis patent buy cialis canada cialis black 800
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
fexofenadine otc fexofenadine 180mg over the counter fexofenadine prices
SPY4D Pola Slot Gacor Hari Ini
Быстромонтируемые строения – это актуальные системы, которые отличаются высокой скоростью строительства и гибкостью. Они представляют собой конструкции, заключающиеся из предварительно произведенных компонентов или узлов, которые способны быть скоро смонтированы на месте строительства.
Быстровозводимые конструкции из сэндвич панелей владеют податливостью также адаптируемостью, что дает возможность просто изменять и модифицировать их в соответствии с потребностями клиента. Это экономически эффективное а также экологически стабильное решение, которое в крайние лета заполучило обширное распространение.
zanaflex 2 mg otc
is cephalexin ok for dogs dog cephalexin side effects keflex allergy to penicillin
SPY4D Situs Slot Gacor Hari Ini
para q sirve cephalexin fish mox cephalexin cephalexin and nexplanon
singulair 5 mg usa
bactrim price
can pregnant women take keflex keflex side effects eye what is another name for keflex
cephalexin 500mg for dogs is cephalexin and keflex the same thing is cephalexin safe for pregnancy
BINGO4D Situs Slot Gacor Hari Ini
KLIX4D Pola Slot Gacor Hari Ini
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
BINGO4D Situs Mudah Menang Hari Ini
KLIX4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
KLIX4D Angka Keluaran Togel Hk, SGP , SDY
YOI4D Situs Slot Gacor Hari Ini
flomax for kidney stones
YOI4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
amoxicillin sinus infection amoxicillin for ear infection how many days amoxicillin 500 mg capsule
augmentin with zithromax augmentin dla dziecka na anginД™ augmentin 625mg for children
augmentin and boils augmentin hasznГЎlati utasГtГЎs augmentin compresse composizione
doxycycline hyclate vs amoxicillin does amoxicillin reduce fever amoxicillin 250 mg dosage for adults
can you give amoxicillin to cats can you take mucinex with amoxicillin amoxicillin for bacterial infection
YOI4D Situs Slot Gacor Hari Ini
what is amoxicillin clavulanate missed a dose of amoxicillin does amoxicillin go bad
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
POL4D Angka Keluaran Togel Hk, SGP , SDY
prednisone and sudafed how quickly does prednisone work can i take aspirin with prednisone
do i take doxycycline with food can you drink alcohol while taking doxycycline doxycycline used for uti
doxycycline for dogs ear infection sinus infection doxycycline does doxycycline treat sinus infections
POL4D Angka Keluaran Togel Hk, SGP , SDY
ciprofloxacin 500mg tablets shelf life ciprofloxacin trade name ciprofloxacin diverticulitis
how long do you stay on prednisone for pmr how many hours apart should you take prednisone 20 mg prednisone
I don’t commonly comment but I gotta tell thankyou for the post on this perfect one : D.
vitamin c and ciprofloxacin ciprofloxacin uses for uti ciprofloxacin 500 with alcohol
POL4D Pola Slot Gacor Hari Ini
POL4D Angka Keluaran Togel Hk, SGP , SDY
POL4D Angka Keluaran Togel Hk, SGP , SDY
should azithromycin be taken with food erythromycin azithromycin azithromycin in store
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
furadantin 100mg generika
POL4D Pola Slot Gacor Hari Ini
generic tadalafil tablet or pill photo or shape cialis 25mg cialis for women
cialis super active vs regular cialis prices cialis cialis cheap generic
50 mg tadalafil cialis no prescription arizona soft gel cialis
zovirax bestellen
POL4D Situs Slot Gacor Gampang Menang Maxwin Hari Ini
purchase viagra usa order sildenafil 50 mg sildenafil online usa
female viagra capsule usa pharmacy online viagra can i buy generic viagra in canada
online viagra usa us online viagra prescription viagra online rx pharmacy
mynet sunar
POL4D Angka Keluaran Togel Hk, SGP , SDY
prednisolone online pharmacy
POL4D Angka Keluaran Togel Hk, SGP , SDY
generic cialis buy cialis without doctor prescription cialis 20 mg sell
top web source for cialis cialis and dapoxetine canada buy cialis brand
epivir billig bestellen
allopurinol ohne rezept kaufen
triamcinolone online-apotheke bestellen
essay writing service best how to start an argumentative essay example write my essay students
essay format cool essay original essay writing service
my essay writer website for essay writing buy essay
essay writing helper how to do an essay custom essay help
I’m often to blogging and i really appreciate your content. The article has actually peaks my interest. I’m going to bookmark your web site and maintain checking for brand spanking new information.
the best essay writer how to write a essay essay with citations example
This was beautiful Admin. Thank you for your reflections.
acyclovir cream 5% generika billig
abilify online-apotheke kaufen
doxycycline billig kaufen
donepezil 5mg coupon
mba application essay writing service transition words for essay essay on help
buy essays good essay starters sat essay scores
what can i write my essay on college admission essay write my essay for me cheap
reflective essay examples recommended essay writing service buy cheap essays
troy hill pharmacy online tramadol pharmacy to buy viagra does target pharmacy carry oxycodone
best drug store foundation vicodin online pharmacy permethrin uk pharmacy
us pharmacy tramadol online rx pharmacy richland washington celebrex pharmacy coupon
cymbalta pharmacy checker Clomid online pharmacy jobs
zithromax online bestellen
cipro
buy tetracycline online pharmacy pharmacie canadienne viagra from boots pharmacy
Стабилизатор — необходимое устройство для защиты от повышенных напряжений
стабилизаторы напряжения http://stabrov.ru.
Стабилизатор напряжения — качественная защита для вашего оборудования
стабилизаторы напряжения http://stabrov.ru.
best online pharmacy to buy viagra philidor rx pharmacy 365 pharmacy viagra
modafinil tablets buy
Зачем нужен стабилизатор напряжения для дистанционного управления электрооборудованием?
электронный стабилизатор напряжения http://www.stabilizatory-napryazheniya-1.ru/.
valtrex 1000 mg usa
Релейный стабилизатор напряжения является одним из простых и наиболее распространенных типов стабилизаторов. В этой статье мы рассмотрим его принцип работы, основные преимущества и недостатки.
стабилизаторы напряжения на 15 квт http://www.stabilizatory-napryazheniya-1.ru.
acyclovir cream 5% ohne rezept billig
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
mestinon 60mg without prescription
walgreen pharmacy hours acyclovir online pharmacy valium pharmacy review
mestinon medication
target pharmacy lexapro cost viagra pharmacy rx one online pharmacy store
yonkers pharmacy oxycodone online pharmacy exam reputable online pharmacy levitra
I think this post makes sense and really helps me, so far I’m still confused, after reading the posts on this website I understand.
For the reason that the admin of this site is working, no uncertainty very quickly it will be renowned, due to its quality contents.
tadalafil grapefruit interaction tadalafil controlled can i take tamsulosin and tadalafil at the same time
allopurinol ohne rezept billig
how much tadalafil tadalafil from mexico tadalafil us next day delivery
what is the difference between cialis and tadalafil tadalafil grapefruit interaction tadalafil dosage for erectile dysfunction
tadalafil t5 generic name for tadalafil tadalafil nitric oxide
Great information shared.. really enjoyed reading this post thank you author for sharing this post .. appreciated
how long does 20mg tadalafil last tadalafil cipla headache from tadalafil
tadalafil pill identifier tadalafil uk tadalafil women
levofloxacin pharmacy
Lainaa 25000 euroa
tadalafil? tadalafil good rx tadalafil kidney
Pretty! This has been a really wonderful post. Many thanks for providing these details.
tadalafil prostate tadalafil cut in half can you take viagra and tadalafil together
Nice post. I learn something totally new and challenging on websites
why is tadalafil so expensive tadalafil release date tadalafil liquid drops
daniel, lee tadalafil tadalafil and mdma tadalafil similares
can you overdose on tadalafil sildenafil or tadalafil which is better v tada super tadalafil tablets 20 mg
is tadalafil viagra tadalafil erfahrungen tadalafil adcirca info
Good post! We will be linking to this particularly great post on our site. Keep up the great writing
symbicort 160/4.5mcg ordine a Torino
Prestito di 750 euro
I carry on listening to the reports speak about getting free online grant applications so I have been looking around for the most excellent site to get one. Could you advise me please, where could i get some?
how much is tadalafil 20 mg how to take tadalafil 20 mg what is the best time to take tadalafil
lanoxin farmГЎcia online ordem em Manaus
tadalafil vs. sildenafil tadalafil taste tadalafil alzheimer
cialis vs generic tadalafil reviews mixing sildenafil and tadalafil tadalafil side efect bruising
can i get pregnant if my husband use tadalafil tadalafil 2.5 mg price difference between tadalafil and sildenafil
Some truly quality articles on this website , saved to favorites.
powiД…zana witryna
500 Euro Kredit
se mer info
tadalafil oral jelly buy tadalafil 20 mg do i need prescription for tadalafil
tadalafil heart rate tadalafil pills sildenafil vardenafil and tadalafil
tadalafil accord 20 mg tadalafil maxigra how long does 5mg tadalafil last
LГҐn 100000 kr
Lainaa 300000 euroa
lien Web
Prestito 12 mila euro
clicca qui per saperne di piГ№
25000 euro lenen
differin
wellbutrin coupon
Lee esto
nimotop
credit 27000 euros
prinivil
LГҐn 500000 kr
I very delighted to find this internet site on bing, just what I was searching for as well saved to fav
Лучшие онлайн казино России на рубли
https://carcam-official.ru/
You’re so awesome! I don’t believe I have read a single thing like that before. So great to find someone with some original thoughts on this topic. Really.. thank you for starting this up. This website is something that is needed on the internet, someone with a little originality!
Онлайн казино на реальные деньги для андроид
https://gozee.ru/
zanaflex
LГҐn 30000 kr
besГёg webstedet
Rüyada 3 Tane Cenaze Görmek Ne Anlama Gelir?
Rüyada Altın Kol Saati Görmek Nedir?
clavulanate 750/250mg kaufen
Онлайн казино на реальные деньги для андроид
https://gozee.ru/
Hi there to all, for the reason that I am genuinely keen of reading this website’s post to be updated on a regular basis. It carries pleasant stuff.
quetiapine generika
to say concerning this paragraph, in my view its
LГҐn 90000
İnstagramda satışlarınızı arttırmanın yolu takipçi satın almaktır, bunun için igtakipcim.com
trouver
This was beautiful Admin. Thank you for your reflections.
cost of baclofen
toradol for headaches
buy vermox in usa
order vermox online
compare colchicine prices
trazodone buy online
can i order paxil online
how to get albuterol cheap
allopurinol 100mg tablets
zithromax cost uk
synthroid mexico
propecia 1
inderal 10 mg price
cialis super active
zoloft 2004
order levitra online in usa
gabapentin 25mg tablets
I think the content you share is interesting, but for me there is still something missing, because the things discussed above are not important to talk about today.
gabapentin 1000mg
paxil for menopause
doxycycline antimalarial
bactrim ds price
PЕЇjДЌka 20000 KДЌ
how to get propecia in south africa
vardenafil 10 mg tablet
ivermectin 1% cream generic
price of accutane without insurance
doxycycline uk pharmacy
cialis generic
albuterol 0.42
augmentin drug
prescription propecia cost
en cliquant ici
budesonide 3 mg cap
over the counter robaxin
orlistat no prescription
zoloft 50mg tabs
zithromax buying
lisinopril 20 mg generic
baclofen 10 mg discount
zithromax over the counter uk
doxycycline order online canada
where can i order doxycycline
suhagra 50
cialis buy online usa
augmentin brand name india
prescription retin a products
where to buy generic propecia online
neurontin singapore
cheap prednisolone
amoxil generic brand name
proair albuterol sulfate online
clonidine hcl for sleeping
can you buy prozac
казино Pokerdom
официальный сайт Покердом
vermox online pharmacy
fluoxetine uk
buy doxycycline 500mg
best rogue online pharmacy
colchicine pills uk
diflucan usa
fluoxetine 5 mg tablets
dapoxetine 60mg buy
doxycycline 75 mg tab
viagra 50mg online
vermox for sale
experimente isso
LГҐna 80000 kr
fluoxetine price nz
500 mg amoxicillin 40 tablet
cost of finasteride
lasix 10 mg tablet
https://anlamlisozleroku.blogspot.com
zithromax 250mg
suhagra 50 mg tablet
medicine trazodone 50 mg
buy augmentin 1000 mg online
robaxin 500mg generic
vermox online pharmacy
buy propranolol without prescription
cost of celexa 20 mg
baclofen 5 mg tablet price
clonidine cost
synthroid 0.125mg tab
lioresal drug
how to get retin a cream
desyrel 100 mg
buy lyrica online australia
silagra 50 mg
amoxicillin rx coupon
buy clonidine usa
amoxicillin 500 tablet
how to get gabapentin
Try to slowly read the articles on this website, don’t just comment, I think the posts on this page are very helpful, because I understand the intent of the author of this article.
albuterol prescription
where to buy vardenafil online
generic propecia cheap
lisinopril 20 25 mg
zyban price us
lisinopril generic cost
pepcid 50mg
propecia pharmacy cost
cialis 5mg price comparison
prednisolone 25mg
propecia discount
prozac pill
robaxin otc canada
bactrim ds 800
ivermectin buy online
propecia 1mg
amoxicillin online pharmacy uk
zoloft canada
synthroid 0.15
paroxetine 0.37
clonidine 50 mcg
where to buy generic propecia
I don’t commonly comment but I gotta tell thankyou for the post on this perfect one : D.
how much is doxycycline cost
cost of paxil 30 mg
finasteride 5mg coupon
allopurinol 100 mg generic
cialis mexico
lyrica 15 mg
inderal australia
amoxicillin prescription discount
doxycycline 500mg tablets
paxil 93712
pulmicort united states
baclofen 10 mgs no prescription
budesonide pills
propranolol 10mg
augmentin xr 1000 mg
retin a cream buy online nz
augmentin tablets 1g
neurontin 100mg caps
vermox tablets where to buy
ventolin prescription australia
buy cheap prednisolone
best price for effexor 75mg
lyrica 900 mg
synthroid 137 mcg tablet
zyvox 0.625mg
prozac 50 mg cost
where can i get accutane cheap
lexapro for sale
where to buy fildena 100
viagra daily use
augmentin 875 mg 125 mg tablets
20mg zoloft
buy lisinopril without a prescription
canadian pharmacy doxycycline
propranolol 60 mg price
reputable online pharmacy uk
brand name clonidine prescription
3000 euro lenen
drug finasteride
tretinoin 1 uk
order azithromycin 500mg online
amoxil buy
hydroxychloroquine 200 mg tab
казино без верификации документов и без паспорт
https://clickrabota.ru
can i buy accutane online
accutane cost uk
furadantin
freespin nasıl kullanılır
Free Spin Bonusu Nasıl Kullanılır
prednisone buy
levitra purchase cheap
levitra 10 mg
how much is paxil pills
azithromycin capsules
600 mg zoloft
cheapest pharmacy to fill prescriptions without insurance
safe canadian pharmacies
albuterol pills
retin a cream 1 % for wrinkles no prescription
amoxicillin 2000 mg
buy lexapro online india
ventolin coupon
colchicine generic pharmacy
paroxetine 40
fluoxetine 80 mg daily
buy brand name lexapro
generic propecia online pharmacy
buy cialis cheap online
propranolol 80 mg capsules
augmentin 875 prescription
amoxicillin 500mg capsule over the counter
cheap augmentin online
zoloft 75 mg
toradol 10mg cost
levitra 20mg pills
40000 euro lenen
fincar
accutane cost in canada
where can i get vermox
clonidine bipolar disorder
lyrica buy online uk
trazodone 150 mg pill
vermox pills
medication lisinopril 20 mg
silagra 100 mg india
buy diflucan without prescription
I appreciate you sharing this blog post. Thanks Again. Cool.
levitra 20mg for sale
colchicine discount coupon
doxycycline 40 mg capsules
xenical 84
doxycycline for sale uk
suhagra 25 mg price in india
trazodone buy
lasix 20 mg daily
doxycycline online canada
clomid online no prescription
viagra cost uk
robaxin 500mg cost
diflucan 50mcg
buy levitra 20 mg
cost of accutane
allopurinol 100mg price
plaquenil generic coupon
quineprox 40 mg
synthroid 15mcg
05 clonidine
order propecia online
synthroid 0.5
fluoxetine 10 mg discount
lowest price tretinoin cream
synthroid 75 mg cost
dapoxetine online pharmacy india
diflucan 300 mg
clonidine capsule
levitra cost australia
cost of sildenafil
prozac brand price
buy zithromax 250mg
augmentin cream
prednisone brand name uk
lisinopril 10 mg cost
baclofen brand name in canada
cost of levitra generic
combivent respimat package insert
80 mg celexa daily
azithromycin 500 mg tablet buy online
accutane 10 mg discount
augmentin 1 mg
gabapentin in canada
cost of prescription orlistat
buy lasix tablets
bactrim 1600
propranolol prescription canada
buy amoxicillin over the counter uk
buy phenergan uk
discount levitra pills
amoxicillin price australia
budecort 0.5 mg
phenergan nz buy
doxycycline 30mg
paxil online uk
buy diflucan prescription med
propecia 5mg india
buy generic propranolol
irbesartan 300mg
toradol 10mg cost
order diflucan
zestril generic
how to purchase cialis online
accutane 40
propranolol 2.5 mg
lyrica generic
buy accutane online uk
cheap silagra
dapoxetine 1mg
otc clonidine
order prednisone without prescription
buy retin a gel uk
dapoxetine order online
medicine furosemide 20 mg
order accutane from canada
0.75 mg albuterol
generic deltasone
vermox otc
pharmacy prices for doxycycline
prescription albuterol online
propranolol over the counter australia
can you buy augmentin over the counter
vermox otc uk
buy amoxicillin uk
buy flucanozole
vermox over the counter usa
1000 euro lenen
budesonide 3 mg price india
brand retin a cream
where to get accutane in south africa
can you buy sildenafil over the counter in uk
amoxicillin fast delivery
fildena india
bupropion tablets uk
finasteride 1mg
prednisone 8 mg
baclofen 40 mg
allopurinol cost uk
cheap accutane for sale
augmentin australia
price of tadalafil 20mg
cost of phenergan
generic augmentin prices
buy inderal online usa
can you buy gabapentin online
doxycycline 300 mg
ventolin 4 mg tabs
albuterol 0.42
can you buy baclofen over the counter
phenergan 25 mg over the counter
generic vermox
how much is toradol
lisinopril drug
levitra rx
amoxicillin 30 capsules price
amoxicillin 650 mg price
synthroid for sale online
fluoxetine brand name india
cost of synthroid medication
azithromycin 1g for sale
desloratadine online bestellen
cheap propecia for sale
doxycycline 100
buy accutane in india
lisinopril 5 mg pill
augmentin capsules 875 mg
levitra 20mg prices
lasix 200 mg
levitra 60 mg
women viagra online no prescription
Холоп 2 скачать торрент. Холоп фильм смотреть онлайн бесплатно в хорошем качестве. Актеры фильма холоп. Холоп фильм 2024 смотреть. Холоп 2 дата выхода в россии в кинотеатрах. Холоп 2 смотреть онлайн бесплатно. Холоп фильм смотреть онлайн бесплатно в хорошем качестве. Холоп 2. Холопы это.
clonidine catapres
livitra
generic lyrica 2017
buy levitra online canada
propranolol 10 mg for sale
how can i get dapoxetine
prozac australia
lisinopril tabs 10mg
where to buy finasteride
synthroid tablets 100 mcg
credit 8500 euros
buy furosemide online australia
viagra pills for sale canada
vermox 100
order zoloft uk
ventolin 2.5
augmentin tablets
vardenafil 10 mg
propranolol rx
finasteride proscar
synthroid cost canada
generic for augmentin
plaquenil malaria
bactrim canada
inderal 20 mg tablet
prednisolone tablets
canada augmentin
where can i buy doxycycline without prescription
retin a 025
propranolol online
levitra 500mg
150 mg diflucan
credito 8000 euros
sildenafil price australia
propecia generic no prescription
gabapentin buy usa
propranolol how to get
ventolin 2mg
diflucan from india
budesonide 200
cialis compare discount price
stromectol covid
buy fildena 50 mg
clonidine sale
inderal 5 mg tablet
price of augmentin 625mg
doxycycline capsule 100mg price
purchase diflucan online
how to get propranolol prescription
finasteride on sale no prescription Specific targeting of lung destruction in LAM and other chronic lung diseases needs better understanding of the pathological mechanisms involved
amoxicillin 500 mg prescription
prednisolone buy online uk
vardenafil online india
buy zithromax uk
order zithromax online canada
generic vardenafil canada
50mg citalopram
amoxicillin 250 mg tablet price
robaxin for sale online
super avana
how to buy clomid in canada
amoxicillin no script
citalopram 10 mg pill
cialis for bph insurance coverage
Prestito di 4500 euro
clonidine 0.1 mg pill
clomid australia
synthroid 112 mcg tab
retin a mexico price
generic 450 bupropion
how to buy azithromycin 250 mg
bactrim online canada
cost of generic cialis
tretinoin india pharmacy
augmentin 1 mg
otc atarax
rate online pharmacies
dilaudid online pharmacy
4 azithromycin cream
dapoxetine india online
50mg citalopram
buy levitra 5mg
synthroid brand name price
6000 euro lenen
zoloft 100 mg
price of propecia in canada
diflucan coupon
cheap wellbutrin
world best pharmacy online store reviews
sildenafil 48 tabs 50 mg price
stromectol price in india
methocarbamol robaxin
cost of 1 viagra pill
quineprox 30 mg
how much is lyrica cost
wellbutrin 10 mg
cymbalta 60 mg purchase
buy albuterol without prescription
can you buy albuterol online
augmentin 125mg
levitra over the counter uk
best generic sildenafil
vermox usa
tadalafil tablets 40mg
accutane pills
cialis supr active
cheap prednisolone tablets
best tretinoin coupon
where to buy finasteride online
doxycycline price canada
innopran xl online
buy accutane online 30mg
how to order doxycycline
amoxicillin no prescription
synthroid 250 mcg price
buy vermox over the counter
can i buy prozac online
accutane 40 mg daily
tadalafil half life
best prices sildenafil
vermox 500mg for sale
10 mg prednisone daily
lexapro
canadian pharmacy service
50 prednisolone
prices for cialis
tretinoin 0.01 gel price
accutane from mexico
credit 7000 euros
ivermectin 18mg
how much is gabapentin 300 mg
how much is bactrim
online pharmacy drop shipping
buy synthroid without a prescription
prednisone 30 mg
top 10 online pharmacy in india
doxycycline 100mg without a prescription
buy silagra uk
allopurinol 100mg uk
furosemide coupon
prinivil drug
robaxin 500
neurontin 600 mg cost
cheap brand levitra
buy tretinoin from canada
desyrel drug
LГҐna 7000 kr
buy propecia australia
purchase neurontin
Eylül Online
paroxetine 20 mg tablet
buy cheap levitra online
glucophage online
over the counter amoxicillin australia
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
buy amoxil online uk
augmentin online buy without prescription
price of augmentin 625
neurontin 100 mg cap
levitra online canada
zyloprim allopurinol
allopurinol 100mg prices
augmentin 800
diflucan for sale
online pharmacy europe
synthroid 0.01mg
clonidine iv
effexor-xr
diflucan 300 mg
cymbalta otc
price of diflucan in south africa
90 vermox
cheapest pharmacy
cialis men
buy suhagra tablet
zoloft rx online
propecia generic version
50 mg lisinopril
amoxicillin buy no prescription
zoloft online india
cost of levitra in canada
accutane 40 mg cost
sildenafil medication
tadalafil 10mg cost
amoxil over the counter
cheap neurontin
accutane 30 mg
buy zoloft from canada
prozac from canada
lyrica 50 mg capsule
where to buy augmentin
Prestito di 600 euro
amoxil over the counter
doxycycline rx coupon
I gotta favorite this web site it seems very helpful invaluable
ventolin australia prescription
augmentin 625
21 amoxicillin 500mg capsules
price of lyrica 100 mg
can i buy amoxicillin
shopping for cialis
where to buy crestor 10 mg
desyrel 100 mg tab
order difucan
zyban coupon
vermox
colchicine 0.5 mg price
where can i buy vardenafil
buy albuterol canada
clonidine hcl 0.1mg tablets
synthroid 25 mcg tablet
where can you get toradol
bactrim 400 800 mg
clonidine generic brand
price of toradol
order accutane over the counter
generic cialis in australia
silagra canada
online doctor prescription cialis
zoloft without script
baclofen 30 mg tab
doxycycline 150 mg price
propecia 1mg cost
buy allopurinol 300 mg online uk
real viagra online usa
LГҐn 500000 kr
synthroid 375 mcg
norvasc 100mg
synthroid 1.25 mg
diflucan pill over the counter
bupropion sr price
best reviewed tadalafil site
augmentin 625 tablets
where can you buy retin a over the counter
tretinoin 0.11
accutane singapore cost
doxycycline tablet
levitra pills canada
ventolin coupon
doxycycline 300 mg cost
buy viagra india
synthroid 0.05 mg
propecia 0.5 mg
orlistat tablets in india
clavulanate 750/250mg online
how much is clomid uk
synthroid 137 mcg price
amoxicillin capsules from mexico
sildenafil 55mg
order vermox online canada
albuterol 108 mcg
cialis san diego
tretinoin cream uk pharmacy
online propecia prescription
accutane 10mg price
vocГЄ pode tentar aqui
neurontin otc
cialis doses
buy ozempic
original cialis pills
semaglutide without prescription
rybelsus lose weight
Nice post. I learn something totally new and challenging on websites
Prestito di 700 euro
rybelsus tablets for weight loss
semaglutide buy uk
buy semaglutide online from india
rybelsus 21 mg
how long does cialis take to work
semaglutide lose weight
wegovy semaglutide tablets 3mg
metronidazole medicament
rybelsus retail price
rybelsus diabetes medication
buy ozempic
noroxin 500mg
semaglutide oral medication
semaglutide lose weight
buy ozempic online no script needed
ozempic tablets 7 mg
generic rybelsus for weight loss
buy semaglutide for weight loss
buy ozempic
credit 5500 euros
buy ozempic online from india
rybelsus diabetes
buy ozempic uk
rybelsus pill
where can i buy ozempic
buy ozempic online
ozempic coupon
wegovy semaglutide tablets cost
rybelsus 21 mg
buy ozempic online cheap
wegovy order
rybelsus mexico
sulfamethoxazole-tmp allergic reaction
wegovy lose weight
wegovy 14
semaglutide pill
buy wegovy in canada
rybelsus mexico
ozempic tablets for weight loss
Prestito 16 mila euro
semaglutide tablets cost
buy semaglutide uk
semaglutide uk
fortsatte
semaglutide without prescription
wegovy buy from canada
buy ozempic from canada online
rybelsus 7mg
wegovy xl
buy ozempic oversees
generic wegovy for weight loss
where can i buy semaglutide
buy semaglutide online cheap
where to buy semaglutide online
buy semaglutide online from india
rybelsus buy from canada
rybelsus order
semaglutide without prescription
ozempic tablets 7 mg
buy rybelsus in canada
buy semaglutide for weight loss
rybelsus tab 14mg
buy rybelsus
semaglutide rx
semaglutide from canada
buy wegovy online no script needed
buy semaglutide online canada
ozempic coupon
ozempic semaglutide
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
LГҐn 60000 kr
buy semaglutide in mexico
ozempic cost
ozempic semaglutide tablets cost
generic semaglutide cost
buy wegovy in canada
rybelsus australia online
wegovy rx
order semaglutide
buy wegovy in mexico
buy wegovy in canada
buy semaglutide online
semaglutide over the counter
rybelsus 7mg
ozempic coupon
semaglutide 7 mg
rybelsus semaglutide tablets 3mg
semaglutide injection
semaglutide diabetes
ozempic semaglutide
rybelsus 3 mg
rybelsus tablets 7 mg
semaglutide where to buy
ozempic pill form
rybelsus online order
where to buy ozempic
buy ozempic uk
wegovy weight loss
semaglutide coupon
rybelsus buy from canada
rybelsus for sale
rybelsus online prescription
epivir 5mg
wegovy pill form
semaglutide weight loss
generic wegovy
LГҐn 30000 kr
generic semaglutide
semaglutide medication
semaglutide pill form
wegovy mexico
ozempic injections
wegovy generic cost
ozempic tab 3mg
wegovy for sale
rybelsus tablets buy
wegovy semaglutide tablets cost
rybelsus 7 mg
semaglutide from canada
buy rybelsus canada
wegovy best price
purchase semaglutide
wegovy online uk
generic ozempic
rybelsus semaglutide tablets
rybelsus without prescription
semaglutide rybelsus
ozempic tab 7mg
wegovy order
semaglutide injection
Greetings! I found your site while researching ‘early pregnancy scan services near me.’ The wealth of information is impressive. Currently on the lookout for an ‘early scan near me,’ I’d love to hear your expert opinion or any suggestions you may have. Thanks a bunch!
rybelsus 3 mg tablet
semaglutide online prescription
wegovy pill form
buy semaglutide online cheap
semaglutide online cheap
semaglutide diabetes
semaglutide 21 mg
LГҐn 8000
semaglutide online
ponstel 100mg
rybelsus online pharmacy
semaglutide tablet
buy wegovy in canada
buy ozempic oversees
ozempic
order ozempic
This is a topic close to my heart cheers, where are your contact details though?
ozempic tablets for weight loss cost
buy semaglutide canada
I truly appreciate your technique of writing a blog. I added it to my bookmark site list and will
ozempic semaglutide tablets cost
semaglutide tablets 7 mg
buy semaglutide for weight loss
rybelsus tablets for weight loss
rybelsus drug
semaglutide pills
semaglutide tab 3mg
where to buy ozempic online
semaglutide canada pharmacy
wegovy xl
rybelsus best price
wegovy medicine
buy rybelsus online from india
buy wegovy
wegovy 21 mg
semaglutide buy online
purchase rybelsus
semaglutide 3 mg
semaglutide wegovy
cephalexin 500mg prices
rybelsus 14mg tablets
wegovy pills
rybelsus sale
semaglutide xl
generic rybelsus
Заключение диплома необходимо для занятости на пост. Иногда появляются сценарии, когда предыдущий документ неприменим для профессиональной деятельности. Покупка документа об образовании в Москве разрешит этот вопрос и предоставит процветание в будущем – https://kupit-diplom1.com/. Существует много причин, стимулирующих приобретение документа об образовании в Москве. После некоторого времени в карьере внезапно может потребоваться университетский диплом. Работодатель имеет право менять требования к работникам и принудить вас сделать выбор – взять диплом или потерять работу. Учеба на дневном отделении вызывает затраты времени и усилий, а дистанционное обучение — потребует средства для проведения экзаменов. В подобных обстоятельствах более выгодно закупить готовую копию. Если у вас уже есть опыт в выбранной сфере и овладели необходимыми компетенциями, нет необходимости затрачивать время на обучение в университете. Плюсы заказа диплома воспринимают мгновенное изготовление, полное совпадение с оригиналом, доступные цены, гарантию трудоустройства, возможность выбора оценок и простую доставку. Наша компания предоставляет возможность каждому человеку получить желаемую профессию. Цена изготовления свидетельств достойна, что делает эту покупку доступной для всех.
ozempic online
buy wegovy online no script
rybelsus order
rybelsus drug
semaglutide 3mg
semaglutide 3mg
wegovy online uk
wegovy australia
buy ozempic
semaglutide from canada
rybelsus tab 7mg
remeron 30mg for sale
purchase semaglutide
ozempic tab 7mg
semaglutide 14mg
rybelsus 7 mg tablet
rybelsus 3 mg
semaglutide generic cost
semaglutide canada pharmacy
rybelsus weight loss
order semaglutide
semaglutide tab 7mg
rybelsus for sale
rybelsus pill form
zovirax 30mg
wegovy online prescription
purchase rybelsus
semaglutide 3mg
buy wegovy online no script
ozempic tablets buy
generic rybelsus
generic wegovy
brand semaglutide
semaglutide tablets
semaglutide where to buy
semaglutide where to buy
wegovy where to buy
purchase wegovy
wegovy over the counter
buy rybelsus
wegovy 3 mg tablet
ozempic tab 3mg
rybelsus online uk
ozempic tablets cost
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
buy semaglutide from canada
buy wegovy
semaglutide tablets cost
wegovy best price
buy semaglutide oversees
semaglutide buy online
buy rybelsus online no script
LГҐn 90000 kr
rybelsus 3 mg tablet
rybelsus online order
wegovy 3 mg tablet
lese
semaglutide weight loss
wegovy 21 mg
wegovy online prescription
rybelsus medicine
buy semaglutide canada
semaglutide online uk
semaglutide online
buy ozempic from canada online
rybelsus online pharmacy
semaglutide online prescription
buy semaglutide
where to buy semaglutide
buy semaglutide cheap
rybelsus 3 mg
wegovy
buy semaglutide
brand viagra 100mg price
semaglutide for weight loss without diabetes
rybelsus online pharmacy
where to buy semaglutide online
best time of day to take metformin
wegovy buy from canada
buy ozempic from canada online
rybelsus prescription
buy semaglutide online canada
wegovy lose weight
rybelsus prescription
generic ozempic for weight loss
generic semaglutide
semaglutide canada
wegovy 21 mg
purchase rybelsus
semaglutide rx
buy semaglutide
zithromax generic
semaglutide diabetes
semaglutide injections
buy ozempic online from india
semaglutide tab 3mg
wegovy semaglutide
cheap semaglutide
semaglutide uk
daddy casino pr4a.ru
rybelsus 3 mg
buy wegovy
rybelsus 7mg
semaglutide online prescription
semaglutide 3 mg
izzzi casino
izzzi casino
buy rybelsus in mexico
buy ozempic cheap
rybelsus buy australia
wegovy order
rybelsus sale
wegovy without prescription
joycasino бонус код
joycasino бонус код
rybelsus tablets cost
rybelsus 7 mg tablet
wegovy tablets for weight loss
казино клубника
казино клубника
buy wegovy
semaglutide where to buy
rybelsus for diabetes
rybelsus buy online
rybelsus buy online
semaglutide medication
semaglutide canada
buy ozempic oversees
wegovy diabetes medication
rybelsus without prescription
казино волна
https://groups.google.com/g/topgamersonline/c/PMlG9sqvQGM
semaglutide over the counter
ozempic tablets for weight loss cost
8000 euro lenen
cheap differin 15g
buy wegovy in mexico
rybelsus australia online
казино восток
казино восток
buy ozempic from india
semaglutide injection
wegovy tablets 7 mg
ozempic pill form
rybelsus 14mg
lucky nugget casino
https://groups.google.com/g/topgamersonline/c/SRj6g3_VvA8
ozempic tab 3mg
rybelsus tab 3mg
where can i buy semaglutide
buy semaglutide
order rybelsus online
https://ipbhelp.ru
[url=http://rybelsustabs.shop/]rybelsus[/url]
[url=http://rybelsus.quest/]wegovy tablets for weight loss cost[/url]
semaglutide uk
Gama Casino официальный сайт
generic ozempic for weight loss
buy ozempic online from india
semaglutide cost
wegovy tablets buy
wegovy pills
metformin anti aging
wegovy rx
semaglutide online pharmacy
daddy casino
wegovy canada pharmacy prices
semaglutide online uk
besök
abilify cost
semaglutide from canada
buy wegovy
ozempic for weight loss
https://ipbhelp.ru
[url=http://rybelsus.guru/]wegovy online order[/url]
semaglutide australia
ozempic for weight loss
rybelsus tab 3mg
rybelsus pills
brand semaglutide
generic rybelsus
buy semaglutide in canada
You have noted very interesting points! ps decent web site. “Formal education will make you a living self-education will make you a fortune.” by Jim Rohn.
semaglutide pills
semaglutide 3 mg tablet
how long before lisinopril works
pill furosemide
http://www.marieltour.ru
buy wegovy from canada
buy ozempic online cheap
where to buy semaglutide online
semaglutide online order
rybelsus online uk
wegovy injection
semaglutide generic
semaglutide diabetes medication
buy ozempic online cheap
zofran united states
daddy casino зеркало
Промокод без отыгрыша
Промокод без отыгрыша
can you take ibuprofen with zoloft
wegovy medicine
klik voor meer informatie
alternative to flagyl
semaglutide online uk
rybelsus buy australia
rybelsus canada
buy rybelsus
rybelsus 21 mg
semaglutide retail price
wegovy 14
order semaglutide online
200 mg gabapentin cat
lasix to torsemide
purchase semaglutide
ozempic semaglutide tablets cost
valtrex prices
wegovy semaglutide tablets 7.5 mcg
cheap ozempic
semaglutide cost
En ugyun fiyata IQOS heets satın al aynı gün kargo imkanı.
glucophage lantus
rybelsus canada pharmacy
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
This is my first time pay a quick visit at here and i am really happy to read everthing at one place
wegovy prescription
order semaglutide
buy wegovy
will cephalexin treat strep
rybelsus
zithromax while pregnant
purchase ozempic
rybelsus xr
wegovy canada pharmacy
buy rybelsus online no script
semaglutide generic cost
rybelsus tablets cost
augmentin 250/125mg no prescription
wegovy tablets cost
gabapentin and tramadol
ozempic pill form
Hi! I’m at work surfing around your blog from my new iphone 4! Just wanted to say I love reading your blog and look forward to all your posts! Keep up the fantastic work!
rybelsus mexico
wegovy canada pharmacy
buy wegovy online no script needed
ozempic semaglutide
buy wegovy online no script needed
how often to take amoxicillin
semaglutide online cheap
I am truly thankful to the owner of this web site who has shared this fantastic piece of writing at at this place.
buy ozempic from india
buy semaglutide
semaglutide tablets cost
rybelsus retail price
wegovy tab 14mg
escitalopram and thc
ozempic for weight loss
wegovy semaglutide tablets cost
wegovy semaglutide
you can find out more
buy wegovy from canada
semaglutide tablets cost
ozempic pill form
semaglutide for weight loss without diabetes
ozempic semaglutide tablets 3mg
buy ozempic online canada
wegovy
semaglutide australia online
semaglutide medicine
rybelsus pill
wegovy australia online
can you take cephalexin if you are allergic to penicillin
semaglutide online
wegovy 3 mg tablet
rybelsus prescription
buy wegovy from canada
wegovy tablets cost
semaglutide online pharmacy
generic ozempic for weight loss
rybelsus for sale
ozempic online
ciprofloxacin drops
rybelsus diabetes medication
Vozol gear 10000 siparişleriniz aynı gün kapınızda.
buy rybelsus online no script needed
rybelsus pill form
generic semaglutide for weight loss
where can i buy ozempic
semaglutide tab 7mg
rybelsus from canada
rybelsus 14mg tablets
semaglutide online cheap
läs mer här
cephalexin behavioral side effects
generic semaglutide
buy ozempic online from india
LГҐn 3000 kr
semaglutide cost
ozempic tab 14mg
wegovy 3 mg
wegovy buy online
wegovy online pharmacy
buy wegovy canada
rybelsus semaglutide tablets 3mg
rybelsus semaglutide tablets
semaglutide pills
buy rybelsus online no script
semaglutide sale
rybelsus diabetes medication
rybelsus tab 7mg
semaglutide diabetes medication
buy semaglutide from canada online
rybelsus tablets for weight loss
ozempic
semaglutide online uk
semaglutide cost
wegovy retail price
wegovy best price
where to buy ozempic online
robaxin 500mg
semaglutide diabetes
flibanserin 500mg
wegovy xr
rybelsus diabetes
rybelsus generic cost
wegovy prescription
ozempic coupon
cephalexin for cellulitis
ozempic semaglutide tablets cost
rybelsus medication
bactrim for uti 3 days
generic wegovy for weight loss
İstanbul Bölgesinde bulunan en iyi masaj salonları ve masaj hizmetini arıyorsan doğru adrestesin diyebiliriz çünkü sizler için en iyi terapistleri ile masaj merkezlerini araştırır ve sadece en iyilerini sitesinde yayınlar.
semaglutide 3 mg tablet
semaglutide for sale
rybelsus xl
wegovy semaglutide tablets 7.5 mcg
rybelsus for diabetes
generic wegovy cost
18000 Euro Kredit
buy ozempic online canada
where to buy ozempic online
rybelsus pill form
semaglutide tab 3mg
rybelsus semaglutide tablets 3mg
semaglutide diabetes
synthroid 05 mg
buy valtrex australia
lisinopril 120 mg
valtrex tablets uk
metformin 93
zithromax no rx
canadianpharmacymeds com
prinivil lisinopril
purchase generic cialis online
klick
zithromax online canada
1000 Euro Kredit
buying synthroid in mexico
cost for valtrex
foreign pharmacy online
valtrex over the counter australia
lisinopril 25 mg price
tadalafil cheap online
В городе Москве купить аттестат – это удобный и быстрый способ достать нужный запись без избыточных проблем. Большое количество компаний предоставляют помощь по изготовлению и реализации дипломов разных образовательных учреждений – https://russkiy-diploms-srednee.com/. Ассортимент свидетельств в городе Москве огромен, включая бумаги о высшем и среднем образовании, аттестаты, дипломы техникумов и вузов. Главное достоинство – способность достать свидетельство подлинный документ, обеспечивающий достоверность и высокое стандарт. Это обеспечивает специальная защита ото подделки и позволяет применять свидетельство для различных нужд. Таким образом, заказ аттестата в городе Москве является надежным и эффективным вариантом для данных, кто хочет достичь успеха в трудовой деятельности.
canadian pharmacy prices
legit canadian pharmacy
synthroid 137 mcg cost
lisinopril generic 20 mg
neurontin dose
prinivil coupon
lisinopril 20 12.5 mg
happy family store rx
generic tadalafil no prescription
azithromycin india
tadalafil pills in india
zithromax over the counter canada
gold pharmacy online
generic cialis us pharmacy
escitalopram oxalate side effects
10000 Euro Kredit
tadalafil 20 mg buy
proventil 300mg
prednisone 5 mg tablet without a prescription
prednisone 5084
no prescription lisinopril
prednisone for sale no prescription
price of lisinopril
zestoretic
buy synthroid cheap
synthroid 137 mg
tadalafil tablets 20 mg price in india
levothyroxine ordering online
tadalafil tablet 10mg price in india
valtrex script
zestril tablet price
professional pharmacy
prednisone 60
prescription prednisone cream
lisinopril online prescription
LГҐn 20000 kr
reputable indian online pharmacy
100 mg tadalafil
valtrex cream price
can i buy prednisone online in uk
generic synthroid online
metformin canada online
synthroid 250 mcg price
how to get cialis without prescription
wellbutrin
lisinopril 20mg buy
online med pharmacy
citalopram 10 mg tablet
synthroid 50mcg coupon
glucophage 500 australia
depakote price
purchase lisinopril online
90000 euro lenen
generic valtrex no prescription
ddavp intranasal dose
synthroid 0.112 mg
lisinopril 60 mg
synthroid 50
cozaar generic walmart
prinivil coupon
liГ© ici
cialis canada fast shipping
synthroid 88 brand prices
canadian pharmacy world coupon
lowest price for synthroid
lisinopril online canadian pharmacy
tadalafil coupon discount
cialis canada online pharmacy
happy family store pharmacy
zestoretic 20
175 mg synthroid
metformin 125 mg
medicine valtrex
safe reliable canadian pharmacy
seroquel cheap
tadalafil 2.5 mg daily
canadian world pharmacy
40 mg prednisone daily
lisinopril generic 10 mg
tadalafil daily 5mg
40 mg lisinopril
metformin hcl 500 mg
online otc pharmacy
lisinopril 12.5 tablet
lisinopril 40 mg for sale
buy prednisone tablets uk
happy family store
online pharmacy pain
where can i buy synthroid online
how do i get cialis
cost of lisinopril 10 mg
metforfim without a prescription
real metformin mexico
synthroid best price
cheapest price for tadalafil
where to buy tadalafil in usa
zestril price
price of synthroid in canada
floxin 20mg
top mail order pharmacies
drugstore com online pharmacy prescription drugs
prednisone no rx
buy valtrex canada
online drugs valtrex
zestoretic
legitimate canadian pharmacies
depakote vs valproic acid
happy family drug store
gabapentin and citalopram
online pharmacy no rx
medication lisinopril 10 mg
zestril 40
where to buy cialis without a prescription
metformin pill
canadian pharmacy discount coupon
online cialis in canada
This is really interesting, You’re a very skilled blogger. I’ve joined your feed and look forward to seeking more of your magnificent post. Also, I’ve shared your site in my social networks!
cheapest price for generic cialis
lisinopril 5 mg tabs
canadian pharmacy 24h com
buy valtrex canada
cozaar medication
drugstore com online pharmacy prescription drugs
LГҐn 6000
zithromax capsules 500mg
canadianpharmacymeds com
best pharmacy
ddavp fever
buspar 2.5mg
prinivil coupon
tadalafil 5mg price in india
buy azithromycin 250 online
buying prednixone for animals
price of 100mcg synthroid
happy family canadian pharmacy
azithromycin tabs 500mg
where can you buy prednisone
buy online pharmacy uk
tadalafil medicine
buy cheap generic cialis
buy prednisone without prescription
lisinopril 10 mg without prescription
credit 9000 euros
lisinopril 40 mg
buy synthroid no prescription
synthroid 125 mcg coupon
happy family store
buy metformin on line
diclofenac sodium topical gel 1
tadalafil online in india
valtrex 500mg price
buying valtrex online uk
ezetimibe wiki
blog qui
usa pharmacy
synthroid 0.075
american online pharmacy
40 mg lisinopril
15000 Euro Kredit
valtrex 1g price
pharmacy order online
rx 535 lisinopril 40 mg
lisinopril 20 12.5 mg
fish augmentin
best no prescription pharmacy
4 azithromycin cream
buy valtrex online no prescription
synthroid prescription
happy family store pharmacy
diltiazem moa
online pharmacy 365
how much is lisinopril 20 mg
azithromycin capsules 500mg
lowest cost generic cialis
how much is lisinopril 40 mg
legitimate canadian mail order pharmacy
flexeril and alcohol
tadalafil tablets 20 mg buy
buy generic tadalafil
valtrex over the counter australia
should i increase my effexor dose
generic valtrex for sale
valtrex costs canada
dozvД›dД›t se vГce
LГҐna 40000 kr
lisinopril 100 mg
canadian pharmacy 24h com safe
northwestpharmacy
metformin 60 tablets 1000 mg
flomax breathing
lisinopril 40 mg cost
cialis online 40 mg
happy family online pharmacy
where to buy lisinopril online
lisinopril average cost
2870 valtrex
tadalafil uk pharmacy
synthroid 0.100 mg
synthroid 25 pill
over the counter valtrex medication
synthroid 0.125
order cialis pills
compare pharmacy prices
what does aripiprazole do
a nurse is providing teaching to a client who has gout and a new prescription for allopurinol
us pharmacy
prednisone 10mg pack
prednisone cream over the counter
drug azithromycin 500 mg
tadalafil tab 10mg
tadalafil generic 10mg
synthroid cost comparison
prednisolone prednisone
LГҐna 2000 kr
synthroid price in india
zithromax 250 mg cost
lisinopril 10 mg cost
lisinopril 20 mg buy
lisinopril for sale uk
tadalafil 5 mg mexico
glucophage tab 1000mg
prednisone 5mg cost
prednisone canadian pharmacy
canada drug pharmacy
lees dit bericht hier
zestoretic 10 12.5 mg
lisinopril from mexico
mexican online mail order pharmacy
steroid prednisone
amitriptyline pill
can you buy valtrex over the counter in australia
azithromycin 100 mg sale
long term use of aspirin icd 10
can you buy azithromycin over the counter
valtrex medicine price
where to buy lisinopril without prescription
synthroid brand name coupon
buy baclofen online
drug metformin
synthroid prescription cost
celebrex generic name
200 mg cialis
Казино с выводом на Киви
cost of synthroid 75 mcg
https://zelpgo.ru
cost of synthroid 200 mcg
isoptin sr
canadian pharmacy happy family store
synthroid 137 mcg
levaquin coupon
16 lisinopril
canada cialis 20 mg
6000 mg metformin
generic cialis for sale online
https://mfc-sosnovskoe.ru
canadian pharmacy generic tadalafil
how does bupropion make you feel
price of cialis daily
synthroid 137 mcg tab
augmentin doses for sinusitis
how much is lisinopril
synthroid coupon
valtrex 550 mg
www canadianonlinepharmacy
lisinopril 10 mg
online pharmacy without insurance
prednisone 8 pills
cialis europe
credito 7500 euros
synthroid 112 mcg india
celecoxib generic for celebrex
lisinopril 2019
how much is cialis
lisinopril 20 mg price online
canadian pharmacy 24
prednisone in mexico
30 mg tadalafil
drug lisinopril
208 lisinopril
regarde ce site web
online shopping pharmacy india
reliable canadian online pharmacy
pharmacy website india
drug lisinopril 5 mg
valtrex tablets over the counter
canadian pharmaceutical prices
metformin online canada
lisinopril tab 20mg cost
canadian metformin
metformin 2019
LГҐna 7000 kr
lisinopril 10 mg cost
buspar half life
canadian online pharmacy no prescription
canada drugstore pharmacy rx
celexa 40mg
buying tadalafil in mexico
20 mg prednisone daily
how to buy lisinopril online
cialis buy australia
canadian pharmacies not requiring prescription
cialis best price australia
metformin 500 mg tablet price in india
lisinopril 2.5 mg cost
chloromycetin 50mg
canadian pharmacy com
where can you get synthroid
LГҐn 30000
canadian pharmacy antibiotics
can you buy valtrex over the counter in australia
valtrex 500 mg cost
buy cialis online in india
lisinopril pills 10 mg
generic for prinivil
canadian pharmacy coupon
foreign online pharmacy
canadian pharmacy service
medication canadian pharmacy
synthroid cheapest price
tadalafil soft gel capsule 20mg
synthroid 0.112 mg
best canadian pharmacy no prescription
buy generic tadalafil 20mg
lisinopril 125 mg
prednisone 12 mg
online pharmacy bc
order prednisone online canada
glucophage 500mg
LГҐn 6000
valtrex online purchase
lisinopril price without insurance
prednisone pak
synthroid 0.112 mcg
tadalafil daily
buy synthroid 150 mcg
legit mexican pharmacy
medication canadian pharmacy
medication lisinopril 20 mg
how to get azithromycin without prescription
lisinopril discount
pharmacy express
Lainaa 500000 euroa
happy family medicine
buy azithromycin 500mg online uk
discount pharmacy online
azithromycin cheap online
average price of cialis 20mg
online pharmacy birth control pills
valtrex cost australia
can i buy valtrex over the counter
zestoretic 20 25mg
tadalafil prices canada
thewiin.com
Hongzhi 황제는 자신감이 넘치는 것을보고 죄책감을 느꼈습니다.
medical pharmacy south
lisinopril 5mg tab
tadalafil 7.5 mg
zestril 2.5 mg tablets
internet pharmacy mexico
my canadian pharmacy
valtrex rx cost
actos compromisorios
synthroid over the counter online
semaglutide stomach pain
synthroid 100 mcg tablet
valtrex 1000 mg cost
where to buy lisinopril
happy family drugstore
buy synthroid cheap
mene tänne
8000 Euro Kredit
lisinopril 80
cialis 40mg
tadalafil 20mg online canada
pharmaceuticals online australia
tadalafil 15mg
zithromax cost in mexico
top online pharmacy 247
buying from canadian pharmacies
valtrex pill
https://zelpgo.ru
synthroid prescription
brand name valtrex cost
azithromycin online pharmacy uk
lisinopril 3760
valtrex purchase online
acarbose emc
how to lose weight on abilify
online pharmacy synthroid
lisinopril 20 mg no prescription
cialis tablets 20mg for sale
metformin south africa
tadalafil over the counter canada
cost for 40 mg lisinopril
lisinopril 10 mg online
80000 euro lenen
prednisone tablets canada
happy family store.net
60 mg prednisone
which pharmacy is cheaper
valtrex generic brand
can i buy prednisone over the counter
valtrex buy online
cialis 20mg best price
buy synthroid online from canada
cardizem 500mg
canada pharmacy coupon
metformin 825 mg
prednisone 2.5 tablet
how to buy prednisone online
best online foreign pharmacy
prednisone online
protonix indications
muscle relaxer robaxin
synthroid 125 mcg coupon
how much is 30 lisinopril
lisinopril 5mg
remeron sleep aid
lisinopril 20 mg canadian
where can i get synthroid
southern pharmacy
Payday loans in Mississippi
azithromycin 500 mg tablet online
synthroid 112 mcg
synthroid pharmacy price
lisinopril 2.5 mg buy online
75 mcg synthroid no prescription
zestril 2.5 mg
price of azithromycin 250 mg
metformin 550 mg
cialis for daily use price
buy metformin usa
cheap valtrex
repaglinide fk
what are the side effects of protonix
synthroid 200 mg
price of tadalafil
7000 Euro Kredit
synthroid 200 mcg tabs
Казино с пополнением с Киви
metformin without presription
valtrex pharmacy
lisinopril tablets india
tadalafil soft gel capsule 20mg
buy metformin uk online
how much is zithromax 500mg
tadalafil online order
medicine azithromycin 500 mg
cialis india generic
cross border pharmacy canada
order cialis canada
buy zestril
tadalafil soft gel
where to buy remeron 15mg
lisinopril cost
buy generic tadalafil
canadian pharmacy
price of zestril
differin without a doctor prescription
buy prednisone online from mexico
cost of metformin uk
azithromycin without prescription online
prednisone 10 mg price
azithromycin 100mg tablet
medication zestoretic
reliable online pharmacy
prednisone 60 mg price
pharmacy website
tadalafil 10 mg coupon
prednisone online sale
tadalafil cialis
canadian pharmacy 365
valtrex medication
tadalafil tablets 20 mg online india
cheapest pharmacy canada
lisinopril online
legal online pharmacies in the us
50000 euro lenen
azithromycin order canada
lisinopril 5
canadian family pharmacy
pharmacy mall
canadian pharmacy mall
prednisone uk buy
zithromax prescription
lisinopril online without a prescription
tadalafil united states
zestril 5 mg tablets
robaxin 750 mg side effects
can you buy valtrex over the counter
tadalafil 20mg canada
synthroid brand price
synthroid price india
prescription cialis prices
online pharmacy delivery dubai
metformin 52080
buy generic cialis online
buy prednisone online uk
tadalafil 80mg
kerendia vs spironolactone
synthroid elderly
how much is metformin
5094 prednisone
brazilian pharmacy online
happy family store rx
purchase lisinopril 40 mg
buy synthyroid online
buy tadalafil 100mg
tadalafil from india to usa
no prescription prednisone
metformin canada over the counter
canadian pharmacies comparison
where to buy glucophage online
pharmacy com canada
trusted canadian pharmacy
https://mfc-sosnovskoe.ru
synthroid 50 mcg tablet
zestril 25 mg
zamiennik sitagliptin
Казино с выводом на Qiwi
valtrex prescription cost
prednisone canada pharmacy
canadian pharmacies comparison
where to buy valtrex generic
synthroid 100 mcg price
buy cialis usa
indian pharmacy paypal
how to get prednisone without a prescription
synthroid
lisinopril 20 mg no prescription
valtrex 500mg uk
aldactone 100 mg cost
zestoretic 10 12.5
lisinopril 20mg buy
medicine valtrex
synthroid overstimulation
valtrex generic canada
international pharmacy no prescription
can i buy cialis online in canada
synthroid brand price